Abstract
This paper proposes and analyzes a mathematical model of an infectious disease system with a piecewise control function concerning threshold policy for disease management strategy. The proposed models extend the classic models by including a piecewise incidence rate to represent control or precautionary measures being triggered once the number of infected individuals exceeds a threshold level. The long-term behaviour of the proposed non-smooth system under this strategy consists of the so-called sliding motion—a very rapid switching between application and interruption of the control action. Model solutions ultimately approach either one of two endemic states for two structures or the sliding equilibrium on the switching surface, depending on the threshold level. Our findings suggest that proper combinations of threshold densities and control intensities based on threshold policy can either preclude outbreaks or lead the number of infecteds to a previously chosen level.
Similar content being viewed by others
1 Introduction
Emerging infectious diseases such as the 2009 A/H1N1 influenza pandemic and the SARS outbreak in 2003 (Cauchemez et al. 2009; Peiris et al. 2004; Skowronski et al. 2005; Tang et al. 2010) or endemic diseases such as HIV or TB in China (CMH 2009; Lu et al. 2008; Liu et al. 2010) threaten public health, despite attempts to contain their world-wide spread by implementation of stringent non-pharmaceutical interventions (NPIs). It is thus important to investigate effective control strategies that can prevent outbreaks or minimize the impact of such outbreaks if their prevention is impossible.
Mathematical modelling can be a useful tool for designing strategies to control rapidly spreading infectious diseases in the absence of an effective treatment, vaccine, or diagnostic test (Anderson and May 1991). The impacts of a variety of intervention measures for SARS (Lipsitch et al. 2003; Pourbohloul et al. 2005) and, more recently, for pandemic influenza outbreaks (Ferguson et al. 2006; Germann et al. 2006; Tang et al. 2010) have been studied. Although such studies provide vital information for public health officials, they do not consider situations when multiple outbreaks are possible. Other studies (Feng and Thieme 1995; Hethcote 2000; Xiao et al. 2011; Liu et al. 2007; Sun et al. 2011) have examined variation of the basic reproduction number or long-term dynamics on control measures to assess the efficacy of different strategies. However, these studies did not consider whether or not the interventions affect the initial epidemic nor the timing for triggering intervention measures. This study addresses questions that arise if disease elimination is not possible in a short time. These include how to reduce disease severity to buy time to, for instance, produce and deploy vaccines, how to control outbreaks when resources are limited, or alternatively, how to keep the number of infecteds at a desired low level for a long time.
Control strategies for the transmission dynamics of infectious diseases have been modelled as control parameters which may either be constant or variable during disease progression in a dynamic system (Feng and Thieme 1995; Hethcote 2000; Xiao and Tang 2010; Xiao et al. 2011; Wang 2006; Wang et al. 2012). However, all such models assume, explicitly or implicitly that interventions have been implemented throughout the disease progression, disregarding the case numbers and the timing of implementation.
However, intervention measures are usually implemented by the government only when the case number reaches a critical level. Usually, when the case number is relatively high, the public may be encouraged to take precautionary measures against the disease, probably in response to media reporting, which may reduce the frequency of potentially infecting contacts and lower the probability of disease transmission among the well-informed population (Cui et al. 2008; Liu et al. 2007; Sun et al. 2011). Note that during spreading of emergent infectious disease the general information disseminated to the public is often restricted to simply reporting the number of infections and deaths, while government agencies for disease control and prevention may attempt to contain the disease (Tchuenche et al. 2011; World Health Organization 2005). It follows that the case number has been used as an index for the public or the authority to change their behaviour or to implement control strategies. Several analyses have suggested that individuals reactively reduced their contact rates, in response to high levels of mortality or the presence of many infectious individuals, during a pandemic (Martin et al., 2007; Tang et al., 2010, 2012; Tchuenche et al., 2011). In such cases, interventions are modelled and represented by using a piecewise defined function which is dependent on the case number.
Very little is known about the effects of this discontinuous control function on the dynamic behaviour of epidemic models for disease control. The main purpose of this paper, therefore, is to formulate a compartmental model to represent circumstances when intervention measures are taken only when the case number reaches a certain critical value by proposing a discontinuous incidence rate. The proposed models aim at precluding the possibility of outbreaks or causing the infection to stabilize at a desired level by changing individual avoidance and contact patterns in response to the reported information of infectious cases. The overall objective is to develop a systematic way of designing simple implementable controls that drive the dynamical systems to a desired globally stable equilibrium.
2 The SIR Model with Threshold Policy
To illustrate our ideas, we begin with a simple compartment SIR model (Anderson and May 1991; Diekmann and Heesterbeek 2000). The reported number of infectious individuals has profound psychological impact on behaviour that seems to reduce the effective contact of susceptible people with infectious individuals (Cui et al. 2008; Liu et al. 2007). At the initial stages of an epidemic, the general population and the public authority are unaware of the disease, and hence the former do not change behaviour and the latter do not implement a control strategy, allowing the disease to spread. When the number of infecteds reaches a critical level I c , people are sufficiently aware of the infection to change their behaviour and some control measures are implemented, resulting in the reduction (f) in transmission. We here include the demographic process to explore the longer-term persistence and endemic dynamics since population movement is an important factor for infectious diseases to spread geographically over time (Khan et al., 2009; Tang et al., 2010, 2012). Let Λ be the (constant) recruitment rate and d be the natural death rate of the population. We consider the dynamics of susceptibles S, infecteds I, and recovereds R. The model equations are
with
where β is the transmission coefficient, and γ is the recovery rate from infection. Model (1) with (2) is a description of the threshold policy (hereafter named TP), which is referred to as an on-off control or as a special and simple case of variable structure control in the control literature (Edwards and Spurgeon, 1998; Filippov, 1988; Utkin, 1978, 1992). A TP leads to a variable structure system with two distinct structures with their own equilibrium points, separated by the threshold level (Utkin 1978). A sliding mode (Utkin 1992) along I c may ensue, if in its vicinity the vector fields of both structures are directed toward each other. The detailed mathematical structure of the proposed threshold policy is laid out in Appendix A. Initially, we only consider the first two equations of model (1) with (2), and denote the structure without intervention (ϵ=0) by free system (S 1) and the structure with intervention (ϵ=1) by control system (S 2). It is not difficult to prove that solutions of the system model (1) are ultimately uniformly bounded by Λ/d, then the attraction region for the system (1) without considering dynamics of the recovered individuals is
We examine all the possible equilibria and their stability for this system (1) with (2). There are two types of equilibria: those that belong to the sliding domain Ω and those that do not. The former is referred to as the sliding equilibrium, whereas the latter is called the natural equilibrium which includes two classes: real equilibria and virtual equilibria. If the equilibrium points are located in their opposite regions, they are named virtual equilibrium points. Otherwise, they are called real equilibrium points (see details in Appendix A or Costa et al., 2000). It is worth mentioning that if the locally stable equilibrium points are virtual, they will never be attained since the dynamics change as soon as the trajectories cross the threshold I c .
For the control system (S 2), we can also easily define the basic reproduction number
It is easy to obtain that the disease-free equilibrium \((\frac{\varLambda}{d}, 0)\) is globally asymptotically stable if R 02≤1; the endemic state \(E^{2}=(\frac{d+\gamma}{\beta(1-f)},\frac{\varLambda\beta(1-f)-d(d+\gamma)}{\beta(1-f)(d+\gamma)})\) is globally asymptotically stable if R 02>1. In particular, E 2 could either be a stable spiral if Δ<0; or be a stable node if Δ≥0, where
Similarly, for the free system (S 1), the basic reproduction number is given by \(R_{01}=\frac{\varLambda \beta }{d(d+\gamma)}\). Moreover, the endemic state \(E^{1}=(\frac{d+\gamma}{\beta}, \frac{\varLambda\beta -d(d+\gamma)}{\beta (d+\gamma)})\) is globally asymptotically stable if R 01>1.
Denote
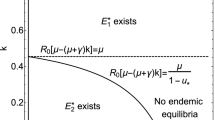
It is easy to verify that when I c <H 1 (i.e., region ϒ 1 in Fig. 1) the endemic state of the free system is a virtual equilibrium, denoted by \(E_{V}^{1}\), and the endemic state of the control system is a regular equilibrium, denoted by \(E_{R}^{2}\), whereas we have the regular equilibium \(E_{R}^{1}\) and the virtual equilibium \(E_{V}^{2}\) for I c >H 2 (i.e., region ϒ 4∪ϒ 5∪ϒ 6). The endemic states for both the free system and the controlled system are virtual equilibria for H 1<I c <H 2 (i.e., region ϒ 2∪ϒ 3, see details in Fig. 1). Note that Fig. 1 shows the bifurcation set for the model (1) with (2) to exhibit the influence of the important parameters (control intensity f and the threshold level I c to trigger control measures) on possible equilibria.
Bifurcation set for the model (1) with (2) with respect to the control intensity f and threshold level I c . Let ϒ 1 be the domain bounded by the curve I c =H 1 (solid curve), the boundaries I c =0 and f=0; ϒ 2 be the domain bounded by the curves I c =H 1, I c =H 2 (dash-dotted curve), and I c =H 3 (dash curve); ϒ 3 be the domain bounded by the curves I c =H 2, I c =H 3, I c =0, and the boundary f=1; ϒ 4 be the domain bounded by the curves I c =H 2, I c =H 3, and the boundary f=0; ϒ 5 be the domain bounded by the curves I c =H 2, I c =H 3, I c =H 4 (dotted line), and the boundary f=1; ϒ 6 be the domain bounded by the line I c =H 4 and the boundaries f=0, f=1 and I c =Λ/d. Parameter values are a=0.6, β=1, d=0.2, γ=0.2
2.1 The Sliding Mode Dynamics
We initially examine the existence of the sliding mode. The manifold Σ is defined as
which is a discontinuity surface between the two different structures of the system. A ‘sliding mode’ exists if there are regions in the vicinity of manifold Σ where the vectors for the two different structures of the system (1) are directed toward each other. Mathematically, there are two basic methods to determine sufficient conditions for a sliding mode to occur on the surface of a discontinuity (see details in Bernardo et al., 2008; Utkin, 1978). We can simply verify that the sliding mode exists since there exists a nonempty set
where the two adjacent vector fields point toward the manifold. Then the set Ω is called the sliding domain. Denote the end points of the sliding domain Ω by point A and B. Let point P and Q be the intersection points between the manifold Σ with line S(t)=0 and line S+I=Λ/d, respectively.
Following the method developed by Utkin (1992), we use a formal procedure to obtain equations describing sliding mode dynamics along the manifold Σ for system (1) with (2) (see details in Appendix A). By means of algebraic manipulations, we can eliminate the control ϵ. Note that σ′=I′=β(1−fϵ)SI−(d+γ)I, and letting σ′=0 gives
Substituting the control ϵ given above and I=I c into the first equation of (1) gives the system dynamics on the switching line
Obviously, Eq. (7) has a unique equilibrium \(S_{*}=\frac{\varLambda-(d+\gamma)I_{c}}{d}\) which is locally asymptotically stable on the switching surface, and then system (1) with (2) has a sliding equilibrium (S ∗,I c ), denoted by E S . It is easy to show that the sliding equilibrium point E S belongs to the sliding domain if (d+γ)/β<S ∗<(d+γ)/(β(1−f)), that is
To investigate its global stability and the long-term dynamics of the system (1) with (2), we initially explore the relationship between the sliding domain Ω given in (6) and the attraction region \({\mathcal{D}}\) given in (3). Simple calculations indicate that the sliding domain is included in the attraction region \({\mathcal{D}}\) if
whilst the sliding domain is excluded within the attraction region \({\mathcal{D}}\) if
Note that when H 3<I c <H 4 (i.e., region ϒ 3∪ϒ 5 in Fig. 1) the sliding domain is partly within the attraction region \({\mathcal{D}}\). Note that H 3≤H 4, H 1≤H 2 for f≥0, and H 2<H 4 for R 01>1. We mention that we do not consider the case of R 01≤1 since in such a case the disease will die out for the free system and the control strategy may not be necessary to consider. Notice that the condition (8) describes whether the sliding equilibrium is in the sliding domain or not, and conditions (9) and (10) determine the relationship between the sliding domain and the attraction region (see details in Fig. 1).
Figure 2(a) and (b) illustrate the regular/virtual equilibria (red dots), sliding equilibrium (black dots), sliding mode (grey lines) and the attraction region \({\mathcal{D}}\). It follows from Fig. 2(a) that the sliding domain is totally, partly, and exclusively in the attraction region for relatively low, middle, and high levels of threshold I c , respectively. It indicates that for middle values of the threshold the sliding equilibrium is feasible and the endemic states for free/control system are virtual. Whereas for the low or high level of thresholds, one of two equilibria is regular, and hence the sliding equilibrium does not exist. Comparing Fig. 2(a) and (b) shows that as control intensity f is strengthened the sliding domain is enlarged.
Evolution of null-isoclines, sliding modes (grey), and sliding equilibria (black dots), the regular/virtual equilibria (red dots), of system (1) with (2) with respect to the control intensity f and threshold level I c . The boundary of the attraction region \({\mathcal{D}}\) is bounded by S+I≤Λ/d, (dashed line). The horizontal and vertical isoclinic curves for the free system S 1 are \(\{(S,I)\in R_{+}^{2}:\ S=\frac{d+\gamma}{\beta}, I<I_{c}\}\) and \(\{(S,I)\in R_{+}^{2}:\ \varLambda-\beta SI-dS=0, I<I_{c}\}\), denoted by \(g_{S_{1}}^{1}\) and \(g_{S_{1}}^{2}\), respectively. The horizontal and vertical isoclinic curves for the control system S 2 are \(\{(S,I)\in R_{+}^{2}:\ S=\frac{d+\gamma}{\beta(1-f)}, I>I_{c}\}\) and \(\{(S,I)\in R_{+}^{2}:\ \varLambda-\beta(1-f) SI-dS=0, I>I_{c}\}\), denoted by \(g_{S_{2}}^{1}\) and \(g_{S_{2}}^{2}\), respectively. Parameter values are a=0.6, β=1, d=0.2, γ=0.2 (Color figure online)
2.2 Global Behaviour
We now consider the asymptotical behaviour of the system (1) with (2). In order to get the richness of the possible dynamics that the system can exhibit, all possible combinations of the control intensity f and the threshold level I c were chosen to build phase diagrams. Note that we only consider the case of R 02>1. That is because the control system itself will stabilize to its disease-free equilibrium for R 02≤1, consequently solution for the system (1) with (2) will definitely hit the switching surface, and finally converge to the endemic state E 1 of the free system. We initially consider the region ϒ 1 to show the asymptotical properties of solutions.
Theorem 1
The equilibrium \(E_{R}^{2}\) is globally asymptotically stable if I c <H 1.
Proof
In the region ϒ 1 where I c <H 1 the two endemic equilibria for two structures belong on the same sides of the switching surface (i.e., the region where I>I c ). So, we have the virtual equilibrium \(E_{V}^{1}\) and the regular equilibrium \(E_{R}^{2}\) (shown in Fig. 3(a)). Although the sliding mode which is included in the attraction region does exist, no sliding equilibrium exists in the sliding domain. In fact, analyzing the Jacobian matrix yields that \(E_{R}^{2}\) is stable in the region of I>I c due to our assumption that R 02>1. The virtual equilibrium \(E_{V}^{1}\) is also asymptotically stable, and trajectories initiating from the region where I<I c tend to it before hitting the switching surface. In the sliding domain Ω given in (6) (here the segment \(\overline{AB}\) in Fig. 3(a)), we have

Therefore, when trajectories hit the sliding domain Ω the state vector starts to move to the right end point of the sliding domain (point B) along the sliding domain (shown in Fig. 3(a)). Then we have the following two claims.
Phase plane S–I for SIR model with demography (1) with (2), showing the sliding domain (\(\overline{AB}\)) and asymptotical equilibrium including regular equilibrium (small blue circles: \(E_{R}^{1}\) or \(E_{R}^{2}\)), virtual equilibrium (small blue circles: \(E_{V}^{1}\) or \(E_{V}^{2}\)) or sliding equilibrium (small red circle: E S ) for different parameter sets. The horizontal isoclinic line (black) \(g_{S_{1}}^{1}\) (\(g_{S_{2}}^{1}\)) and the vertical isoclinic curve (green) \(g_{S_{1}}^{2}\) (\(g_{S_{2}}^{2}\)) are plotted for the free (control) system S 1 (S 2). The blue curves represent the orbits in the phase plane indicating the asymptotical equilibrium. Parameters values are a=0.6,β=1,d=0.2,γ=0.2, and other parameters are chosen such that dynamics in all region ϒ i (i=1,…,6) shown in Fig. 1 are exhibited. (a) I c =0.5, f=0.65 for region ϒ 1; (b) I c =1.15, f=0.65 for region ϒ 2; (c) I c =1.2, f=0.85 for region ϒ 3; (d) I c =1.6, f=0.65 for region ϒ 4; (e) I c =2.1, f=0.65 for region ϒ 5; (f) I c =2.7, f=0.65 for region ϒ 6 (Color figure online)
Claim 1
The trajectory initiating from the point \(B (\frac{d+\gamma}{\beta(1-f)},I_{c})\) will not hit the sliding domain again.
In fact, in the region I>I c shown in Fig. 3(a), the segment \(\overline{BB_{1}}\) can be a non-tangent segment for the control system S 2, where point B 1 is the middle point of the segment \(\overline{BE_{R}^{2}}\), since the line \(S=\frac{d+\gamma}{\beta(1-f)}\) is an isoclinic line along which I′=0. We note that the trajectory starting from point B either tends to the stable equilibrium \(E_{R}^{2}\) directly or spirally since \(E_{R}^{2}\) could be a stable node or a focus in the region I>I c . If the latter happens, all intersection points of the orbit and the segment \(\overline{BB_{1}}\) are placed in order in the segment \(\overline{BB_{1}}\), and are above the point B. Hence, a trajectory starting at B cannot hit the sliding domain \(\overline{AB}\) or the segment \(\overline{PA}\). So, we conclude that trajectory starting at B cannot form a cycle.
Claim 2
No limit cycle surrounds the regular equilibrium \(E_{R}^{2}\) and the sliding mode \(\overline{AB}\).
Denote the right-hand sides of the first two equations of model (1) by f 1 and f 2.
For I<I c , we choose function \(D=\frac{1}{SI}\) as a Dulac function and calculate
For I>I c , we still choose the function \(D=\frac{1}{SI}\) and perform the same calculation as above. By using the Lemma for the non-smooth system given in Appendix B, we can preclude the existence of a limit cycle which surrounds the regular equilibrium \(E_{R}^{2}\) and the sliding domain \(\overline{AB}\).
Hence, the combination of Claim 1, Claim 2, and local stability of \(E_{R}^{2}\) implies that it is globally asymptotically stable. The proof is complete. □
Theorem 2
The sliding equilibrium E S is globally stable if H 1<I c <H 2.
Proof
According to the above argument in Sect. 2.1, we know that in this case the sliding equilibrium E S exists and is stable in the sliding domain. If the sliding domain is totally within the attraction region \({\mathcal{D}}\) (shown in Fig. 3(b)), we can prove that no limit cycle surrounds the sliding domain \(\overline{AB}\) by using a similar method to Claim 2 in Theorem 1. If the sliding domain exceeds the attraction region \({\mathcal{D}}\) (shown in Fig. 3(c)), then on the one hand, all the trajectories initiating from the region I>I c will either hit the sliding domain and tend to the sliding equilibrium, or enter into the region I<I c by crossing through the segment \(\overline{PA}\). On the other hand, all the trajectories initiating from the region I<I c ultimately hit the sliding domain due to its stable equilibrium lying in the region where I>I c , and hence tend to the sliding equilibrium E S . So, E S is globally asymptotically stable. The proof is complete. □
Theorem 3
The equilibrium \(E_{R}^{1}\) is globally stable if I c >H 2.
Proof
When I c <H 3, it follows from (9) that the sliding domain is totally included in the attraction region (shown in Fig. 3(d)). In region ϒ 4 shown in Fig. 1, we have the regular equilibrium \(E_{R}^{1}\) and the virtual equilibrium \(E_{V}^{2}\) which lie in the region I<I c . We can prove that the equilibrium \(E_{R}^{1}\) is globally asymptotically stable by using a similar method to that for Theorem 1.
When H 3<I c <H 4 (the region ϒ 5 shown in Fig. 1), then the sliding domain exceeds the attraction region \({\mathcal{D}}\) (shown in Fig. 3(e)). In such a case, all the trajectories initiating from the region I<I c either tend to equilibrium \(E_{R}^{1}\) or hit the sliding domain, and hence move to the left end point (point A) of the sliding domain along the sliding domain. Using a similar method to Claim 1 of Theorem 1, we can prove that the trajectory initiating from the point A will not hit the sliding domain again but converge to the equilibrium \(E_{R}^{1}\). Then the equilibrium \(E_{R}^{1}\) is globally asymptotically stable.
When I c >H 4 (the region ϒ 6 shown in Fig. 1), then the sliding mode is precluded in the attraction region \({\mathcal{D}}\) (shown in Fig. 3(f)). Obviously, trajectories initiating from outside of the attraction region will eventually enter into the attraction region no matter whether they hit the sliding domain or not. Also, trajectories initiating from the region I>I c will ultimately enter into the region I<I c since its stable equilibrium \(E_{V}^{2}\) is in the region I<I c . Further, \(E_{R}^{1}\) is an unique stable equilibrium in the region I<I c , hence it is globally stable. Therefore, the equilibrium \(E_{R}^{1}\) is globally asymptotically stable for I c >H 2. The proof is complete. □
In summary, when we consider demographic processes the solutions of the non-smooth system ultimately converge to either one of two endemic states for two structures or the sliding equilibrium in the sliding domain on the switching surface, depending on the threshold level. This result could suggest a possible control strategy should elimination of the emerging infectious disease be impossible. That is, it follows from Theorem 2 that the proper combinations of threshold level and control intensities based on threshold policy can lead the number of infecteds to a previously chosen level.
2.3 The Special Case: SIR Model without Demography
In this subsection, we are interested in the dynamics of system (1) with (2) without demography (i.e., A=0, and d=0) and examine how the epidemic size (i.e., N−S ∞) changes with this variable structure control. Then total population size N is constant here and we have S(t)+I(t)≤N for all t. Using a similar method to that in Sect. 2.1, we obtain the sliding domain
where ρ=γ/β,ρ 1=γ/(β(1−f)). It indicates that the sliding mode does not exist for I c >N−ρ. The system dynamics on the switching line are described by the following equation:
which implies that the sliding mode has no equilibrium. Moreover, it means that when trajectories hit the sliding domain Ω the state vector starts to move along the switching surface asymptotically converging to the point A (the left end point of the sliding domain) in the phase plane (as shown in Fig. 5). According to the relationship between threshold level I c and control intensity f, there are three cases to consider. In the following, we choose N=1 to illustrate the trend of trajectories in the phase plane.
Case (1): Suppose I c <1−ρ 1. Then the sliding mode exists and its domain Ω is given by (12). Let V 1 be the region bounded by the integral curve \(\widetilde{BE}\) and the segments \(\overline{BQ}\) and \(\overline{EQ}\), V 2 be the region bounded by the integral curves \(\widetilde{AD}\), \(\widetilde{BE}\) and the segments \(\overline{AB}\) and \(\overline{ED}\) (as shown in Fig. 5(a)). Note that any trajectory initiating from V 2 hits the sliding domain Ω directly, while trajectories initiating from V 1 will cause an outbreak in the sense that the number of infecteds initially exceeds I c , then decreases and finally hits the sliding domain. In the sliding domain, there is a rapid alternation of intervention with intervention free, resulting in shorter periods of both modalities. To inhibit occurrence of an outbreak, we could strengthen intervention measures by increasing control intensity f such that the value of ρ 1 increases and reaches 1−I c , that is, the sliding domain is prolonged to the right such that its right end point B coincides with point Q (as shown in Fig. 5(b)). Therefore, any trajectory initiating from the region I<I c cannot cause an outbreak in the sense that the number of infecteds exceeds the given threshold level I c .
Case (2): Suppose 1−ρ 1≤I c <1−ρ. The calculation shows that the sliding mode exists and its domain becomes
Let M 1 be the domain bounded by the integral curves \(\widetilde{AC}\), \(\widetilde{AD,}\) and the segment \(\overline{CD}\). As in Fig. 5(b), any trajectory initiating from M 1 in Fig. 6(a) will hit the sliding domain Ω and slide to its left end point A (ρ,I c ), and finally tend to \((S_{1}^{\infty}, 0)\) with \(S_{1}^{\infty}\) satisfying
Trajectories initiating from other regions never behave either like those with no interventions, or like those with interventions before hitting the segment \(\overline{PA}\) of the switching surface, then switch to follow the free system and finally fall down to the S-axes (as shown in Fig. 6(a)).
Case (3): Suppose I c ≥1−ρ. In such case, a sliding mode does not exist. Any trajectory starting from the region where I<I c does not hit the switching surface, while trajectories starting from the region where I≥I c hit the switching surface first, and then converge to the S-axes (shown in Fig. 6(b)). In such a scenario where the critical level I c is set to be relatively high, intervention measures are not triggered, but an outbreak with the infection spreading sufficiently to significantly deplete the susceptible population is possible. Therefore, it is essential to determine the critical level at which to trigger intervention measures in order to control outbreaks.
Note that trajectories of model (1) with (2) without demography hitting the sliding domain, if the sliding mode exists, will slide along the Ω until leaving it, finally approaching a fixed point. Other trajectories behave like either those that are intervention free or those with interventions initially, and then switch to follow the free system and eventually fall down to the S-axes. These results, although seemingly exhibiting no qualitative difference when compared to the SIR model without switching from the point of view of ultimate trends, raise particularly interesting issues during the disease spreading process (i.e., disease evolution in its initial stage is consequently distinct). In particular, if the threshold level is set properly (as shown in Fig. 5(b)), this sliding mode control strategy could inhibit occurrence of outbreaks in a short term and lead the final size (represented by 1−S ∞ here) to decline in the long term. It then suggests an effective control measure for preventing outbreaks to combat an emerging infectious disease.
Remark
Handel et al. (2007) investigated the best control strategy to adopt for a situation when multiple outbreaks are likely to occur. They showed that the best strategy was to apply intervention measures in such a way that the number of susceptibles reaches exactly the threshold level. That is, their control intensity f h should be chosen such that (S ∞,I ∞)=(ρ,0), by using the first integral of the system their control intensity is as follows:
On the basis of this control intensity f h and the initial data (S 0,I 0), the maximum number of infecteds \(I_{h}^{\max}\) gives
We now try to derive the control intensity f by using our approach described above. For a fair comparison, we choose the control intensity such that the maximum number of infecteds is equivalent to \(I_{h}^{\max}\) given in (17). To this end, we choose the control parameter f such that the right end point B of the sliding domain coincides with or exceeds the point Q (as shown in Fig. 6(a)), that is, ρ/(1−f)≥1−I c is satisfied. It then follows from case (2) that the number of infecteds cannot exceed the given threshold level I c (here the \(I_{h}^{\max}\)). So, the optimal control intensity (the minimum) yields f 1=1−ρ/(1−I c ). Using \(I_{c}=I_{h}^{\max}\), then the control intensity reads
Further study shows that f 1 is equivalent to f h at \(S_{0}^{*}\), where \(S_{0}^{*}\) satisfies
And f 1 is less than f h for \(S_{0}<S_{0}^{*}\), whilst f 1 is greater than f h for \(S_{0}>S_{0}^{*}\). Note that although the control intensify f 1 may be greater than that obtained by Handel et al. (2007) for a relatively large initial number of susceptibles, the duration of implementation of control is quite limited and short, compared to the whole epidemic process needed to implement control in Handel et al.’s measure. In fact, we can only trigger a control strategy when the number of infecteds reaches the threshold level (here \(I_{c}=I_{h}^{\max}\)). It is not difficult to calculate the duration of intervention measures being implemented in our approach (see details in Appendix C).
3 Conclusion and Discussion
In this paper, we have proposed a mathematical model of infectious disease systems with a piecewise control function concerning a threshold policy for a disease management strategy. Note that human behaviour may adapt in relation to information on epidemic prevalence and the threat of endemic disease when such information is relatively complete. However, in the initial stage of the emerging infectious disease the information disseminated to the public is only the reported number of infections and deaths (Tchuenche et al. 2011; World Health Organization 2005). Thus, our formulation used the number of infections to be the threshold level for the public or the authority to change their behaviour or to implement control strategies. Further, given (Martin et al. 2007; Tang et al. 2010) that individuals reactively reduced their contact rates in response to high numbers of cases or high levels of mortality, the proposed models extend the classic models by including non-smooth incidence rates to represent control or precautionary measures being triggered once the number of infected individuals exceeds a threshold level. The global properties of the models are discussed. Our findings suggest that proper combinations of threshold densities and control intensities can either preclude outbreaks or lead the number of infecteds to a previously chosen level.
Dynamical systems subject to a TP are also referred to as variable structure systems in control terms. A TP results in a variable structure system with two distinct structures. In fishery management, a so-called TP is actually intermediate between the well-known constant escapement and constant harvest rate policies (Costa et al., 2000; Meza et al., 2005, 2002; Quinn and Deriso, 2000). In current HIV therapy policy, anti-retroviral therapy (ART) will be initiated whenever CD4 T cell counts are below 350 cells/mm3 (CMH 2009; Zhang et al. 2005). Although several studies have modelled such piecewise intervention measures in epidemiological models (Tchuenche et al. 2011), they do not investigate the long-term asymptotic behaviour for the whole system. This study is devoted to the investigation of the long-term dynamic behaviour of epidemic models with piecewise incidence rates, on the basis of a simple SIR-type model with/without demography, with the aim of generating possible strategies.
Using the classic SIR-type model, Handel et al. (2007) found that the best strategy for multiple infectious disease outbreaks was to apply intervention measures in such a way that the number of susceptibles reaches exactly the threshold level. However, their proposed interventions should last for the whole epidemic process, and moreover the timing of triggering intervention measures is not determined. This threshold policy applied to infectious disease management is realized by replacing continuous incidence rates (Handel et al. 2007) with a piecewise function which depends on the case number and the previously given threshold. Our main results obtained in Sect. 2.2 can, on the one hand, show that outbreaks are not possible, or on the other hand, determine the timings for switching on/off intervention measures. Further, when considering demographic processes we show that the solutions ultimately approach one of two endemic states for two structures or the sliding equilibrium on the switching surface, depending on the threshold level. It is worth mentioning that choosing an appropriate threshold level for making the decision to trigger the intervention and for its suspension is crucial (Cauchemez et al., 2009; Day et al., 2006; Tang et al., 2010, 2012).
It is important to emphasize that this policy is robust to uncertainties or intrinsic constraints in the model parameters. In fact, real systems may possess infrequent or inaccurate measurements of population densities. Nonetheless, if these uncertainties (or constraints) are bounded in the vicinity of the threshold and the vector fields maintained are directed toward each other, then the policy remains effective (Utkin 1978, 1992).
The work presented in this paper is just a first approach to the dynamics of disease management when the threshold policy, as often applied in the control literature, is considered, i.e. the density dependent piecewise control function is formulated. Our results show that the dynamics can be rather stable and useful from the disease control point of view, i.e. precluding outbreaks and resulting in the number of infecteds being reduced to a previously given level for appropriate threshold levels.
References
Anderson, R. M., & May, R. M. (1991). Infectious diseases of humans, dynamics and control. Oxford: Oxford University.
Bernardo, M. D., Budd, C. J., Champneys, A. R., et al. (2008). Bifurcations in nonsmooth dynamical systems. SIAM Rev., 50, 629–701.
Boukal, D. S., & Krivan, V. (1999). Lyapunov functions for Lotka–Volterra predator–prey models with optimal foraging behavior. J. Math. Biol., 39, 493–517.
Cauchemez, S., et al. (2009). Household transmission of 2009 pandemic influenza A (H1N1) virus in the United States. N. Engl. J. Med., 361, 2619–2627.
China Ministry of Health and joint UN programme on HIV/AIDS WHO. (2009). Estimates for the HIV/AIDS Epidemic in China, 2009.
Costa, M. I. S., Kaskurewicz, E., Bhaya, A., & Hsu, L. (2000). Achieving global convergence to an equilibrium population in predator–prey systems by the use of a discontinuous harvesting policy. Ecol. Model., 128, 89–99.
Cui, J., Tao, X., & Zhu, H. (2008). An SIS infection model incorporating media coverage. Rocky Mt. J. Math., 38(5), 1323–1334.
Day, T., Park, A., Madras, N., Gumel, A., & Wu, J. H. (2006). When is quarantine a useful control strategy for emerging infectious diseases? Am. J. Epidemiol., 163, 479–485.
Diekmann, O., & Heesterbeek, J. A. P. (2000). Mathematical epidemiology of infectious diseases: model building, analysis and interpretation. Chichester: Wiley.
Edwards, C., & Spurgeon, S. K. (1998). Systems and control book series: Vol. 7. Sliding mode control: theory and applications (pp. 15–20). London: Taylor and Francis.
Feng, Z., & Thieme, H. R. (1995). Recurrent outbreaks of childhood diseases revisited: the impact of isolation. Math. Biosci., 128, 93–130.
Ferguson, N. M., Cummings, D. A., et al. (2006). Strategies for mitigating an influenza pandemic. Nature, 442, 448–452.
Filippov, A. F. (1988). Differential equations with discontinuous righthand sides. Dordrecht: Kluwer Academic.
Germann, T. C., Kadau, K., Longini, K., & Macken, C. A. (2006). Mitigation strateties for pandemic influenza in the United States. Proc. Natl. Acad. Sci. USA, 103, 5935–5940.
Handel, A., Longini, I. M. Jr, & Antia, R. (2007). What is the best control strategy for multiple infectious disease outbreaks? Proc. R. Soc. B, 274, 833–837.
Hethcote, H. W. (2000). The mathematics of infectious disease. SIAM Rev., 42, 599–653.
Krivan, V. (1996). Optimal foraging and predator–prey dynamics. Theor. Popul. Biol., 49, 265–290.
Krivan, V. (1998). Effects of optimal antipredator behavior of prey on predator–prey dynamics: the role of refuges. Theor. Popul. Biol., 53, 131–142.
Khan, K., Arino, J., Hu, W., et al. (2009). Spread of a novel influenza A (H1N1) virus via global airline transportation. N. Engl. J. Med., 361(2), 212.
Liu, R., Wu, J., & Zhu, H. (2007). Media/psychological impact on multiple outbreaks of emerging infectious diseases. Comput. Math. Methods Med., 8(3), 153–164.
Liu, L., Zhao, X. Q., & Zhou, Y. (2010). A tuberculosis model with seasonality. Bull. Math. Biol., 72, 931–952.
Lipsitch, M., et al. (2003). Transmission dynamics and control of severe acute respiratory syndrome. Science, 300, 1966–1970. doi:10.1126/science.1086616.
Lu, L., Jia, M. H., et al. (2008). The changing face of HIV in China. Nature, 455, 609–611.
Martin, C., Bootsma, J., & Ferguson, N. M. (2007). The effect of public health measures on the 1918 influenza pandemic in U.S. cities. Proc. Natl. Acad. Sci. USA, 104(18), 7588–7593.
Meza, M. E. M., Bhaya, A., Kaszkurewicz, E. K., Silveira, D. A., & Costa, M. I. (2005). Threshold policies control for predator–prey systems using a control Liapunov function approach. Theor. Popul. Biol., 67, 273–284.
Meza, M. E. M., Costa, M. I. S., Bhaya, A., & Kaszkurewicz, E. (2002). Threshold policies in the control of predator–prey models. In Preprints of the 15th triennial world congress (IFAC) (pp. 1–6). Barcelona, Spain.
Peiris, J. S. M., Guan, Y., & Yuen, K. Y. (2004). Severe acute respiratory syndrome. Nat. Med., 10(Suppl. 12), S88.
Pourbohloul, B., Meyers, L. A., Skowronski, D. M., Krajden, M., Patrick, D. M., & Brunham, R. C. (2005). Modeling control strategies of respiratory pathogens. Emerg. Infect. Dis., 11, 1249–1256.
Quinn, T. J., & Deriso, R. B. (2000). Biological resource management series. Quantitative fish dynamics (pp. 439–442). Oxford: Oxford University Press.
Sun, C., Yang, W., Arino, J., & Khan, K. (2011). Effect of media-induced social distancing on disease transmission in a two patch setting. Math. Biosci. doi:10.1016/j.mbs.2011.01.005.
Skowronski, D. M., Astell, C., Brunham, R. C., et al. (2005). Severe acute respiratory syndrome (SARS): a year in review. Annu. Rev. Med., 56, 357–381.
Tang, S. Y., Xiao, Y. N., et al. (2010). Community-based measures for mitigating the 2009 H1N1 pandemic in China. PLoS ONE, 5, 1–11 (e10911).
Tang, S. Y., Xiao, Y. N., Yuan, L., Cheke, R., & Wu, J. (2012). Campus quarantine (Fengxiao) for curbing emergent infectious diseases: lessons from mitigating A/H1N1 in Xi’an, China. J. Theor. Biol., 295, 47–58.
Tchuenche, J. M., Dube, N., Bhunu, C. P., Smith, R. J., & Bauch, C. (2011). The impact of media coverage on the transmission dynamics of human influenza. BMC Public Health, 11(Suppl. 1), S5.
Utkin, V. I. (1978). Sliding modes and their applications in variable structure systems. Moscow: Mir.
Utkin, V. I. (1992). Sliding modes in control and optimization. Berlin: Springer.
Wang, W. (2006). Backward bifurcation of an epidemic model with treatment. Math. Biosci., 201, 58–71.
Wang, X., Xiao, Y., Wang, J., & Lu, X. (2012). A mathematical model of effects of environmental contamination and presence of volunteers on hospital infections in China. J. Theor. Biol., 293, 161–173.
World Health Organization. (2005). Combating emerging infectious diseases in the South-East Asia region. Regional office for New Delhi.
Xiao, Y., & Tang, S. (2010). Dynamics of infection with nonlinear incidence in a simple vaccination model. Nonlinear Anal., Real World Appl., 11, 4154–4163.
Xiao, Y., Zhou, Y., & Tang, S. (2011). Modelling disease spread in dispersal networks at two levels. IMA J. Math. Appl. Med. Biol., 28, 227–244.
Zhang, F., Pan, J., & Yu, L. (2005). Current progress of China’s free ART program. Cell Res., 15, 877–882.
Acknowledgements
The authors thank Professor R.A. Cheke for his kind help and comments and thank Professor Jaume Llibre for discussing the Lemma in Appendix B. The authors are supported by the National Mega-project of Science Research No. 2012ZX10001-001, by the National Natural Science Foundation of China (NSFC, 11171268(YX), 11171199(ST)), and by the Fundamental Research Funds for the Central Universities (08143042 (YX), GK 201003001 (ST)). The authors thank the anonymous referees for useful comments.
Author information
Authors and Affiliations
Corresponding author
Appendices
Appendix A: Mathematical Definition of the Threshold Policy
A threshold policy can be defined as follows: Control is suppressed when a state variable is below a previously chosen threshold density; above the threshold, control is applied (Utkin 1978). A TP leads to a variable structure system with two distinct structures. In mathematical terms (choosing a plane system as an example for illustrative purposes), it can be written as
where Z (Z∈R 2) is the state vector, f∈C(R 2,R 2) is a continuous function, and the control u τ is defined as
where u 1 is a continuous function, τ(Z):R 2→R is a threshold dependent on the state vector, and u τ is discontinuous. The ‘controlled system’ is one in which the control u τ =u 1 is applied, and the ‘free system’ is one in which no control (i.e. u τ =0) is applied. The manifold Σ is defined as
then Σ is the set of points in R 2. Let
Definition 1
Let \(Z_{G^{i}}^{eq}\) be such that \(f^{i}(Z_{G^{i}}^{eq}, u_{i})=0\) for some u i in (20). Then \(Z_{G^{i}}^{eq}\) is called a real equilibrium if it belongs to G i and a virtual equilibrium if it belongs to G j,j≠i, where f i(Z,u i ) corresponds to the function f(Z,u τ ) in the region G i.
A ‘sliding mode’ exists if there are regions in the vicinity of manifold Σ where the vectors f 1(Z,0) and f 2(Z,u 1) are directed toward each other.
Equivalent Control Method
Using a method developed by Utkin (1992), we describe how to obtain equations describing sliding mode dynamics along the manifold Σ for system (20). Assume that a sliding mode exists on manifold Σ given in (22), we now find a continuous control such that, given an initial condition of the state vector on this manifold, it yields identical equality to zero of the time derivative of vectors τ(Z) along trajectories of system (20):
which implies that a motion starting in τ(Z(t 0))=0 will proceed along the trajectories that lie on the manifold Σ.
Suppose that a solution of the system of the algebraic equation (23) with respect to control does exist. This solution, referred to as ‘equivalent control’ \(\bar{u}(Z, t)\), is substituted for u τ in system (20)
This equation is regarded as the ‘sliding mode dynamics’ describing the reduced-order motion on the discontinuity surface Σ. If the sliding mode dynamics have a stable equilibrium, then it is referred to as the sliding equilibrium or equilibrium attained through a sliding mode. Note, however, that it is not necessary that the sliding mode dynamics present a stable equilibrium (Boukal and Krivan, 1999; Krivan, 1996, 1998). The above procedure will be called the ‘equivalent control method’.
Appendix B: Non-Existence of Limit Cycle
In the case of smooth dynamical systems, the Dulac function is used to prove non-existence of limit cycles, it could be applicable to model (1) with (2) where the vector field is neither smooth nor continuous at the line I=I c . Indeed, denote the right-hand sides of the first two equations of model (1) by f 1 and f 2, then we have
Lemma
If there is a continuous function D in int \(R_{+}^{2}\) such that D is continuously differentiable when I≠I c , and
then system (1) with (2) does not have a limit cycle.
Proof
Suppose that there exists a limit cycle Γ (shown in Fig. 4) which surrounds the regular equilibrium \(E_{R}^{2}\) and the sliding mode \(\overline{AB}\), and crosses the manifold Σ with a period T. Denote its part below I=I c by Γ 1 and its part above the line I=I c by Γ 2. Denote the intersection points of the limit cycle Γ and the line I=I c by A 1 and B 1, the intersection points of Γ and the auxiliary line I=I c −ϵ (I=I c +ϵ) by T 1 and T 2 (T 3 and T 4), where ϵ>0 is sufficiently small. Let G 1 be the region bounded by Γ 1 and the segment T 1 T 2, G 2 be the region bounded by Γ 2 and the segment T 3 T 4. We denote the boundary of G 1 and G 2 by L 1 and L 2, respectively, with the directions indicated in the figure. In particular, denote the right-hand side of (1) in the region of I<I c (or I>I c ) by f 1(x) (or f 2(x)) where \(f^{i}(x)=(f_{1}^{i}(x), f_{2}^{i}(x))\), i=1,2.
Phase plane S–I for SIR model without demography, showing the switching line (\(\overline{PQ}\)), sliding domain (\(\overline{AB}\)) and the typical orbits (pink curves) for Case (1). Parameter values are γ=0.3; β=1.1; I c =0.4. (a) f=0.4; (b) f=0.55. Curve \(\widetilde{AC}\) (pink) is the integral curve passing the point A in the region I>I c , and curves \(\widetilde{DAF}\) (pink), \(\widetilde{BE}\) (pink) are integral curves passing the point A and B in the region I<I c , respectively. The thin blue curves represent the general orbits in the phase plane (Color figure online)
Phase plane S–I for SIR model without demography, showing the switching line (\(\overline{PQ}\)), sliding domain (\(\overline{AB}\)) and the typical orbits (pink curves). Parameter values are γ=0.2; β=1; f=0.7; I c =0.5. (a): I c =0.5 for Case (2); (b): I c =0.85 for Case (3). The thick pink curves in (a) are defined in the Fig. 5 and the thin blue curves represent the general orbits in the phase plane. The thick pink curve in (b) is an integral curve passing point Q (Color figure online)
Let the Dulac function be D=1/(SI). By Green’s theorem, we have

Note that Γ 1 is part of the limit cycle Γ of system (1) with (2) in the region of I<I c and let t 1 and t 2 be the timings at which the orbit Γ 1 passes the point T 1 and T 2 in the phase plane S–I, respectively. Similarly, we have

Let G 0⊂G 1 and \(\xi={{\int\hspace{-0.2cm}\int}_{G_{0}}} (\frac{\partial(Df_{1}^{1})}{\partial S}+\frac{ \partial(Df_{2}^{1})}{ \partial I} )\,dS\,dI\), then we know ξ<0 from the condition of the lemma, and furthermore,

which is
Suppose the abscissas of the points A 1,B 1,T 1,T 2,T 3,T 4 are \(\underline{S}, \overline{S}, \underline{S}+h_{1}(\epsilon), \overline{S}-h_{2}(\epsilon), \underline{S}+h_{4}(\epsilon), \overline{S}-h_{3}(\epsilon)\), respectively, where h i (ϵ) (i=1,2,3,4) is continuous with respect to ϵ and satisfies h i (0)=0 and lim ϵ→0 h i (ϵ)=0. Thus, we get

Similarly, we have

Therefore,
which contradicts (25). This rules out the existence of the limit cycle Γ surrounding the sliding mode and the regular equilibrium. □
Appendix C: Duration of Interventions Being Implemented
It is easy to obtain the number of susceptibles S 1 when the number of infecteds reaching the threshold level (here I c =I max), where S 1 satisfies
Integrating the sliding dynamics (13) from (S 1,I c ) along the sliding mode yields
When a trajectory slides from (S 1,I c ) to (γ/(β(1−f 1)),I c ), the time duration gives
where f 1 is given in (18).
Rights and permissions
About this article
Cite this article
Xiao, Y., Xu, X. & Tang, S. Sliding Mode Control of Outbreaks of Emerging Infectious Diseases. Bull Math Biol 74, 2403–2422 (2012). https://doi.org/10.1007/s11538-012-9758-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11538-012-9758-5