Abstract
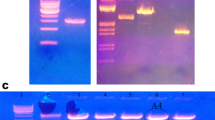
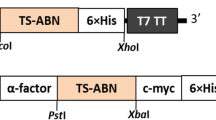
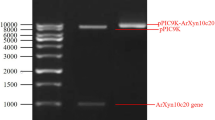
α-l-Arabinofuranosidases with an orchestral action of xylanolytic enzymes degrades the xylan in plant cell wall. In this study, heterologous expression, biochemical characterization, and synergistic action of α-l-Arabinofuranosidase from previously identified.Geobacillus vulcani GS90 (GvAbf) was investigated. The recombinant α-l-Arabinofuranosidase was overexpressed in Escherichia coli BL21 (λDE) and purified via His-tag Ni-affinity and size-exclusion chromatography. Optimum activity of the purified α-l-Arabinofuranosidase was obtained at pH 5 and at 70 °C. The GvAbf was active in a broad pH and temperature ranges; pH 4–9 and 30–90 °C, respectively. In addition, it retained most of its activity after an hour incubation at 70 °C and remained relatively stable at pH 3–6. GvAbf was quite stable against various metal ions. The kinetic parameters of GvAbf was obtained as Vmax and Km; 200 U/mg and 0.2 mM with p-nitrophenyl-α-l-arabinofuranoside and 526 U/mg and 0.1 mM with sugar beet arabinan, respectively. The synergistic action of GvAbf was studied with commercially available xylanase on juice enrichment of apples, grapes, oranges, and peaches. The best juice enrichment in terms of clarity, reducing sugar content, and yield, was achieved with GvAbf and xylanase together compared to treatment with xylanase and GvAbf alone in all fruits. The treatment with GvAbf and xylanase together lead to an increased juice yield by 26.56% (apple), 30.88% (grape), 40.00% (orange) and 32.20% (peach) as well as having a significant effect on juice clarity by an increase of % transmittance 47.26, 25.98, 41.77, and 44.97, respectively. The highest reducing sugar level of fruit juices also obtained with GvAbf and xylanase together compared to treatment with xylanase and GvAbf alone in all types of fruits. GvAbf and xylanase together as simultaneous synergistic manner may have an exciting potential for application in fruit juice processing.



Similar content being viewed by others
References
Taiz L, Zeiger E (2010) Plant physiology, 3rd edn. Sinauer Associates, Sunderland
Kaji A (1984) l-Arabinosidases. Chem Biochem 42:383–394
Hoffmam ZB, Oliveira LC, Cota J, Alvarez TM, Diogo JA, de Oliveira Neto M, Citadini APS, Leite VBP, Squina FM, Murakami MT, Ruller R (2013) Characterization of a hexameric exo-acting GH51 α-l-Arabinofuranosidase from the mesophilic Bacillus subtilis. Mol Biotechnol 55:260–267
Hashimoto T, Nakata Y (2003) Synergistic degradation of arabinoxylan with alpha-l-Arabinofuranosidase, xylanase and alpha-xylosidase from soy sauce koji mold, Aspergillus oryzae, in high salt condition. J Biosci Bioeng 95:164–169
Raweesri P, Riangrungrojana P, Pinphanichakarn P (2008) α-l-Arabinofuranosidase from Streptomyces sp. PC22: purification, characterization and its synergistic action with xylanolytic enzymes in the degradation of xylan and agricultural residues. Biores Technol 99:8981–8986
Yang W, Bai Y, Yang P, Luo H, Huang H, Meng K, Shi P, Wang Y, Yao B (2015) A novel bifunctional GH51 exo-α-l-Arabinofuranosidase/endo-xylanase from Alicyclobacillus sp. A4 with significant biomass-degrading capacity. Biotechnol Biofuels 8:1–11
Park J-M, Jang M-U, Oh GW, Lee E-H, Kang J-H, Song Y-B, Han NS, Kim T-J (2015) Synergistic action modes of arabinan degradation by exo- and endo-arabinosyl hydrolases. J Microbiol Biotechnol 25:227–233
Bouraoui H, Desrousseaux M-L, Ioannou E, Alvira P, Manaï M, Rémond C, Dumon C, Fernandez‑Fuentes N, O’Donohue MJ (2016) The GH51 α-l-Arabinofuranosidase from Paenibacillus sp. THS1 is multifunctional, hydrolyzing main-chain and side-chain glycosidic bonds in heteroxylans. Biotechnol Biofuels 9:1–14
Aryan A-P, Wilson B, Strauss C-R, Williams P-J (1987) The properties of glycosidases of Vitis vinifera and a comparison of their β-glucosidase activity with that of exogenous enzymes: an assessment of possible applications in enology. Am J Enol Vitic 38:182–188
Saha B (2000) Alpha-l-Arabinofuranosidases: biochemistry, molecular biology and application in biotechnology. Biotechnol Adv 18:403–423
Campbell GL, Bedford MR (1992) Enzyme applications for monogastric feeds: a review. Can J Anim Sci 72:449–466
Viikari L, Kantelinen A, Sundquist J, Linko M (1994) Xylanases in bleaching from an idea to the industry. FEMS Microbiol Rev 13:335–350
Numan M, Bhosle N (2006) Alpha-l-Arabinofuranosidases: the potential applications in biotechnology. J Ind Microbiol Biotechnol 33:247–260
Himmel ME, Ding SY, Johnson DK, Adney WS, Nimlos MR, Brady JW, Foust TD (2007) Biomass recalcitrance: engineering plants and enzymes for biofuels production. Science 315:804–807
Nawel B, Saida B, Estelle C, Hakima H, Duchironb F (2011) Production and partial characterization of xylanase produced by Jonesia denitrificans isolated in Algerian soil. Process Biochem 46:519–525
Makkonen HP, Nakas JP (2005) Use of xylanase and Arabinofuranosidase for arabinose removal from unbleached kraft pulp. Biotechnol Lett 27:1675–1679
Adigüzel AO, Tunçer M (2016) Production, characterization and application of a xylanase from Streptomyces sp. AOA40 in fruit juice and bakery industries. J Food Biotechnol 30:189–218
Rosmine E, Sainjan NC, Silvester R, Alikkunju A, Varghese SA (2017) Statistical optimisation of xylanase production by estuarine Streptomyces sp. and its application in clarification of fruit juice. J Genet Eng Biotechnol 15:393–401
Lee RC, Hrmova M, Burton RA, Lahnstein J, Fincher GB (2003) Bifunctional family 3 glycoside hydrolases from barley with alpha-lArabinofuranosidase and alpha-d-xylosidase activity: characterization, primary structures and COOH-terminal. J Biol Chem 278:5377–5387
Rahman AK, Kato K, Kawai S, Takamizawa K (2003) Substrate specificity of the alpha-l-Arabinofuranosidase from Rhizomucor pusillus HHT-1. Carbohydr Res 338:1469–1476
Debeche T, Cummings N, Connerton I, Debeire P, O’Donohue MJ (2000) Genetic and biochemical characterization of a highly thermostable alpha-l-Arabinofuranosidase from Thermobacillus xylanilyticus. Appl Environ Microb 66:1734–1736
Canakci S, Kacagan M, Inan K, Belduz AO, Saha BC (2008) Cloning, purification, and characterization of a thermostable alpha-l-Arabinofuranosidase from Anoxybacillus kestanbolensis AC26Sari. Appl Microbiol Biotechnol 81:61–68
Morozkina EV, Slutskaya ES, Fedorova TV, Tugay TI, Golubeva LI, Koroleva OV (2010) Extremophilic microorganisms: biochemical adaptation and biotechnological application (review). Appl Biochem Microbiol 46:1–14
Lee SH, Lee YE (2014) Cloning, expression, and characterization of a thermostable GH51 α-l-Arabinofuranosidase from Paenibacillus sp. DG-22. J Microbiol Biotechnol 24:236–244
Shi H, Ding H, Huang Y, Wang L, Zhang Y, Li X, Wang F (2014) Expression and characterization of a GH43 endoarabinanase from Thermotoga thermarum. BMC Biotechnol 14:1–9
Ahmed S, Luis AS, Bras JLA, Ghosh A, Gautam S, Gupta MN et al (2013) A novel α-l-Arabinofuranosidase of family 43 glycoside hydrolase (Ct43Araf) from Clostridium thermocellum. PLoS One 8(9):e73575
Wang W, Mai-Gisondi G, Stogios PJ, Kaur A, Xu X, Cui H, Turunen O, Savchenko A, Master ER (2014) Elucidation of the molecular basis for arabinoxylan-debranching activity of a thermostable family GH62 α-l-Arabinofuranosidase from Streptomyces thermoviolaceus. Appl Environ Microbiol 80:5317–5329
Kaur A-P, Nocek B-P, Xu X, Lowden M-J, Leyva J-F, Stogios P-J, Cui H, Leo R-D, Powlowski J, Tsang A, Savchenko A (2015) Functional and structural diversity in GH62 α-l-Arabinofuranosidases from the thermophilic fungus Scytalidium thermophilum. Microb Biotechnol 8:419–433
Bezalel L, Shoham Y, Rosenberg E (1993) Characterization and delignification activity of a thermostable α-l-Arabinofuranosidase from Bacillus Stearothermophilus. Appl Microbiol Biotechnol 40:57–62
Gilead S, Shoham Y (1995) Purification and characterization of α-l-Arabinofuranosidase from Bacillus stearothermophilus T-6. Appl Environ Microbiol 61:170–174
Canakci S, Belduz AO, Saha BC, Yasar A, Ayaz FA, Yayli N (2007) Purification and characterization of a highly thermostable α-l-Arabinofuranosidase from Geobacillus Caldoxylolyticus Tk4. Appl Microbiol Biotechnol 75:813–820
Sürmeli Y (2013) Investigation of alkaline and thermal stability of alpha-l-Arabinofuranosidase produced by directed evolution. Unpublished master thesis, İzmir Institute of Technology, İzmir
Yavuz E, Gunes H, Harsa S, Yenidunya AF (2004) Identification of extracellular enzyme producing thermophilic bacilli from Balcova (Agamemnon) geothermal site by ITS rDNA RFLP. J Appl Microbiol 97:810–817
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein–dye binding. Anal Biochem 72:248–251
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 3:426–428
Olfa E, Mondher M, Issam S, Ferid L, Nejib MM (2007) Induction, properties and application of xylanase activity from Sclerotinia Sclerotiorum S2 fungus. J Food Biochem 31:96–107
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729
Shallom D, Belakhov V, Solomon D, Shoham G, Baasov T, Shoham Y (2002) Detailed kinetic analysis and identification of the nucleophile in alpha-l-Arabinofuranosidase from Geobacillus Stearothermophilus T-6, a family 51 glycoside hydrolase. J Biol Chem 277:43667–43673
Lim Y-R, Yoon R-Y, Seo E-S, Kim Y-S, Park C-S, Oh D-K (2010) Hydrolytic properties of a thermostable α-l-Arabinofuranosidase from Caldicellulosiruptor saccharolyticus. J Appl Microbiol 109:1188–1197
Taylor EJ, Smith NL, Turkenburg JP, D’Souza S, Gilbert HJ, Davies GJ (2006) Structural insight into the ligand specificity of a thermostable family 51 Arabinofuranosidase, Araf51, from Clostridium thermocellum. Biochem J 395:31–37
Miyazaki K (2005) Hyperthermophilic alpha-l-Arabinofuranosidase from Thermotoga maritima Msb8: molecular cloning, gene expression, and characterization of the recombinant protein. Extremophiles 9:399–406
dos Santos C, Squina F, Navarro A, Oldiges D, Leme A, Ruller R et al (2011) Functional and biophysical characterization of a hyperthermostable GH51 α-l-Arabinofuranosidase from Thermotoga petrophila. Biotech Lett 33:131–137
Squina FM, Santos CR, Ribeiro DA, Cota J, de Oliveira RR, Ruller R, Mort A, Murakami MT, Prade RA (2010) Substrate cleavage pattern, biophysical characterization and low-resolution structure of a novel hyperthermostable arabinanase from Thermotoga petrophila. Biochem Biophys Res Commun 399:505–511
Sharma HP, Patel H, Sugandha (2017) Enzymatic added extraction and clarification of fruit juices—a review. Crit Rev Food Sci Nutr 57:1215–1227
Huang D, Liu J, Qi Y, Yang K, Xu Y, Feng L (2017) Synergistic hydrolysis of xylan using novel xylanases, β-xylosidases, and an α-l-Arabinofuranosidase from Geobacillus thermodenitrificans NG80-2. Appl Microbiol Biotechnol 101:6023–6037
Cakmak U, Ertunga NS (2016) Gene cloning, expression, immobilization and characterization of endo-xylanase from Geobacillus sp. TF16 and investigation of its industrial applications. J Mol Catal B Enzym 133:S288–S298
Nagar S, Mittal A, Gupta VK (2012) Enzymatic clarification of fruit juices (apple, pineapple, and tomato) using purified Bacillus pumilus SV-85S xylanase. Biotechnol Bioprocess Eng 17:1165–1175
Bajaj BK, Manhas K (2012) Production and characterization of xylanase from Bacillus licheniformis P11 (C) with potential for fruit juice and bakery industry. Biocatal Agric Biotechnol 4:330–337
Acknowledgements
The authors would like to thank Biotechnology and Bioengineering Research Center at İzmir Institute of Technology for the facilities and technical support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
İlgü, H., Sürmeli, Y. & Şanlı-Mohamed, G. A thermophilic α-l-Arabinofuranosidase from Geobacillus vulcani GS90: heterologous expression, biochemical characterization, and its synergistic action in fruit juice enrichment. Eur Food Res Technol 244, 1627–1636 (2018). https://doi.org/10.1007/s00217-018-3075-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00217-018-3075-7