Abstract
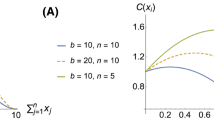
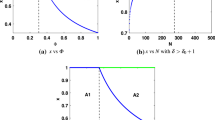
Some social insects exhibit an exceptionally high degree of polyandry. Alternative hypotheses exist to explain the benefits of multiple mating through enhanced colony performance. This study critically extends theoretical analyses of the hypothesis that enhanced division of labour confers fitness benefits to the queen that are sufficient to explain the observed mating frequencies of social insects. The effects of widely varying numbers of tasks and matings were systematically investigated in two alternative computer simulation models. One model was based on tasks that have to be performed to maintain an optimal trait value, while the other model was based on tasks that only have to be sufficiently performed to exceed a minimum trait value to confer full fitness returns. Both model versions were evaluated assuming a broad and a narrow response threshold distribution. The results consistently suggest a beneficial effect of multiple mating on colony performance, albeit with quickly diminishing returns. An increasing number of tasks decreased performance of colonies with few patrilines but not of more genetically diverse colonies. Instead, a performance maximum was found for intermediate task numbers. The results from the two model versions and two response threshold distributions did not fundamentally differ, suggesting that the type of tasks and the breadth of response thresholds do not affect the benefit of multiple mating. In general, our results corroborate previous models that have evaluated simpler task/patriline scenarios. Furthermore, selection for an intermediate number of tasks is indicated that could constrain the degree of division of labour. We conclude that enhanced division of labour may have favoured the evolution of multiple mating but is insufficient to explain the extreme mating numbers observed in some social insects, even in complex task scenarios.



Similar content being viewed by others
References
Baer B. and Schmid-Hempel P. 1999. Experimental variation in polyandry affects parasite loads and fitness in a bumble-bee. Nature 397: 151-154
Bastian J. and Esch H. 1970. The nervous control of the indirect flight muscles of the honey bee. Z. vergl. Physiol. 67: 307-324
Beekman M., Sumpter, D.J.T. and Ratnieks, F.L.W. 2001. Phase transition between disordered and ordered foraging in Pharaoh’s ants. Proc. Natl Acad. Sci. 98: 9703-9706
Bertram S.M., Gorelick R. and Fewell J.H. 2003. Colony response to graded resource changes: an analytical model of the influence of genotype, environment, and dominance. Theor. Pop. Biol. 64: 151-162
Beshers S.N. and Fewell J.H. 2001. Models of division of labor in social insects. Annu. Rev. Entomol. 46: 413-440
Beye M., Gattermeier I., Hasselmann M., Gempe T., Schioett M., Baines J.F., Schlipalius D., Mougel F., Emore C., Rueppell O., Sirvio A., Guzman-Novoa E., Hunt G., Solignac M. and Page R.E. 2006. Exceptionally high levels of recombination across the honey bee genome. Genome Res. 16: 1339-1344
Breed M.D., Robinson G.E. and Page R.E. 1990. Division of labor during honey bee colony defense. Behav. Ecol. Sociobiol. 27: 395-401
Brown M.J.F. and Schmid-Hempel P. 2003. The evolution of female multiple mating in social Hymenoptera. Evolution 57: 2067-2081
Camazine S. 1993. The regulation of pollen foraging by honey bees: How foragers assess the colony’s need for pollen. Behav. Ecol. Sociobiol. 32: 265-273
Chadwick P. 1931. Ventilation of the hive. Glean. Bee Cult. 59: 356-358
Chapman N.C., Oldroyd B.P. and Hughes W.O.H. 2007. Differential responses of honeybee (Apis mellifera) patrilines to changes in stimuli for the generalist tasks of nursing and foraging. Behav. Ecol. Sociobiol. 61: 1185-1194
Crozier R.H. and Consul P.C. 1976. Conditions for genetic polymorphism in social hymenoptera under selection at the colony level. Theor. Popul. Biol. 10:1-9
Crozier R.H. and Page R.E. 1985. On being the right size: male contributions and multiple mating in social Hymenoptera. Behav. Ecol. Sociobiol. 18: 105-115
Dreller C. and Tarpy D.R. 2000. Perception of the pollen need by foragers in a honeybee colony. Anim. Behav. 59: 91–96
Fewell J.H. and Page R.E. 1993. Genotypic variation in foraging responses to environmental stimuli by honey-bees, Apis mellifera. Experientia 49: 1106-1112
Fewell J.H. and Winston M.L. 1992. Colony state and regulation of pollen foraging in the honey-bee, Apis mellifera L. Behav. Ecol. Sociobiol. 30: 387-393
Fjerdingstad E.J. and Boomsma, J.J. 2000. Queen mating frequency and relatedness in young Atta sexdens colonies. Insect. Soc. 47: 354-356
Fournier D., Battaille G., Timmermans I. and Aron S. 2008. Genetic diversity, worker size polymorphism and division of labour in the polyandrous ant Cataglyphis cursor. Anim. Behav. 75: 151-158
Fuchs S. and Moritz R.F.A. 1999. Evolution of extreme polyandry in the honeybee Apis mellifera L. Behav. Ecol. Sociobiol. 45: 269-275
Fuchs S. and Schade V. 1994. Lower performance in honeybee colonies of uniform paternity. Apidologie 25: 155-169
Goode K., Huber Z., Mesce K.A. and Spivak M. 2006. Hygienic behavior of the honey bee (Apis mellifera) is independent of sucrose responsiveness and foraging ontogeny. Horm. Behav. 49: 391-397
Graham S., Myerscough M.R., Jones J.C. and Oldroyd B.P. 2006. Modelling the role of intracolonial genetic diversity on regulation of brood temperature in honey bee (Apis mellifera L.) colonies. Insect. Soc. 53: 226-232
Hölldobler B. and Wilson E.O. 1990. The Ants. The Belknap Press of Harvard University Press, Cambridge. 732 pp
Hughes W.O.H. and Boomsma J.J. 2004. Genetic diversity and disease resistance in leaf-cutting ant societies. Evolution 58: 1251-1260
Hughes W.O.H. and Boomsma J.J. 2007. Genetic polymorphism in leaf-cutting ants is phenotypically plastic. Proc. R. Soc. Lond. B. 274: 1625-1630
Jaffe R., Kronauer D.J.C., Kraus F.B., Boomsma J.J. and Moritz R.F.A. 2007. Worker caste determination in the army ant Eciton burchellii. Biol. Lett. 3: 513-516
Jeanson R., Fewell J.H., Gorelick R. and Bertram S.M. 2007. Emergence of increased division of labor as a function of group size. Behav. Ecol. Sociobiol. 62: 289-298
Johnson B.R. 2008a. Within-nest temporal polyethism in the honey bee. Behav. Ecol. Sociobiol. 62: 777-784
Johnson B.R. 2008b. Global information sampling in the honey bee. Naturwissenschaften 95: 523-530
Jones J.C., Myerscough M.R., Graham S. and Oldroyd B.P. 2004. Honey bee nest thermoregulation: Diversity promotes stability. Science 305: 402-404
Jones J.C., Nanork P. and Oldroyd B.P. 2007. The role of genetic diversity in nest cooling in a wild honey bee, Apis florea. J. Comp. Physiol. A. 193: 159-165.
Kallenberg O. 1997. Foundations of Modern Probability. Springer-Verlag, New York. 515 pp
Kolmes S.A. 1985. A quantitative study of the division of labour among worker honeybees. Z. Tierpsychol. 68: 287-302
Kronauer D.J.C., Johnson R.A. and Boomsma J.J. 2007. The evolution of multiple mating in army ants. Evolution 61: 413-422
Kryger P., Kryger U. and Moritz R.F.A. 2000. Genotypical variability for the tasks of water collecting and scenting in a honey bee colony. Ethology 106: 769-779
Lindauer M. 1952. Ein Beitrag zur Frage der Arbeitsteilung im Bienenstaat. Z. vergl. Physiol. 34: 299-345
Lindauer M. 1954. Temperaturregulierung und Wasserhaushalt im Bienenstaat. Z. vergl. Physiol. 36: 391-432
Mattila H.R., Burke K.M. and Seeley T.D. 2008. Genetic diversity within honeybee colonies increases signal production by waggle-dancing foragers. Proc. R. Soc. Lond. B. 275: 809-816
Mattila H.R. and Seeley T.D. 2007. Genetic diversity in honey bee colonies enhances productivity and fitness. Science 317: 362-364
Maynard Smith J. and Szathmáry E. 1995. The Major Transitions in Evolution. W.H. Freeman, San Francisco. 360 pp
Moritz R.F.A. 1985. The effect of multiple mating on the worker-queen conflict in Apis mellifera. Behav. Ecol. Sociobiol. 16: 375-377
Myerscough M.R. and Oldroyd B.P. 2004. Simulation models of the role of genetic variability in social insect task allocation. Insect. Soc. 51: 146-152
Oldroyd B.P. and Fewell J.H. 2007. Genetic diversity promotes homeostasis in insect colonies. Trend Ecol. Evol. 22: 408-413
Oldroyd B.P., Sylvester H.A., Wongsiri S. and Rinderer T.E. 1994. Task specialization in a wild-bee, Apis florea (Hymenoptera, Apidae), revealed by RFLP banding. Behav. Ecol. Sociobiol. 34: 25-30
Oster G.F. and Wilson E.O. 1978. Caste and Ecology in the Social Insects. Princeton University Press, Princeton. 352 pp
Page R.E. 1980. The evolution of multiple mating behavior by honey bee queens Apis mellifera L. Genetics 96: 263-273
Page R.E. and Fondrk M.K. 1995. The effects of colony level selection on the social-organization of honey-bee (Apis mellifera L) colonies-colony level components of pollen hoarding. Behav. Ecol. Sociobiol. 36: 135-144
Page R.E. and Mitchell S.D. 1998. Self-organization and the evolution of division of labor. Apidologie 29: 171-190
Page R.E., Robinson G.E., Fondrk M.K. and Nasr M.R. 1995. Effects of worker genotypic diversity on honey-bee colony development and behaviour (Apis mellifera L). Behav. Ecol. Sociobiol. 36: 387-396
Pankiw T. 2004a. Brood pheromone regulates foraging activity of honey bees (Hymenoptera: Apidae). J. Econ. Entomol. 97: 748–751
Pankiw T. 2004b. Cued in: Honey bee pheromones as information flow and colony decision making. Apidologie 35: 217–226
Pankiw T. 2007. Brood pheromone modulation of pollen forager turnaround time in the honey bee (Apis mellifera L.). J. Insect Behav. 20: 173-180
Pankiw T. and Page R.E. 1999. The effects of genotype, age, and caste on response thresholds to sucrose and foraging behavior of honey bees. J. Comp. Physiol. A. 185: 207-213
Pankiw T. and Page R.E. 2001. Brood pheromone modulates sucrose response thresholds in honeybees (Apis mellifera L.). Behav. Ecol. Sociobiol. 49: 206–213
Pankiw T., Page R.E. and Fondrk M.K. 1998. Brood pheromone stimulates pollen foraging in honey bees (Apis mellifera). Behav. Ecol. Sociobiol. 44: 193–198
Pankiw T. and Rubink W.L. 2002. Pollen foraging response to brood pheromone by Africanized and European honey bees (Apis mellifera L.). Ann. Entomol. Soc. Am. 95: 761–767
Pankiw T., Tarpy D.R. and Page R.E. 2002. Genotype and rearing environment affect honeybee perception and foraging behaviour. Anim. Behav. 64: 663-672
Ratnieks F.L.W. and Boomsma J.J. 1995. Facultative sex allocation by workers and the evolution of polyandry by queens in social Hymenoptera. Am. Nat. 145: 969-993
Robinson G.E. and Page R.E. 1988. Genetic determination of guarding and undertaking in honeybee colonies. Nature 333: 356-358
Robinson G.E. and Page R.E. 1989. Genetic determination of nectar foraging, pollen foraging, and nest-site scouting in honey bee colonies. Behav. Ecol. Sociobiol. 24: 317-323
Robinson G.E. and Page R.E. 1995. Genotypic constraints on plasticity for corpse removal in honey-bee colonies. Anim. Behav. 49: 867-876.
Rueppell O., Johnson N. and Rychtár J. 2008. Variance-based selection may explain general mating patterns in social insects. Biol. Lett. 4: 270-273
Schmid-Hempel P. 1998. Parasites in Social Insects. Princeton University Press, Princeton. 392 pp
Schneider S.S. 1987. The modulation of worker activity by the vibration dance of the honeybee Apis mellifera. Ethology 74: 211-218
Schneider S.S., Stamps J.A. and Gary N.E. 1986. The vibration dance of the honey bee. I. Communication regulating foraging on two time scales. Anim. Behav. 34: 366-85
Seeley T.D. and Tarpy D.R. 2007. Queen promiscuity lowers disease within honeybee colonies. Proc. R. Soc. Lond. B. 274: 67-72
Sherman P.W., Seeley T.D. and Hudson K.R. 1988. Parasites, pathogens, and polyandry in social Hymenoptera. Am. Nat. 131: 602-610
Shykoff J.A. and Schmid-Hempel P. 1991. Parasites and the advantage of genetic variability within social insect colonies. Proc. R. Soc. Lond. B. 243: 55–58
Tarpy D.R. and Page R.E. 2001. The curious promiscuity of queen honey bees (Apis mellifera): evolutionary and behavioral mechanisms. Ann. Zool. Fenn. 38: 255-265
Tofts C. and Franks N.R. 1992. Doing the right thing - Ants, honeybees and naked mole-rats. Trends Ecol. Evol. 7: 346-349
van Baalen M. and Beekman M. 2006. The costs and benefits of genetic heterogeneity in resistance against parasites in social insects. Am. Nat. 167: 568-577
Waibel M., Floreano D., Magnenat S. and Keller L. 2006. Division of labour and colony efficiency in social insects: effects of interactions between genetic architecture, colony kin structure and rate of perturbations. Proc. R. Soc. B. 273: 1815-1823
Wattanachaiyingcharoen W., Oldroyd B.P., Wongsiri S., Palmer K. and Paar R. 2003. A scientific note on the mating frequency of Apis dorsata. Apidologie 34: 85-86
Weidenmuller A. 2004. The control of nest climate in bumblebee (Bombus terrestris) colonies: interindividual variability and self reinforcement in fanning response. Behav. Ecol. 15: 120-128
Wiernasz D.C., Hines J., Parker D.G. and Cole B.J. 2008. Mating for variety increases foraging activity in the harvester ant, Pogonomyrmex occidentalis. Mol. Ecol. 17: 1137-1144
Wiernasz D.C., Perroni D.L. and Cole B.J. 2004. Polyandry and fitness in the western harvester ant, Pogonomyrmex occidentalis. Mol. Ecol. 13: 1601-1606
Wilfert L., Gadau J. and Schmid-Hempel P. 2007. Variation in genomic recombination rates among animal taxa and the case of social insects. Heredity 98: 189-197
Wilson E.O. 1976. Behavioral discretization and the number of castes in an ant species. Behav. Ecol. Sociobiol. 1: 141-154
Acknowledgments
We would like to thank all members of the UNCG Math-Bio group for their comments, suggestions, and lively discussion. This study was improved by the comments of two anonymous reviewers of an earlier version of this manuscript but potential errors remain our own. Financial support was provided by NSF (#0634182 and #0615502). The work performed adheres to all applicable ethical standards and government regulations.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gove, R., Hayworth, M., Chhetri, M. et al. Division of labour and social insect colony performance in relation to task and mating number under two alternative response threshold models. Insect. Soc. 56, 319–331 (2009). https://doi.org/10.1007/s00040-009-0028-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00040-009-0028-y