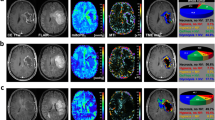
Abstract
Glioblastoma (GBM) is the most aggressive primary brain tumor with a short median survival. Tumor recurrence is a clinical expectation of this disease and usually occurs along the resection cavity wall. However, previous clinical observations have suggested that in cases of ischemia following surgery, tumors are more likely to recur distally. Through the use of a previously established mechanistic model of GBM, the Proliferation Invasion Hypoxia Necrosis Angiogenesis (PIHNA) model, we explore the phenotypic drivers of this observed behavior. We have extended the PIHNA model to include a new nutrient-based vascular efficiency term that encodes the ability of local vasculature to provide nutrients to the simulated tumor. The extended model suggests sensitivity to a hypoxic microenvironment and the inherent migration and proliferation rates of the tumor cells are key factors that drive distal recurrence.








Similar content being viewed by others
Notes
We denote this as f to represent fuel for the cells, to avoid reusing n which is already assigned to necrotic cells.
It is well known that nutrient concentrations in blood (such as glucose concentration) fluctuate throughout a single day; however, we are interested in modeling tumor growth over many days and months, so only consider the average nutrient concentration across these daily fluctuations.
References
Adeberg S, König L, Bostel T, Harrabi S, Welzel T, Debus J, Combs S E (2014) Glioblastoma recurrence patterns after radiation therapy with regard to the subventricular zone. Int J Radiat Oncol Biol Phys 90 4:886–893
Ansarizadeh F, Singh M, Richards D (2017) Modelling of tumor cells regression in response to chemotherapeutic treatment. Appl Math Model 48:96–112
Armento A, Ehlers J, Schötterl S, Naumann U (2017) Molecular mechanisms of glioma cell motility. Exon Publications 73–93
Barazzuol L, Burnet NG, Jena R, Jones B, Jefferies SJ, Kirkby NF (2010) A mathematical model of brain tumour response to radiotherapy and chemotherapy considering radiobiological aspects. J Theor Biol 262(3):553–565
Bette S, Barz M, Huber T, Straube C, Schmidt-Graf F, Combs SE, Delbridge C, Gerhardt J, Zimmer C, Meyer B et al (2018) Retrospective analysis of radiological recurrence patterns in glioblastoma, their prognostic value and association to postoperative infarct volume. Sci Rep 8(1):4561
Bette S, Wiestler B, Kaesmacher J, Huber T, Gerhardt J, Barz M, Delbridge C, Ryang Y-M, Ringel F, Zimmer C et al (2016) Infarct volume after glioblastoma surgery as an independent prognostic factor. Oncotarget 7(38):61945
Boujelben A, Watson M, McDougall S, Yen Y-F, Gerstner ER, Catana C, Deisboeck T, Batchelor TT, Boas D, Rosen B et al (2016) Multimodality imaging and mathematical modelling of drug delivery to glioblastomas. Interface Focus 6(5):20160039
Burnet N, Jena R, Jefferies S, Stenning S, Kirkby N (2006) Mathematical modelling of survival of glioblastoma patients suggests a role for radiotherapy dose escalation and predicts poorer outcome after delay to start treatment. Clin Oncol 18(2):93–103
Chamberlain M (2011) Radiographic patterns of relapse in glioblastoma. J Neuro-oncol 101(2):319–323
Cocosco CA, Kollokian V, Kwan R K-S, Pike GB, Evans AC (1997) Brainweb: Online interface to a 3d mri simulated brain database. In NeuroImage, Citeseer
Collins DL, Zijdenbos AP, Kollokian V, Sled JG, Kabani NJ, Holmes CJ, Evans AC (1998) Design and construction of a realistic digital brain phantom. IEEE Trans Med Imaging 17(3):463–468
Curtin L, Hawkins-Daarud A, Van Der Zee KG, Swanson KR, Owen MR (2020) Speed switch in glioblastoma growth rate due to enhanced hypoxia-induced migration. Bull Math Biol 82(3):1–17
de Rioja VL, Isern N, Fort J (2016) A mathematical approach to virus therapy of glioblastomas. Biol Direct 11(1):1
Delbeke D, Meyerowitz C, Lapidus R, Maciunas R, Jennings M, Moots P, Kessler R (1995) Optimal cutoff levels of f-18 fluorodeoxyglucose uptake in the differentiation of low-grade from high-grade brain tumors with pet. Radiology 195(1):47–52
Frieboes HB, Lowengrub JS, Wise S, Zheng X, Macklin P, Bearer EL, Cristini V (2007) Computer simulation of glioma growth and morphology. Neuroimage 37:S59–S70
Gallaher JA, Massey SC, Hawkins-Daarud A, Noticewala SS, Rockne RC, Johnston SK, Gonzalez-Cuyar L, Juliano J, Gil O, Swanson KR et al (2020) From cells to tissue: How cell scale heterogeneity impacts glioblastoma growth and treatment response. PLoS Comput Biol 16(2):e1007672
Harpold H, Alvord E, Swanson K (2007) The evolution of mathematical modeling of glioma proliferation and invasion. J Neuropathol Exp Neurol 66(1):1–9
Hawkins-Daarud A, Rockne R, Anderson A, Swanson KR (2013) Modeling tumor-associated edema in gliomas during anti-angiogenic therapy and its impact on imageable tumor. Front Oncol 3:66
Keunen O, Johansson M, Oudin A, Sanzey M, Rahim S, Fack F, Thorsen F, Taxt T, Bartos M, Jirik R et al (2011) Anti-vegf treatment reduces blood supply and increases tumor cell invasion in glioblastoma. Proc Natl Acad Sci 108(9):3749–3754
Kwan RK-S, Evans AC, Pike GB (1996) An extensible mri simulator for post-processing evaluation. In: Visualization in biomedical computing, Springer, pp 135–140
Kwan R-S, Evans AC, Pike GB (1999) Mri simulation-based evaluation of image-processing and classification methods. IEEE Trans Med Imaging 18(11):1085–1097
Leder K, Pitter K, LaPlant Q, Hambardzumyan D, Ross BD, Chan TA, Holland EC, Michor F (2014) Mathematical modeling of pdgf-driven glioblastoma reveals optimized radiation dosing schedules. Cell 156(3):603–616
Liberti MV, Locasale JW (2016) The warburg effect: how does it benefit cancer cells? Trends Biochem Sci 41(3):211–218
Louis D, Ohgaki H, Wiestler O, Cavenee W (2016) WHO Classification of Tumours of the Central Nervous System, Revised. Fourth Edition. International Agency for Research on Cancer
Macklin P, Lowengrub J (2007) Nonlinear simulation of the effect of microenvironment on tumor growth. J Theor Biol 245(4):677–704
Martínez-González A, Calvo G, Romasanta L, Pérez-García V (2012) Hypoxic cell waves around necrotic cores in glioblastoma: a biomathematical model and its therapeutic implications. Bull Math Biol 74(12):2875–2896
Neufeld Z, von Witt W, Lakatos D, Wang J, Hegedus B, Czirok A (2017) The role of allee effect in modelling post resection recurrence of glioblastoma. PLoS Comput Biol 13(11):e1005818
Pardo R, Martinez-Gonzalez A, Perez-Garcia VM (2016) Nonlinear ghost waves accelerate the progression of high-grade brain tumors. Commun Nonlinear Sci Numer Simul 39:360–380
Raza SM, Lang FF, Aggarwal BB, Fuller GN, Wildrick DM, Sawaya R (2002) Necrosis and glioblastoma: a friend or a foe? A review and a hypothesis. Neurosurgery 51(1):2–13
Rockne RC, Trister AD, Jacobs J, Hawkins-Daarud AJ, Neal ML, Hendrickson K, Mrugala MM, Rockhill JK, Kinahan P, Krohn KA et al (2015) A patient-specific computational model of hypoxia-modulated radiation resistance in glioblastoma using 18f-fmiso-pet. J R Soc Interface 12(103):20141174
Roniotis A, Sakkalis V, Tzamali E, Tzedakis G, Zervakis, M, Marias K (2012) Solving the pihna model while accounting for radiotherapy. In: Advanced Research Workshop on In Silico Oncology and Cancer Investigation-The TUMOR Project Workshop (IARWISOCI), 2012 5th International, IEEE, pp 1–4
Rutter EM, Stepien TL, Anderies BJ, Plasencia JD, Woolf EC, Scheck AC, Turner GH, Liu Q, Frakes D, Kodibagkar V et al (2017) Mathematical analysis of glioma growth in a murine model. Sci Rep 7(1):1–16
Scribner E, Saut O, Province P, Bag A, Colin T, Fathallah-Shaykh HM (2014) Effects of anti-angiogenesis on glioblastoma growth and migration: model to clinical predictions. PLoS One 9(12):e115018
Silbergeld D, Chicoine M (1997) Isolation and characterization of human malignant glioma cells from histologically normal brain. J Neurosurg 86(3):525–531
Stark AM, van de Bergh J, Hedderich J, Mehdorn HM, Nabavi A (2012) Glioblastoma: clinical characteristics, prognostic factors and survival in 492 patients. Clin Neurol Neurosurg 114(7):840–845
Stein AM, Demuth T, Mobley D, Berens M, Sander LM (2007) A mathematical model of glioblastoma tumor spheroid invasion in a three-dimensional in vitro experiment. Biophys J 92(1):356–365
Stupp R, Hegi M, Mason W, van den Bent M, Taphoorn M, Janzer R, Ludwin S, Allgeier A, Fisher B, Belanger K et al (2009) Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase iii study: 5-year analysis of the eortc-ncic trial. Lancet Oncol 10(5):459–466
Subramanian S, Gholami A, Biros G (2019) Simulation of glioblastoma growth using a 3d multispecies tumor model with mass effect. J Math Biol 79(3):941–967
Swan A, Hillen T, Bowman J, Murtha A (2018) A patient-specific anisotropic diffusion model for brain tumour spread. Bull Math Biol 80(5):1259–1291
Swanson K (1999) Mathematical Modeling of the Growth and Control of Tumors. PhD thesis, University of Washington
Swanson KR, Alvord E Jr, Murray J (2000) A quantitative model for differential motility of gliomas in grey and white matter. Cell Prolif 33(5):317–29
Swanson KR, Bridge C, Murray J, Alvord E (2003) Virtual and real brain tumors: using mathematical modeling to quantify glioma growth and invasion. J Neurol Sci 216(1):1–10
Swanson KR, Rockne R, Claridge J, Chaplain M, Alvord E, Anderson A (2011) Quantifying the role of angiogenesis in malignant progression of gliomas: in silico modeling integrates imaging and histology. Cancer Res 71(24):7366–7375
Swanson KR, Rostomily R, Alvord E (2008) A mathematical modelling tool for predicting survival of individual patients following resection of glioblastoma: a proof of principle. Br J Cancer 98(1):113–119
Thiepold A, Luger S, Wagner M, Filmann N, Ronellenfitsch M, Harter P, Braczynski A, Dützmann S, Hattingen E, Steinbach J et al (2015) Perioperative cerebral ischemia promote infiltrative recurrence in glioblastoma. Oncotarget 6(16):14537
Warburg O (1925) The metabolism of carcinoma cells. J Cancer Res 9(1):148–163
Yamaguchi T, Kanno I, Uemura K, Shishido F, Inugami A, Ogawa T, Murakami M, Suzuki K (1986) Reduction in regional cerebral metabolic rate of oxygen during human aging. Stroke 17(6):1220–1228
Yang Y, Hou L, Li Y, Ni J, Liu L (2013) Neuronal necrosis and spreading death in a drosophila genetic model. Cell Death Dis 4(7):e723
Zagzag D, Lukyanov Y, Lan L, Ali MA, Esencay M, Mendez O, Yee H, Voura EB, Newcomb EW (2006) Hypoxia-inducible factor 1 and vegf upregulate cxcr4 in glioblastoma: implications for angiogenesis and glioma cell invasion. Lab Investig 86(12):1221
Zuniga R, Torcuator R, Jain R, Anderson J, Doyle T, Ellika S, Schultz L, Mikkelsen T (2009) Efficacy, safety and patterns of response and recurrence in patients with recurrent high-grade gliomas treated with bevacizumab plus irinotecan. J Neuro-oncol 91(3):329
Acknowledgements
The authors gratefully acknowledge funding from the National Cancer Institute (R01CA164371, U54CA193489) and the School of Mathematical Sciences at the University of Nottingham.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Recurrence Results of Other PIHNA Simulations
Recurrence Results of Other PIHNA Simulations
We present the results of PIHNA simulations that were not shown in the main text. The trends in distant recurrence patterns that we observe in the main text all hold in these simulations, supporting our observations regarding \(D_h/D_c\), \(\beta \), \(\gamma \), \(D_c\) and \(\rho \) (Figs. 9, 10, 11, 12, 13, 14).
Recurrence location classified for various \(D_c\), \(\rho \), \(\beta \) and levels of ischemia for \(D_h = D_c\) for \(\gamma = 0.005\)/day and \(\gamma =0.5\)/day. We see that higher values of \(\beta \) and lower levels of \(\gamma \) lead to a larger proportion of distant recurrences in \(D_c\) and \(\rho \) parameter space. Higher migration rates, \(D_c\), and lower proliferation rates, \(\rho \), lead to more distantly recurring simulated tumors
Recurrence location classified for various \(D_c\), \(\rho \), \(\beta \) and levels of ischemia for \(D_h = 10D_c\) for \(\gamma = 0.005\)/day and \(\gamma =0.5\)/day. We see that higher values of \(\beta \) and lower levels of \(\gamma \) lead to a larger proportion of distant recurrences in \(D_c\) and \(\rho \) parameter space. Higher migration rates, \(D_c\), and lower proliferation rates, \(\rho \), lead to more distantly recurring simulated tumors
Recurrence location classified for various \(D_c\), \(\rho \), \(\beta \) and levels of ischemia for \(D_h = 100D_c\) for \(\gamma = 0.005\)/day and \(\gamma =0.5\)/day. We see that higher values of \(\beta \) and lower levels of \(\gamma \) lead to a larger proportion of distant recurrences in \(D_c\) and \(\rho \) parameter space. Higher migration rates, \(D_c\), and lower proliferation rates, \(\rho \), lead to more distantly recurring simulated tumors
Recurrence location classified for various \(D_c\), \(\rho \), \(\beta \) and levels of ischemia for \(D_h = 10D_c\) for \(\gamma = 0.05\)/day. In these simulations, perioperative ischemia was set at 10% of the pre-resection value. We see a larger proportion of local recurrence in these figures compared with those in the main text (see Fig. 4)
Recurrence location classified for various \(D_c\), \(\rho \), \(\beta \) and levels of ischemia for \(D_h = 10D_c\) for \(\gamma = 0.005\)/day. In these simulations, perioperative ischemia was set at 10% of the pre-resection value. We see a larger proportion of local recurrence in these figures compared with those in the main text (see Fig. 5)
Recurrence location classified for various \(D_c\), \(\rho \) and \(D_h\) levels for \(\beta = 0.5\rho \) and \(\gamma = 0.05\)/day. In these simulations, perioperative ischemia was set at 10% of the pre-resection value. We see a larger proportion of local recurrence in these figures compared with those in the main text (see Fig. 6)
Rights and permissions
About this article
Cite this article
Curtin, L., Hawkins-Daarud, A., Porter, A.B. et al. A Mechanistic Investigation into Ischemia-Driven Distal Recurrence of Glioblastoma. Bull Math Biol 82, 143 (2020). https://doi.org/10.1007/s11538-020-00814-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11538-020-00814-y