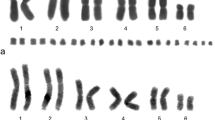
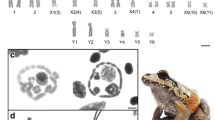
Abstract
It is notable that the occurrence of multiple sex chromosomes differs significantly between major lineages of amniote vertebrates. In this respect, birds are especially conspicuous, as multiple sex chromosomes have not been observed in this lineage so far. On the other hand, in mammals, multiple sex chromosomes have evolved many times independently. We hypothesize that this contrast can be related to the different involvement of sex-specific sex chromosomes in female meiosis subjected to the female meiotic drive under male versus female heterogamety. Essentially, the male-specific Y chromosome is not involved in female meiosis and is therefore sheltered against the effects of the female meiotic drive affecting the X chromosome and autosomes. Conversely, the Z and W sex chromosomes are both present in female meiosis. Nonrandom segregation of these sex chromosomes as a consequence of their rearrangements connected with the emergence of multiple sex chromosomes would result in a biased sex ratio, which should be penalized by selection. Therefore, the emergence of multiple sex chromosomes should be less constrained in the lineages with male rather than female heterogamety. Our broader phylogenetic comparison across amniotes supports this prediction. We suggest that our results are consistent with the widespread occurrence of female meiotic drive in amniotes.



Similar content being viewed by others
References
Axelsson E, Albrechtsen E, van AP, Li L, Megens HJ, Vereijken ALJ, Crooijmans RPMA, Groenen MAM, Ellegren H, Willerslev E, Nielsen R (2010) Segregation distortion in chicken and the evolutionary consequences of female meiotic drive in birds. Heredity 105:290–298
Bachtrog D (2013) Y-chromosome evolution: emerging insights into processes of Y-chromosome degeneration. Nat Rev Genet 14:113–124
Chaiprasertsri N, Uno Y, Peyachoknagul S, Prakhongcheep O, Baicharoen S, Charernsuk S, Nishida C, Matsuda Y, Koga A, Srikulnath K (2013) Highly species-specific centromeric repetitive DNA sequences in lizards: molecular cytogenetic characterization of a novel family of satellite DNA sequences isolated from the water monitor lizard (Varanus salvator macromaculatus, Platynota). J Hered 104:798–806
Charlesworth D, Charlesworth B, Marais G (2005) Steps in the evolution of heteromorphic sex chromosomes. Heredity 95:118–128
Christidis L (1990) Animal cytogenetics 4: Chordata 3 B: Aves. Gebrüder Borntraeger, Berlin
de Villena FPM, Sapienza C (2001) Female meiosis drives karyotypic evolution in mammals. Genetics 159:1179–1189
Dinkel BJ, O’Laughlin-Phillips EA, Fechheimer NS, Jaap RG (1979) Gametic products transmitted by chickens heterozygous for chromosomal rearrangements. Cytogenet Cell Genet 23:124–136
Ellegren H (2010) Evolutionary stasis: the stable chromosomes of birds. Trends Ecol Evol 25:283–291
Ezaz T, Stiglec R, Veyrunes F, Marshall Graves JA (2006) Relationships between vertebrate ZW and XY sex chromosome systems. Curr Biol 16:R736–R743
Ezaz T, Quinn AE, Sarre SD, O’Meally D, Georges A, Graves JAM (2009) Molecular marker suggests rapid changes of sex-determining mechanisms in Australian dragon lizards. Chromosom Res 17:91–98
Ferguson-Smith MA, Trifonov V (2007) Mammalian karyotype evolution. Nat Rev Genet 8:950–962
Fishman L, Saunders A (2008) Centromere-associated female meiotic drive entails male fitness costs in monkeyflowers. Science 322:1559–1562
Gamble T (2010) A review of sex determining mechanisms in geckos (Gekkota: Squamata). Sex Dev 4:88–103
Gorman GC (1973) The chromosomes of the Reptilia, a cytotaxonomic interpretation. In: Chiarelli AB, Capanna E (eds) Cytotaxonomy and vertebrate evolution. Academic Press Inc, New York, pp 349–424
Graves JAM (2006) Sex chromosome specialization and degeneration in mammals. Cell 124:901–914
Griffin DK, Robertson LBW, Tempest HG, Skinner BM (2007) The evolution of the avian genome as revealed by comparative molecular cytogenetics. Cytogenet Genome Res 117:64–77
Hedges SB, Dudley J, Kumar S (2006) TimeTree: a public knowledge-base of divergence times among organisms. Bioinformatics 22:2971–2972
Kasai F, O’Brien PCM, Martin S, Ferguson-Smith MA (2012) Extensive homology of chicken macrochromosomes in the karyotypes of Trachemys scripta elegans and Crocodylus niloticus revealed by chromosome painting despite long divergence times. Cytogenet Genome Res 136:303–307
Kawai A, Nishida-Umehara C, Ishijima J, Tsuda Y, Ota H, Matsuda Y (2007) Different origins of bird and reptile sex chromosomes inferred from comparative mapping of chicken Z-linked genes. Cytogenet Genome Res 117:92–102
Kejnovský E, Michalovova M, Steflova P, Kejnovska I, Manzano S, Hobza R, Kubat Z, Kovarik J, Jamilena M, Vyskot B (2013) Expansion of microsatellites on evolutionary young Y chromosome. PLoS ONE 8:e45519
Kitano J, Peichel CL (2012) Turnover of sex chromosomes and speciation in fishes. Environ Biol Fish 94:549–558
Kobayashi T, Yamada F, Hashimoto T, Abe S, Matsuda Y, Kuroiwa A (2008) Centromere repositioning in the X chromosome of XO/XO mammals, Ryukyu spiny rat. Chromosom Res 16:587–593
Li WH, Yi S, Makova K (2002) Male-driven evolution. Curr Opin Genet Dev 12:650–656
Matsuda Y, Nishida-Umehara C, Tarui H, Kuroiwa A, Yamada K, Isobe T, Ando J, Fujiwara A, Hirao Y, Nishimura O, Ishijima J, Hayashi A, Saito T, Murakami T, Murakami Y, Kuratani A, Agata K (2005) Highly conserved linkage homology between birds and turtles: bird and turtle chromosomes are precise counterparts of each other. Chromosom Res 13:601–615
Murphy WJ, Larkin DM, Everts-van der Wind A, Bourque G, Tesler G, Auvil L, Beever JE, Chowdhary BP, Gilbert F, Gatzke L, Hitte C, Meyers SN, Milan D, Ostrander EA, Page G, Parker HG, Raudsepp T, Rogatcheva MB, Schook LB, Skow LC, Welge M, Wormack JE, O’Brien SJ, Pevzner PA, Lewin HA (2005) Dynamics of mammalian chromosome evolution inferred from multispecies comparative maps. Science 309:613–617
Nakatani Y, Takeda H, Kohara Y, Morishita S (2007) Reconstruction of the vertebrate ancestral genome reveals dynamic genome reorganization in early vertebrates. Genome Res 17:1254–1265
Nguyen P, Sýkorová M, Šíchová J, Kůta V, Dalíková M, Čapková-Frydrychová R, Neven LG, Sahara K, Marec F (2013) Neo-sex chromosomes and adaptive potential in tortricid pests. Proc Natl Acad Sci U S A 110:6931–6936
O’Brien SJ, Menninger JC, Nash WG (eds) (2006) Atlas of mammalian chromosomes. Wiley, New York
O’Meally D, Patel HR, Stiglec R, Sarre SD, Georges A, Graves JAM, Ezaz T (2010) Non-homologous sex chromosomes of birds and snakes share repetitive sequences. Chromosom Res 18:787–800
O’Neill RJ, Eldridge MDB, Metcalfe CJ (2004) Centromere dynamics and chromosome evolution in marsupials. J Hered 95:375–381
Ohno S (1967) Sex chromosomes and sex-linked genes. Springer, Berlin
Ohno S (1969) Evolution of sex chromosomes in mammals. Annu Rev Genet 3:495–524
Olmo E (2008) Trends in the evolution of reptilian chromosomes. Integr Comp Biol 48:486–493
Pala I, Naurin S, Stervander M, Hasselquist D, Bensch S, Hansson B (2011) Evidence of a neo-sex chromosome in birds. Heredity 108:264–272
Pokorná M, Kratochvíl L (2009) Phylogeny of sex–determining mechanisms in squamate reptiles: are sex chromosomes an evolutionary trap? Zool J Linnean Soc 156:68–183
Pokorná M, Rábová M, Ráb P, Ferguson-Smith MA, Rens W, Kratochvíl L (2010) Differentiation of sex chromosomes and karyotypic evolution in the eye-lid geckos (Squamata: Gekkota: Eublepharidae), a group with different modes of sex determination. Chromosom Res 18:809–820
Pokorná M, Kratochvíl L, Kejnovský E (2011a) Microsatellite distribution on sex chromosomes at different stages of heteromorphism and heterochromatinization in two lizard species (Squamata: Eublepharidae: Coleonyx elegans and Lacertidae: Eremias velox). BMC Genet 12:90
Pokorná M, Giovannotti M, Kratochvíl L, Kasai F, Trifonov VA, O’Brien PC, Caputo V, Olmo E, Ferguson-Smith MA, Rens W (2011b) Strong conservation of the bird Z chromosome in reptilian genomes is revealed by comparative painting despite 275 million years divergence. Chromosoma 120:455–468
Pokorná M, Giovannotti M, Kratochvíl L, Caputo V, Olmo E, Ferguson-Smith MA, Rens W (2012) Conservation of chromosomes syntenic with avian autosomes in squamate reptiles revealed by comparative chromosome painting. Chromosoma 121:409–418
Rens W, O’Brien PC, Grutzner F, Clarke O, Graphodatskaya D, Tsend-Ayush E, Trifonov V, Selton H, Wallis MC, Johnston S, Veyrunez F, Graves JAM, Ferguson-Smith MA (2007) The multiple sex chromosomes of platypus and echidna are not completely identical and several share homology with the avian Z. Genome Biol 8:R243
Rice WR (1996) Evolution of the Y sex chromosome in animals: Y chromosomes evolve through the degeneration of autosomes. Bioscience 46:331–343
Rovatsos M, Altmanová M, Pokorná M, Kratochvíl L (2014a) Conserved sex chromosomes across adaptively radiated Anolis lizards. Evolution. doi:10.1111/evo.12357
Rovatsos M, Pokorná M, Altmanová M, Kratochvíl L (2014b) Cretaceous park of sex determination: sex chromosomes are conserved across iguanas. Biol Lett (in press)
Rutkowska J, Lagisz M, Nakagawa S (2012) The long and the short of avian W chromosomes: no evidence for gradual W shortening. Biol Lett 8:636–638
Toder R, O’Neill RJ, Wienberg J, O’Brien PC, Voullaire L, Graves JAM (1997) Comparative chromosome painting between two marsupials: origins of an XX/XY1Y2 sex chromosome system. Mamm Genome 8:418–422
Traut W, Sahara K, Marec F (2007) Sex chromosomes and sex determination in Lepidoptera. Sex Dev 1:332–346
Valenzuela N, Adams DC (2011) Chromosome number and sex determination coevolve in turtles. Evolution 65:1808–1813
Vicoso B, Emerson JJ, Zektser Y, Mahajan S, Bachtrog D (2013) Comparative sex chromosome genomics in snakes: differentiation, evolutionary strata, and lack of global dosage compensation. PLoS Biol 11:e1001643
Volff JN, Schartl M (2001) Variability of genetic sex determination in poeciliid fishes. Genetica 111:101–110
Vorontsov NN, Lyapunova EA, Borissov YM, Dovgal VE (1984) Variability of sex chromosomes in mammals. Genetica 52:361–372
Voss SR, Kump DK, Putta S, Pauly N, Reynolds A, Henry RJ, Basa S, Walker JA, Smith JJ (2011) Origin of amphibian and avian chromosomes by fission, fusion, and retention of ancestral chromosomes. Genome Res 21:1306–1312
Wells DE, Gutierrez L, Xu Z, Krylov V, Macha J, Blankenburg KP, Hitchens M, Bellot LJ, Spivey M, Stemple DL, Kowis A, Ye Y, Pasternak S, Owen J, Tran T, Slavikova R, Tumova L, Tlapakova T, Seifertova E, Scherer SE, Sater AK (2011) A genetic map of Xenopus tropicalis. Dev Biol 354:1–8
Wurster DH, Benirschke K (1970) Indian muntjac, Muntiacus muntjak: a deer with a low diploid chromosome number. Science 168:1364–1366
Yoshida K, Kitano J (2012) The contribution of female meiotic drive to the evolution of neo–sex chromosomes. Evolution 66:3198–3208
Acknowledgement
We are grateful to the editor, Darren K. Griffin, for the kind invitation to include our work in this special issue. This study was supported by grants GAČR 506/10/0718 and GA UK 591712. This article represents part seven of the series “Evolution of sex determining systems in lizards” by Martina Pokorná and Lukáš Kratochvíl.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Responsible Editor: Darren K. Griffin and Beth A. Sullivan.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Table S1
List of species with multiple sex chromosomes (XLSX 27 kb)
Table S2
Numbers of identified independent origins of multiple sex chromosomes, numbers of species with known multiple neo-sex chromosomes, numbers of karyotyped species and numbers of species with known sex chromosomes across amniote lineages (XLSX 15 kb)
Rights and permissions
About this article
Cite this article
Pokorná, M., Altmanová, M. & Kratochvíl, L. Multiple sex chromosomes in the light of female meiotic drive in amniote vertebrates. Chromosome Res 22, 35–44 (2014). https://doi.org/10.1007/s10577-014-9403-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10577-014-9403-2