Abstract
Key message
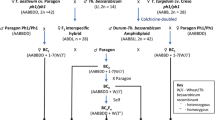
A variety of Thinopyrum bessarabicum introgressions in both hexaploid and tetraploid wheats were generated and characterized by molecular cytogenetic analysis. Six wheat-J genome recombinants were identified with ND-FISH and GISH.
Abstract
Diploid wheatgrass, Thinopyrum bessarabicum (2n = 2x = 14, EbEb or JbJb or JJ), is a well-known alien source of salinity tolerance and disease resistance for wheat improvement. The true genetic potential and effect of such introgressions into wheat can be best studied in chromosomal addition or substitution lines. Here, we report the generation and characterization of various categories of Th. bessarabicum derivatives in both hexaploid and tetraploid cultivated wheats. Sequential non-denaturing fluorescence in situ hybridization (ND-FISH) and genomic in situ hybridization (GISH) are robust techniques to visualize the size of alien introgressions and breakpoints. We identified a complete set of monosomic addition lines into both bread wheat and durum wheat, except for 7J in durum wheat, by sequential ND-FISH and GISH. We also characterized alien derivatives belonging to various classes including mono-telosomic additions, disomic additions, monosomic substitutions, double monosomic substitutions, monosomic substitution–monosomic additions, double monosomic additions, and multiple monosomic additions into both bread and durum wheats. In addition, various wheat-Th. bessarabicum recombinant chromosomes were also detected in six alien derivatives. These wheat-Th. bessarabicum derivatives will provide useful cytogenetic resources for improvement of both hexaploid and tetraploid wheats.





Similar content being viewed by others
References
Able JA, Langridge P (2006) Wild sex in the grasses. Trends Plant Sci 11:261–263. https://doi.org/10.1016/j.tplants.2006.04.004
Ardalani S, Mirzaghaderi G, Badakhshan H (2016) A Robertsonian translocation from Thinopyrum bessarabicum into bread wheat confers high iron and zinc contents. Plant Breed 135:286–290. https://doi.org/10.1111/pbr.12359
Cuadrado Á, Golczyk H, Jouve N (2009) A novel, simple and rapid nondenaturing FISH (ND-FISH) technique for the detection of plant telomeres. Potential used and possible target structures detected. Chromosome Res 17:755. https://doi.org/10.1007/s10577-009-9060-z
Dewey DR (1984) The genomic system of classification as a guide to intergeneric hybridization with the perennial Triticeae. In: Gustafson JP (ed) Gene manipulation in plant improvement. Springer, Boston, pp 209–279. https://doi.org/10.1007/978-1-4613-2429-4_9
Du P, Zhuang L, Wang Y, Yuan L, Wang Q, Wang D, Dawadondup Tan L, Shen J, Xu H, Zhao H, Chu C, Qi Z (2016) Development of oligonucleotides and multiplex probes for quick and accurate identification of wheat and Thinopyrum bessarabicum chromosomes. Genome 60:93–103. https://doi.org/10.1139/gen-2016-0095
Dubcovsky J, Dvorak J (2007) Genome plasticity a key factor in the success of polyploid wheat under domestication. Science 316:1862–1866. https://doi.org/10.1126/science.1143986
Feldman M (1988) Cytogenetic and molecular approaches to alien gene transfer in wheat. In: Miller TE, Koebner RMD (eds) Proceedings of the 7th international wheat genetics symposium, Cambridge, UK, pp 23–32
Friebe B, Jiang J, Raupp WJ, McIntosh RA, Gill BS (1996) Characterization of wheat–alien translocations conferring resistance to diseases and pests: current status. Euphytica 91:59–87. https://doi.org/10.1007/BF00035277
Fu S, Chen L, Wang Y, Li M, Yang Z, Qiu L, Yan B, Ren Z, Tang Z (2015) Oligonucleotide probes for ND-FISH analysis to identify rye and wheat chromosomes. Sci Rep 5:10552. https://doi.org/10.1038/srep10552
Gill KS, Gill BS, Endo TR (1993) A chromosome region-specific mapping strategy reveals gene-rich telomeric ends in wheat. Chromosoma 102:374–381. https://doi.org/10.1007/BF00360401
Giorgi B (1978) A homoeologous pairing mutant isolated in Triticum durum cv. Cappelli. Mutat Breed Newsl 11:4–5
Giorgi B, Cuozzo L (1980) Homoeologous pairing in a ph mutant of tetraploid wheat crossed with rye. Cereal Res Commun 8:485–490
Gorham J, McDonnell E, Budrewicz E, Jones RW (1985) Salt tolerance in the Triticeae: growth and solute accumulation in leaves of Thinopyrum bessarabicum. J Exp Bot 36:1021–1031. https://doi.org/10.1093/jxb/36.7.1021
Grewal S, Yang C, Edwards SH, Scholefield D, Ashling S, Burridge AJ, King IP, King J (2018) Characterisation of Thinopyrum bessarabicum chromosomes through genome-wide introgressions into wheat. Theor Appl Genet 131:389–406. https://doi.org/10.1007/s00122-017-3009-y
Gyawali Y, Zhang W, Chao S, Xu S, Cai X (2019) Delimitation of wheat ph1b deletion and development of ph1b-specific DNA markers. Theor Appl Genet 132:195–204. https://doi.org/10.1007/s001
Han F, Fedak G, Benabdelmouna A, Armstrong K, Ouellet T (2003) Characterization of six wheat × Thinopyrum intermedium derivatives by GISH, RFLP, and multicolor GISH. Genome 46:490–495. https://doi.org/10.1139/g03-032
Harlan JR, de Wet JM (1971) Toward a rational classification of cultivated plants. Taxon 20:509–517
Hassani HS, Caligair PD, Miller T (2003) The chromosomal assessment of salt tolerant substituted Tritipyrum using genomic fluorescent in situ hybridization (FISH). Iran J Biotech 1:169–178
Jauhar PP, Chibbar RN (1999) Chromosome-mediated and direct gene transfers in wheat. Genome 42:570–583. https://doi.org/10.1139/g99-045
Jauhar PP, Peterson T (2006) Cytological analyses of hybrids and derivatives of hybrids between durum wheat and Thinopyrum bessarabicum, using multicolour fluorescent GISH. Plant Breed 125:19–26. https://doi.org/10.1111/j.1439-0523.2006.01176.x
Jauhar PP, Peterson TS (2013) Synthesis and characterization of advanced durum wheat hybrids and addition lines with Thinopyrum chromosomes. J Hered 104:428–436. https://doi.org/10.1093/jhered/ess143
Jiang J, Friebe B, Gill B (1994) Recent advances in alien gene transfer in wheat. Euphytica 73:199–212. https://doi.org/10.1007/BF00036700
Kato A (1999) Air drying method using nitrous oxide for chromosome counting in maize. Biotech Histochem 74:160–166. https://doi.org/10.3109/10520299909047968
Khlestkina EK (2014) Current applications of wheat and wheat–alien precise genetic stocks. Mol Breed 34:273–281. https://doi.org/10.1007/s11032-014-0049-8
King I, Law C, Cant K, Orford S, Reader S, Miller T (1997) Tritipyrum, a potential new salt-tolerant cereal. Plant Breed 116:127–132. https://doi.org/10.1111/j.1439-0523.1997.tb02166.x
Linc G, Gaál E, Molnár I, Icsó D, Badaeva E, Molnár-Láng M (2017) Molecular cytogenetic (FISH) and genome analysis of diploid wheatgrasses and their phylogenetic relationship. PLoS ONE 12:e0173623. https://doi.org/10.1371/journal.pone.0173623
Nemeth C, Yang C, Kasprzak P, Hubbart S, Scholefield D, Mehra S, Skipper E, King IP, King J (2015) Generation of amphidiploids from hybrids of wheat and related species from the genera Aegilops, Secale, Thinopyrum and Triticum as a source of genetic variation for wheat improvement. Genome 58:71–79. https://doi.org/10.1139/gen-2015-0002
Okamoto M (1957) Asynaptic effect of chromosome V. Wheat Inf Serv 5:6
Patokar C, Sepsi A, Schwarzacher T, Kishii M, Heslop-Harrison J (2016) Molecular cytogenetic characterization of novel wheat–Thinopyrum bessarabicum recombinant lines carrying intercalary translocations. Chromosoma 125:163–172. https://doi.org/10.1007/s00412-015-0537-6
Pu J, Wang Q, Shen Y, Zhuang L, Li C, Tan M, Bie T, Chu C, Qi Z (2015) Physical mapping of chromosome 4J of Thinopyrum bessarabicum using gamma radiation-induced aberrations. Theor Appl Genet 128:1319–1328. https://doi.org/10.1007/s00122-015-2508-y
Qi L, Friebe B, Zhang P, Gill BS (2007) Homoeologous recombination, chromosome engineering and crop improvement. Chromosome Res 15:3–19. https://doi.org/10.1007/s10577-006-1108-8
Qi Z, Du P, Qian B, Zhuang L, Chen H, Chen T, Shen J, Guo J, Feng Y, Pei Z (2010) Characterization of a wheat–Thinopyrum bessarabicum (T2JS-2BS.2BL) translocation line. Theor Appl Genet 121:589–597. https://doi.org/10.1007/s00122-010-1332-7
Riley R, Chapman V (1958) Genetic control of the cytologically diploid behaviour of hexaploid wheat. Nature 182:713–715. https://doi.org/10.1038/182713a0
Schneider A, Molnár I, Molnár-Láng M (2008) Utilisation of Aegilops (goatgrass) species to widen the genetic diversity of cultivated wheat. Euphytica 163:1–19. https://doi.org/10.1007/s10681-007-9624-y
Sears ER (1972) Chromosome engineering in wheat. In: Stadler symposia, vol 4. University of Missouri, Columbia, USA, pp 23–38
Sears ER (1976) Genetic control of chromosome pairing in wheat. Ann Rev Genet 10:31–51
Sears ER (1977) Genetics society of Canada award of excellence lecture an induced mutant with homoeologous pairing in common wheat. Can J Genet Cytol 19:585–593. https://doi.org/10.1139/g77-063
Sears ER (1981) Transfer of alien genetic material to wheat. In: Evans LT, Peacock WJ (eds) Wheat science - today and tomorrow. Cambridge University Press, Cambridge, pp 75–89
Sears ER (1984) Mutations in wheat that raise the level of meiotic chromosome pairing. In: Gustafson JP (ed) Gene manipulation in plant improvement. Plenum Press, New York, pp 295–300
Sepsi A, Molnár I, Szalay D, Molnár-Láng M (2008) Characterization of a leaf rust-resistant wheat–Thinopyrum ponticum partial amphiploid BE-1, using sequential multicolor GISH and FISH. Theor Appl Genet 116:825–834. https://doi.org/10.1007/s00122-008-0716-4
Shen Y, Shen J, Zhuang L, Wang Y, Pu J, Feng Y, Chu C, Wang X, Qi Z (2013) Physical localization of a novel blue-grained gene derived from Thinopyrum bessarabicum. Mol Breed 31:195–204. https://doi.org/10.1007/s11032-012-9783-y
Singh AK, Zhang P, Dong C, Li J, Trethowan R, Sharp P (2019) Molecular cytogenetic characterization of stem rust and stripe rust resistance in wheat–Thinopyrum bessarabicum-derived doubled haploid lines. Mol Breed 39:125. https://doi.org/10.1007/s11032-019-1034-z
Song L, Lu Y, Zhang J, Pan C, Yang X, Li X, Liu W, Li L (2016) Cytological and molecular analysis of wheat–Agropyron cristatum translocation lines with 6P chromosome fragments conferring superior agronomic traits in common wheat. Genome 59:840–850. https://doi.org/10.1139/gen-2016-0065
Tang Z, Yang Z, Fu S (2014) Oligonucleotides replacing the roles of repetitive sequences pAs1, pSc119. 2, pTa-535, pTa71, CCS1, and pAWRC. 1 for FISH analysis. J Appl Genet 55:313–318. https://doi.org/10.1007/s13353-014-0215-z
Wang X, Lai J, Liu G, Chen F (2002) Development of a SCAR marker for the Ph1 locus in common wheat and its application. Crop Sci 42:1365–1368. https://doi.org/10.2135/cropsci2002.1365
Wang R-C, Larson SR, Jensen KB (2010) Analyses of Thinopyrum bessarabicum, T. elongatum, and T. junceum chromosomes using EST-SSR markers. Genome 53:1083–1089. https://doi.org/10.1139/G10-088
William M, Mujeeb-Kazi A (1993) Thinopyrum bessarabicum: biochemical and cytological markers for the detection of genetic introgression in its hybrid derivatives with Triticum aestivum L. Theor Appl Genet 86:365–370. https://doi.org/10.1007/BF00222103
William M, Mujeeb-Kazi A (1995) Biochemical and molecular diagnostics of Thinopyrum bessarabicum chromosomes in Triticum aestivum germ plasm. Theor Appl Genet 90:952–956. https://doi.org/10.1007/BF00222908
Xu S, Jin Y, Klindworth D, Wang R-C, Cai X (2009) Evaluation and characterization of seedling resistances to stem rust Ug99 races in wheat–alien species derivatives. Crop Sci 49:2167–2175. https://doi.org/10.2135/cropsci2009.02.0074
Zhang P, Friebe B, Lukaszewski AJ, Gill BS (2001) The centromere structure in Robertsonian wheat-rye translocation chromosomes indicates that centric breakage-fusion can occur at different positions within the primary constriction. Chromosoma 110:335–344. https://doi.org/10.1007/s004120100159
Zhang J-Y, Li X-M, Wang RR-C, Cortes A, Rosas V, Mujeeb-Kazi A (2002) Molecular cytogenetic characterization of Eb-genome chromosomes in Thinopyrum bessarabicum disomic addition lines of bread wheat. Int J Plant Sci 163:167–174. https://doi.org/10.1086/324531
Zhang P, Dundas IS, Xu SS, Friebe B, McIntosh RA, Raupp WJ (2017) Chromosome engineering techniques for targeted introgression of rust resistance from wild wheat relatives. In: Periyannan S (ed) Wheat rust diseases. Methods in molecular biology. Humana Press, Totowa. https://doi.org/10.1007/978-1-4939-7249-4_14
Acknowledgements
The authors are thankful to the Grains Research and Development Corporation (GRDC), Australia, and BBSRC-UK for funding under Grant BB/100260X/1. The authors express their sincere thanks to Professors Ian King and Julie King, University of Nottingham, UK, for sharing seeds of the amphidiploids of Th. bessarabicum. AKS is grateful to the University of Sydney for the award of a Ph.D. studentship and for providing the platform for conducting these research activities. Authors would thank Prof. R. A. McIntosh for review of this manuscript.
Author information
Authors and Affiliations
Contributions
AKS, PS, PZ, CD, and RT conceived the project. AKS conducted the experiments and drafted the manuscript. PZ performed GISH experiments. JL assisted in the Oligo-FISH experiments. SS assisted in backcrossing and FISH analysis. PZ, CD, PS, and RT reviewed the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by P. Heslop-Harrison.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Singh, A.K., Zhang, P., Dong, C. et al. Development and molecular cytogenetic characterization of Thinopyrum bessarabicum introgression lines in hexaploid and tetraploid wheats. Theor Appl Genet 133, 2117–2130 (2020). https://doi.org/10.1007/s00122-020-03581-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-020-03581-5