Abstract
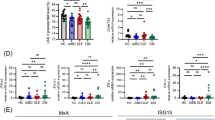
Lichen planus (LP) is a common inflammatory skin disease of unknown etiology. Reports of a common transactivation of quiescent human endogenous retroviruses (HERVs) support the connection of viruses to the disease. HERVs are ancient retroviral sequences in the human genome and their transcription is often deregulated in cancer and autoimmune diseases. We explored the transcriptional activity of HERV sequences as well as the antiviral restriction factor and interferon-inducible genes in the skin from LP patients and healthy control (HC) donors. The study included 13 skin biopsies from patients with LP and 12 controls. Real-time PCR assay identified significant decrease in the HERV-K gag and env mRNA expression levels in LP subjects, when compared to control group. The expressions of HERV-K18 and HERV-W env were also inhibited in the skin of LP patients. We observed a strong correlation between HERV-K gag with other HERV sequences, regardless the down-modulation of transcripts levels in LP group. In contrast, a significant up-regulation of the cytidine deaminase APOBEC 3G (apolipoprotein B mRNA-editing), and the GTPase MxA (Myxovirus resistance A) mRNA expression level was identified in the LP skin specimens. Other transcript expressions, such as the master regulator of type I interferon-dependent immune responses, STING (stimulator of interferon genes) and IRF-7 (interferon regulatory factor 7), IFN-β and the inflammassome NALP3, had increased levels in LP, when compared to HC group. Our study suggests that interferon-inducible factors, in addition to their role in innate immunity against exogenous pathogens, contribute to the immune control of HERVs. Evaluation of the balance between HERV and interferon-inducible factor expression could possibly contribute to surveillance of inflammatory/malignant status of skin diseases.



Similar content being viewed by others
References
Allen IC, Scull MA, Moore CB, Holl EK, McElvania-TeKippe E, Taxman DJ, Guthrie EH, Pickles RJ, Ting JP (2009) The NLRP3 inflammasome mediates in vivo innate immunity to influenza A virus through recognition of viral RNA. Immunity 30:556–565
Assinger A, Yaiw KC, Gottesdorfer I, Leib-Mosch C, Soderberg-Naucler C (2013) Human cytomegalovirus (HCMV) induces human endogenous retrovirus (HERV) transcription. Retrovirology 10:132
Bannert N, Kurth R (2004) Retroelements and the human genome: new perspectives on an old relation. Proc Natl Acad Sci USA 101:14572–14579
Belshaw R, Katzourakis A, Paces J, Burt A, Tristem M (2005) High copy number in human endogenous retrovirus families is associated with copying mechanisms in addition to reinfection. Mol Biol Evol 22:814–817
Bessis D, Moles JP, Basset-Seguin N, Tesniere A, Arpin C, Guilhou JJ (2004) Differential expression of a human endogenous retrovirus E transmembrane envelope glycoprotein in normal, psoriatic and atopic dermatitis human skin. Br J Dermatol 151:737–745
Crincoli V, Di Bisceglie MB, Scivetti M, Lucchese A, Tecco S, Festa F (2011) Oral lichen planus: update on etiopathogenesis, diagnosis and treatment. Immunopharmacol Immunotoxicol 33:11–20
De Vries HJ, van Marle J, Teunissen MB, Picavet D, Zorgdrager F, Bos JD, Weel J, Cornelissen M (2006) Lichen planus is associated with human herpesvirus type 7 replication and infiltration of plasmacytoid dendritic cells. Br J Dermatol 154:361–364
Dewannieux M, Blaise S, Heidmann T (2005) Identification of a functional envelope protein from the HERV-K family of human endogenous retroviruses. J Virol 79:15573–15577
Esnault C, Heidmann O, Delebecque F, Dewannieux M, Ribet D, Hance AJ, Heidmann T, Schwartz O (2005) APOBEC3G cytidine deaminase inhibits retrotransposition of endogenous retroviruses. Nature 433:430–433
Gao S, von der Malsburg A, Dick A, Faelber K, Schroder GF, Haller O, Kochs G, Daumke O (2011) Structure of myxovirus resistance protein a reveals intra- and intermolecular domain interactions required for the antiviral function. Immunity 35:514–525
Garcia-Montojo M, de la Hera B, Varade J, de la Encarnacion A, Camacho I, Dominguez-Mozo M, Arias-Leal A, Garcia-Martinez A, Casanova I, Izquierdo G, Lucas M, Fedetz M, Alcina A, Arroyo R, Matesanz F, Urcelay E, Alvarez-Lafuente R (2014) HERV-W polymorphism in chromosome X is associated with multiple sclerosis risk and with differential expression of MSRV. Retrovirology 11:2
Hohn O, Hanke K, Bannert N (2013) HERV-K (HML-2), the best preserved family of HERVs: endogenization, expression, and implications in health and disease. Front Oncol 3:246
Ishikawa H, Ma Z, Barber GN (2009) STING regulates intracellular DNA-mediated, type I interferon-dependent innate immunity. Nature 461:788–792
Kowalczyk MJ, Danczak-Pazdrowska A, Szramka-Pawlak B, Zaba R, Silny W, Osmola-Mankowska A (2012) Expression of selected human endogenous retroviral sequences in skin and peripheral blood mononuclear cells in morphea. Arch Med Sci 8:819–825
Lee WJ, Kwun HJ, Jang KL (2003) Analysis of transcriptional regulatory sequences in the human endogenous retrovirus W long terminal repeat. J Gen Virol 84:2229–2235
Lee YN, Malim MH, Bieniasz PD (2008) Hypermutation of an ancient human retrovirus by APOBEC3G. J Virol 82:8762–8770
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25:402–408
Lodi G, Giuliani M, Majorana A, Sardella A, Bez C, Demarosi F, Carrassi A (2004) Lichen planus and hepatitis C virus: a multicentre study of patients with oral lesions and a systematic review. Br J Dermatol 151:1172–1181
Maliniemi P, Vincendeau M, Mayer J, Frank O, Hahtola S, Karenko L, Carlsson E, Mallet F, Seifarth W, Leib-Mosch C, Ranki A (2013) Expression of human endogenous retrovirus-w including syncytin-1 in cutaneous T-cell lymphoma. PLoS One 8(10):e76281
Manghera M, Douville RN (2013) Endogenous retrovirus-K promoter: a landing strip for inflammatory transcription factors? Retrovirology 10:16
Mariani R, Chen D, Schrofelbauer B, Navarro F, Konig R, Bollman B, Munk C, Nymark-McMahon H, Landau NR (2003) Species-specific exclusion of APOBEC3G from HIV-1 virions by Vif. Cell 114(1):21–31
Moles JP, Tesniere A, Guilhou JJ (2005) A new endogenous retroviral sequence is expressed in skin of patients with psoriasis. Br J Dermatol 153:83–89
Moyes DL, Martin A, Sawcer S, Temperton N, Worthington J, Griffiths DJ, Venables PJ (2005) The distribution of the endogenous retroviruses HERV-K113 and HERV-K115 in health and disease. Genomics 86:337–341
Nagata S, Hanayama R, Kawane K (2010) Autoimmunity and the clearance of dead cells. Cell 140:619–630
Pereira NZ, Cardoso EC, Oliveira LM, de Lima JF, Branco AC, Ruocco RM, Zugaib M, de Oliveira Filho JB, Duarte AJ, Sato MN (2013) Upregulation of innate antiviral restricting factor expression in the cord blood and decidual tissue of HIV-infected mothers. PLoS One 8(12):e84917
Rebora A (2005) Human endogenous retroviruses and their possible impact on dermatology. J Am Acad Dermatol 52(5):E7
Shaker OG, Hantar N, El-Tahlawi S, El-Tawdi A, El-Hadidi H, Hantar S, El-Refai A, William R (2009) Detection of myxovirus resistance protein A in lichen planus lesions and its relationship to hepatitis C virus. Br J Dermatol 160:980–983
Sharma A, Bialynicki-Birula R, Schwartz RA, Janniger CK (2012) Lichen planus: an update and review. Cutis 90:17–23
Singh S, Kaye S, Gore ME, McClure MO, Bunker CB (2009) The role of human endogenous retroviruses in melanoma. Br J Dermatol 161:1225–1231
Sutkowski N, Conrad B, Thorley-Lawson DA, Huber BT (2001) Epstein-Barr virus transactivates the human endogenous retrovirus HERV-K18 that encodes a superantigen. Immunity 15:579–589
Turcanova VL, Bundgaard B, Hollsberg P (2009) Human herpesvirus-6B induces expression of the human endogenous retrovirus K18-encoded superantigen. J Clin Virol 46:15–19
Wagner G, Rose C, Sachse MM (2013) Clinical variants of lichen planus. J Dtsch Dermatol Ges 11:309–319
Acknowledgments
We are grateful to all individuals who participated in the study. This work was supported by Fundação de Amparo a Pesquisa do Estado de São Paulo (2011/20740-3) and the Laboratório de Investigação Médica, Unidade 56 do Hospital das Clínicas da Faculdade de Medicina de São Paulo.
Conflict of interest
None declared.
Author information
Authors and Affiliations
Corresponding author
Additional information
M. A. de Sousa Nogueira and C. F. Biancardi Gavioli contributed equally to this work.
Rights and permissions
About this article
Cite this article
de Sousa Nogueira, M.A., Biancardi Gavioli, C.F., Pereira, N.Z. et al. Human endogenous retrovirus expression is inversely related with the up-regulation of interferon-inducible genes in the skin of patients with lichen planus. Arch Dermatol Res 307, 259–264 (2015). https://doi.org/10.1007/s00403-014-1524-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00403-014-1524-0