Abstract
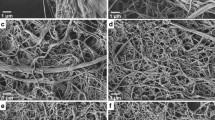
Periodate–chlorite oxidized bleached hardwood kraft pulp fibre samples with six levels of charge densities ranging from 0.5 to 1.8 mmol/g were gradually disintegrated to microfibrils using a high-shear homogenizer. The disintegration kinetics and mechanisms were studied by a flow fractionation method, and the properties of the resulting particles were determined using low shear viscosity and transmittance measurements. The particles formed during the disintegration were visualized with a charge-coupled device camera and by field-emission scanning electron microscopy. The result showed that cellulose fibres with a low charge density disintegrated at a low rate and produced ragged fibres and bunches of microfibrils via bursting of the fibre walls, whereas those with a higher charge density broke down at a high rate and microfibrils were formed through swelling and the creation of balloon structures. A carboxyl content of 1.2 mmol/g was found to be the threshold value for the efficient formation of high aspect ratio microfibrils and also for the change in the disintegration mechanism in the high-shear homogenizer.








Similar content being viewed by others
References
Ankerfors M, Lindström T (2009) Method for providing a nanocellulose involving modifying cellulose fibers. Pat. WO 2009126106
Araki J, Wada M, Kuga S (2001) Steric stabilization of a cellulose microcrystal suspension by poly(ethylene glycol) grafting. Langmuir 17:21–27. doi:10.1021/la001070m
Besbes I, Alila S, Boufi S (2011) Nanofibrillated cellulose from TEMPO-oxidized eucalyptus fibres: effect of the carboxyl content. Carbohydr Polym 84:975–983. doi:10.1016/j.carbpol.2010.12.052
Cuissinat C, Navard P (2006) Swelling and dissolution of cellulose part 1: free floating cotton and wood fibres in N-methylmorpholine-N-oxide–water mixtures. Macromol Symp 244:1–18. doi:10.1002/masy.200651201
Dash R, Elder T, Ragauskas AJ (2012) Grafting of model primary amine compounds to cellulose nanowhiskers through periodate oxidation. Cellulose 19:2069–2079. doi:10.1007/s10570-012-9769-2
Gehmayr V, Potthast A, Sixta H (2012) Reactivity of dissolving pulps modified by TEMPO-mediated oxidation. Cellulose 19:1125–1134. doi:10.1007/s10570-012-9729-x
Haapala A, Laitinen O, Karinkanta P et al (2013) Optical characterisation of size, shape and fibrillarity from microfibrillar and microcrystalline cellulose, and fine ground wood powder fractions. Appita J 66:331–339
Henriksson M, Henriksson G, Berglund LA, Lindström T (2007) An environmentally friendly method for enzyme-assisted preparation of microfibrillated cellulose (MFC) nanofibers. Eur Polym J 43:3434–3441. doi:10.1016/j.eurpolymj.2007.05.038
Hoch C (1950) Degradation of cellulose as revealed microscopically. Text Res J 20:141–151
Iotti M, Gregersen ØW, Moe S, Lenes M (2011) Rheological studies of microfibrillar cellulose water dispersions. J Polym Environ 19:137–145. doi:10.1007/s10924-010-0248-2
Iwamoto S, Abe K, Yano H (2008) The effect of hemicelluloses on wood pulp nanofibrillation and nanofiber network characteristics. Biomacromolecules 9:1022–1026. doi:10.1021/bm701157n
Jonoobi M, Mathew AP, Oksman K (2012) Producing low-cost cellulose nanofiber from sludge as new source of raw materials. Ind Crops Prod 40:232–238. doi:10.1016/j.indcrop.2012.03.018
Katz S, Beatson R, Scallan A (1984) The determination of strong and weak acidic groups in sulfite pulps. Sven Papperstidning 87:48–53
Kekäläinen K, Illikainen M, Niinimäki J (2012) Morphological changes in never-dried kraft fibers under mechanical shearing. Cellulose 19:879–889. doi:10.1007/s10570-012-9670-z
Kekäläinen K, Liimatainen H, Illikainen M et al (2014) The role of hornification in the disintegration behaviour of TEMPO-oxidized bleached hardwood fibres in a high-shear homogenizer. Cellulose 21:1163–1174. doi:10.1007/s10570-014-0210-x
Kim U-J, Kuga S (2001) Ion-exchange chromatography by dicarboxyl cellulose gel. J Chromatogr A 919:29–37. doi:10.1016/S0021-9673(01)00800-7
Kim U-J, Kuga S, Wada M et al (2000) Periodate oxidation of crystalline cellulose. Biomacromolecules 1:488–492. doi:10.1021/bm0000337
Klemm D, Kramer F, Moritz S et al (2011) Nanocelluloses: a new family of nature-based materials. Angew Chem Int Ed 50:5438–5466. doi:10.1002/anie.201001273
Laitinen O (2011) Utilization of tube flow fractionation method in fibre and particle analysis. Dissertation, University of Oulu
Laitinen O, Niinimäki J (2014) Fractional study of the microfibrillated cellulose. Tappi J (in press)
Larsson PA, Gimåker M, Wågberg L (2008) The influence of periodate oxidation on the moisture sorptivity and dimensional stability of paper. Cellulose 15:837–847. doi:10.1007/s10570-008-9243-3
Larsson PA, Berglund LA, Wågberg L (2014) Highly ductile fibres and sheets by core-shell structuring of the cellulose nanofibrils. Cellulose 21:323–333. doi:10.1007/s10570-013-0099-9
Lasseuguette E, Roux D, Nishiyama Y (2008) Rheological properties of microfibrillar suspension of TEMPO-oxidized pulp. Cellulose 15:425–433. doi:10.1007/s10570-007-9184-2
Liimatainen H, Visanko M, Sirviö JA et al (2012) Enhancement of the nanofibrillation of wood cellulose through sequential periodate–chlorite oxidation. Biomacromolecules 13:1592–1597. doi:10.1021/bm300319m
Liimatainen H, Ezekiel N, Sliz R et al (2013a) High-strength nanocellulose–TALC hybrid barrier films. ACS Appl Mater Interfaces 5:13412–13418. doi:10.1021/am4043273
Liimatainen H, Sirviö J, Pajari H et al (2013b) Regeneration and recycling of aqueous periodate solution in dialdehyde cellulose production. J Wood Chem Technol 33:258–266. doi:10.1080/02773813.2013.783076
Mishra SP, Manent A-S, Chabot B, Daneault C (2012) Production of nanocellulose from native cellulose—various options utilizing ultrasound. Bioresources 7:422–436
Moigne N, Bikard J, Navard P (2010) Rotation and contraction of native and regenerated cellulose fibers upon swelling and dissolution: the role of morphological and stress unbalances. Cellulose 17:507–519. doi:10.1007/s10570-009-9395-9
Pääkkö M, Ankerfors M, Kosonen H et al (2007) Enzymatic hydrolysis combined with mechanical shearing and high-pressure homogenization for nanoscale cellulose fibrils and strong gels. Biomacromolecules 8:1934–1941. doi:10.1021/bm061215p
Pöhler T, Lappalainen T, Tammelin T et al (2010) Influence of fibrillation method on the character of nanofibrillated cellulose (NFC). In: TAPPI international conference on nanotechnology for the forest product industry, 27–29 September, Dipoli Congress Centre, Espoo, Finland
Potthast A, Kostic M, Schiehser S et al (2007) Studies on oxidative modifications of cellulose in the periodate system: molecular weight distribution and carbonyl group profiles. Holzforschung 61:662–667. doi:10.1515/HF.2007.099
Potthast A, Schiehser S, Rosenau T, Kostic M (2009) Oxidative modifications of cellulose in the periodate system—reduction and beta-elimination reactions. Holzforschung 63:12–17. doi:10.1515/HF.2009.108
Qua EH, Hornsby PR, Sharma HSS, Lyons G (2011) Preparation and characterisation of cellulose nanofibres. J Mater Sci 46:6029–6045. doi:10.1007/s10853-011-5565-x
Saito T, Nishiyama Y, Putaux J-L et al (2006) Homogeneous suspensions of individualized microfibrils from TEMPO-catalyzed oxidation of native cellulose. Biomacromolecules 7:1687–1691. doi:10.1021/bm060154s
Saito T, Kimura S, Nishiyama Y, Isogai A (2007) Cellulose nanofibers prepared by TEMPO-mediated oxidation of native cellulose. Biomacromolecules 8:2485–2491. doi:10.1021/bm0703970
Saito T, Hirota M, Tamura N et al (2009) Individualization of nano-sized plant cellulose fibrils by direct surface carboxylation using TEMPO catalyst under neutral conditions. Biomacromolecules 10:1992–1996. doi:10.1021/bm900414t
Sirviö J, Hyväkkö U, Liimatainen H et al (2011) Periodate oxidation of cellulose at elevated temperatures using metal salts as cellulose activators. Carbohydr Polym 83:1293–1297. doi:10.1016/j.carbpol.2010.09.036
Sneck A (2011) New approach to classification of cellulose fibrils and suitable methods for their characterization. In: TAPPI international conference on nanotechnology for renewable materials, Sheraton Crystal City, Washington, DC
Syverud K, Chinga-Carrasco G, Toledo J, Toledo PG (2011) A comparative study of Eucalyptus and Pinus radiata pulp fibres as raw materials for production of cellulose nanofibrils. Carbohydr Polym 84:1033–1038. doi:10.1016/j.carbpol.2010.12.066
Tejado A, Alam MN, Antal M et al (2012) Energy requirements for the disintegration of cellulose fibers into cellulose nanofibers. Cellulose 19:831–842. doi:10.1007/s10570-012-9694-4
Uetani K, Yano H (2011) Nanofibrillation of wood pulp using a high-speed blender. Biomacromolecules 12:348–353
Varma AJ, Chavan VB (1995) A study of crystallinity changes in oxidised celluloses. Polym Degrad Stab 49:245–250. doi:10.1016/0141-3910(95)87006-7
Varma AJ, Kulkarni MP (2002) Oxidation of cellulose under controlled conditions. Polym Degrad Stab 77:25–27
Wågberg L, Decher G, Norgren M et al (2008) The Build-up of polyelectrolyte multilayers of microfibrillated cellulose and cationic polyelectrolytes. Langmuir 24:784–795. doi:10.1021/la702481v
Yang H, Alam MN, Ven TGM (2013) Highly charged nanocrystalline cellulose and dicarboxylated cellulose from periodate and chlorite oxidized cellulose fibers. Cellulose 20:1865–1875. doi:10.1007/s10570-013-9966-7
Zhang J, Jiang N, Dang Z et al (2008) Oxidation and sulfonation of cellulosics. Cellulose 15:489–496. doi:10.1007/s10570-007-9193-1
Acknowledgments
The authors acknowledge the Graduate School in Chemical Engineering (GSCE), the Tauno Tönning Research Foundation, and the Finnish Foundation for Technology Promotion for their financial support. We also thank Mr. Jani Österlund and Mr. Samuli Honkaniemi for their help during the experiments. Metso Automation (Finland) is acknowledged for providing the technology used in the flow fractionation technique.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kekäläinen, K., Liimatainen, H. & Niinimäki, J. Disintegration of periodate–chlorite oxidized hardwood pulp fibres to cellulose microfibrils: kinetics and charge threshold. Cellulose 21, 3691–3700 (2014). https://doi.org/10.1007/s10570-014-0363-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-014-0363-7