Abstract
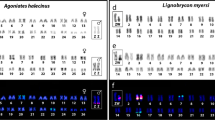
Distribution of sex-determining mechanisms across Australian agamids shows no clear phylogenetic segregation, suggesting multiple transitions between temperature-dependent (TSD) and genotypic sex determination (GSD). These taxa thus present an excellent opportunity for studying the evolution of sex chromosomes, and evolutionary transitions between TSD and GSD. Here we report the hybridization of a 3 kb genomic sequence (PvZW3) that marks the Z and W microchromosomes of the Australian central bearded dragon (Pogona vitticeps) to chromosomes of 12 species of Australian agamids from eight genera using fluorescence in-situ hybridization (FISH). The probe hybridized to a single microchromosome pair in 11 of these species, but to the tip of the long arm of chromosome pair 2 in the twelfth (Physignathus lesueurii), indicating a micro-macro chromosome rearrangement. Three TSD species shared the marked microchromosome, implying that it is a conserved autosome in related species that determine sex by temperature. C-banding identified the marked microchromosome as the heterochromatic W chromosome in two of the three GSD species. However, in Ctenophorus fordi, the probe hybridized to a different microchromosome from that shown by C-banding to be the heterochromatic W, suggesting an independent origin for the ZW chromosome pair in that species. Given the haphazard distribution of GSD and TSD in this group and the existence of at least two sets of sex microchromosomes in GSD species, we conclude that sex-determining mechanisms in this family have evolved independently, multiple times in a short evolutionary period.


Similar content being viewed by others
Abbreviations
- DAPI:
-
4′,6-diamidino-2-phenylindole
- dUTP:
-
2′-deoxyuridine 5′-triphosphate
- ESD:
-
environmental sex determination
- FISH:
-
fluorescence in-situ hybridization
- GSD:
-
genotypic sex determination
- PCR:
-
polymerase chain reaction
- SSC:
-
standard saline citrate
- TSD:
-
temperature sex determination
- v/v:
-
volume/volume
References
Bull JJ (1983) Evolution of sex determining mechanisms. Benjamin/Cummings, Menlo Park, CA
Charlesworth B (1991) The evolution of sex chromosomes. Science 251:1030–1033
Charnier M (1966) Action de la temperature sur la sex-ratio chez l’embryon d’Agama agama (Agamidae, Lacertilien). Societe de Biologie de L’Ouest Africain 160:620–622
Cogger HG (2000) Reptiles and Amphibians of Australia, 6th edn. Reed New Holland, Sydney
Conover DO, Kynard BE (1981) Environmental sex determination: interaction of temperature and genotype in a fish. Science 213:577–579
Cooper SJB, Bull CM, Gardner MG (1997) Characterisation of microsatellite loci from the social monogamous lizard Tiliqua rugosa using a PCR-based isolation technique. Mol Ecol 6:793–795
Devlin RH, Nagahama Y (2002) Sex determination and sex differentiation in fish: an overview of genetic, physiological, and environmental influences. Aquaculture 208:191–364
El Mouden EH, Znair M, Pieau C (2001) Effects of incubation temperature on embryonic development and sex determination in the North African lizard, Agama impalearis. Herpetol J 11:72–77
Ezaz T, Quinn AE, Miura I, Sarre SD, Georges A, Graves JAM (2005) The dragon lizard Pogona vitticeps has ZZ/ZW micro-sex chromosomes. Chromosome Res 13:763–776
Ezaz T, Stiglec R, Veyrunes F, Graves JAM (2006) Relationships between vertebrate ZW and XY sex chromosome systems. Curr Biol 16:736–743
Ganesh S, Raman R (1995) Sex reversal by testosterone and not by estradiol or temperature in Calotes versicolor, the lizard lacking sex chromosomes. J Exp Zool 271:139–144
Graves JAM (2006) Sex chromosome specialization and degeneration in mammals. Cell 124:901–912
Halverson J, Spelman LH (2002) Sex determination. In: Murphy JB, Ciofi C, de la Panouse C, Walsh T (eds) Biology and conservation of Komodo Dragons. Smithsonian Institution, Washington DC, pp 165–177
Harlow PS (1996) A harmless technique for sexing hatchling lizards. Herpetol Rev 27:71–72
Harlow PS (2000) Incubation temperature determines hatchling sex in Australian rock dragons (Agamidae: Genus Ctenophorus). Copeia 4:958–964
Harlow PS (2001) Ecology of sex determining mechanisms in Australian agamid lizards. PhD thesis, School of Biological Sciences, Macquarie University, Sydney, Australia
Harlow PS (2004) Temperature-dependent sex determination in lizards. In: Valenzuela N, Lance VA (eds) Temperature-dependent Sex Determination in Vertebrates. Smithsonian Institution, Washington DC, pp 11–20
Harlow PS, Shine R (1999) Temperature-dependent sex determination in the frillneck lizard, Chlamydosaurus kingii (Agamidae). Herpetologica 55:205–212
Harlow PS, Taylor JE (2000) Reproductive ecology of the jacky dragon (Amphibolurus muricatus): an agamid lizard with temperature-dependent sex determination. Aust Ecol 25:640–652
Hugall AF, Foster R, Hutchinson M, Lee MSY (2008) Phylogeny of Australasian agamid lizards based on nuclear and mitochondrial genes: implications for morphological evolution and biogeography. Biol J Linn Soc 93:343–358
Janzen FJ, Krenz JG (2004) Phylogenetics: which was first, TSD or GSD? In: Valenzuela N, Lance VA (eds) Temperature-dependent Sex Determination in Vertebrates. Smithsonian Institution, Washington DC, pp 121–130
Kawai A, Nishida-Umehara C, Ishijima J, Tsuda Y, Ota H, Matsuda Y (2007) Different origins of bird and reptile sex chromosomes inferred from comparative mapping of chicken Z-linked genes. Cytogenet Genome Res 117:92–102
Kawai A, Ishijima J, Chizuko C et al. (2008) The ZW sex chromosomes of Gekko hokouensis (Gekkonidae, Squamata) represent highly conserved homology with those of avian species. Chromosoma. doi 10.1007/s00412-008-0176-2.
Matsubara K, Tarui H, Toriba M et al (2006) Evidence for different origin of sex chromosomes in snakes, birds, and mammals and step-wise differentiation of snake sex chromosomes. Proc Natl Acad Sci USA 103:18190–18195
Matsuda M, Nagahama Y, Shinomiya A et al (2002) DMY is a Y-specific DM-domain gene required for male development in the medaka fish. Nature 417:559–563
Modi WS, Crews D (2005) Sex chromosomes and sex determination in reptiles. Curr Opin Genet Dev 15:660–665
Ohno S (1967) Sex chromosomes and sex-linked genes. Springer-Verlag, Heidelberg
Organ C, Janes DE (2008) Evolution sex chromosome in Sauropsids. Integr Comp Biol 48:512–519
Pieau C (1971) Sur la proportion sexuelle chez les embryons de deux Cheloniens (Testudo graeca L. et Emys orbicularis L.) issus d’oeufs incubes artificiellement. C R Acad Sci Paris 272:3071–3074
Quinn AE, Georges A, Sarre SD, Guarino F, Ezaz T, Graves JAM (2007) Temperature sex reversal implies sex gene dosage in a reptile. Science 316:411
Radder RS, Quinn AE, Georges A, Sarre SD, Shine R (2008) Genetic evidence for co-occurrence of chromosomal and thermal sex-determining systems in a lizard. Biol Lett 4:176–178
Sinclair AH, Berta P, Palmer MS et al (1990) A gene from the human sex-determining region encodes a protein with homology to a conserved DNA-binding motif. Nature 346:240–244
Solari AJ (1994) Sex Chromosomes and Sex Determination in Vertebrates. CRC Press, Boca Raton, FL
Stow AJ, Sunnucks P, Briscoe DA, Gardner MG (2001) The impact of habitat fragmentation on dispersal of Cunningham’s skink (Egernia cunninghami): Evidence from allelic and genotypic analyses of microsatellites. Mol Ecol 10:867–878
Takehana Y, Hamaguchi Y, Sakaizumi M (2008) Different origins of ZZ/ZW sex chromosomes in closely related medaka fishes, Oryzias javanicus and O. hubbsi. Chromosome Res 16:801–811
Uller T, Olsson M (2006) No seasonal sex-ratio shift despite sex-specific fitness returns of hatching date in a lizard with genotypic sex determination. Evolution 60:2131–2136
Uller T, Mott B, Odierna G, Olsson M (2006) Consistent sex ratio bias of individual female dragon lizards. Biol Lett 2:569–572
Witten GJ (1983) Some karyotypes of Australian agamids (Reptilia: Lacertilia). Aust J Zool 31:533–540
Zeng X-M, Wang Y-Z, Liu Z-J et al (1997) Karyotypes on nine species in the genus Phrynocephalus, with discussion of karyotypic evolution of Chinese Phrynocephalus. Acta Zool Sinica 43:399–410
Acknowledgements
We thank T. Uller for providing specimens of Ctenophorus fordi. Animal collection, handling, and sampling were performed according to the guidelines of the Australian Capital Territory Animal Welfare Act 1992 (Section 40) and licences issued by the Queensland, South Australia, Victoria, Australian Capital Territory, and New South Wales state governments. All experiments were performed with the approval of the University of Canberra Animal Experimentation Ethics Committee (Proposal CEAE 04/04) and the Australian National University Animal Experimentation Ethics Committee (Proposals R.CG.02.00 and R.CG.08.03). This work is supported by Australian Research Council Discovery Grants (DP0449935 and DP0881196) awarded to J.A.M.G., S.S. and A.G, and S.S. and A.G respectively.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Herbert Macgregor.
Rights and permissions
About this article
Cite this article
Ezaz, T., Quinn, A.E., Sarre, S.D. et al. Molecular marker suggests rapid changes of sex-determining mechanisms in Australian dragon lizards. Chromosome Res 17, 91–98 (2009). https://doi.org/10.1007/s10577-008-9019-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10577-008-9019-5