Abstract
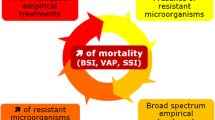
Sepsis in the intensive care unit (ICU) presents a great challenge to any critical care clinician. Patients admitted to the ICU are especially vulnerable to sepsis due to the nature of the underlying pathology that warranted admission to the ICU and deranged physiological function coupled with invasive procedures. Nosocomial infections are common in patients admitted to the ICU, and with these infections come the burden of multidrug-resistant organisms. Antimicrobial resistance (AMR) is now a global emergency that warrants the attention of every health-care professional. AMR has escalated to epic proportions and solutions to this problem are now a matter of “life and death.” The ICU also represents the “breeding ground” of antibiotic-resistant organisms due to the high broad-spectrum antibiotic consumption. Many would argue that broad-spectrum antimicrobials are overprescribed in this patient population, but do all patients admitted to the ICU warrant such therapy? Is there evidence that narrower-spectrum antimicrobial agents can be employed in specific ICU populations coupled with surveillance strategies? The aims of this review are to focus on strategies with the aim of optimizing antimicrobial use within ICUs, and to highlight the importance of differentiating ICU populations with regard to the use of antimicrobial agents.
Similar content being viewed by others
Introduction
Never before has the term “run out of options” been so relevant and life-threatening. With the continuing and relentless development of antimicrobial resistance, time is rapidly running out to find possible solutions to this colossal problem. Our options for future antimicrobial development are limited with less time, effort, and money dedicated to antimicrobial research and development. Seventy years ago, Sir Alexander Fleming correctly predicted the development of antimicrobials when he stated, “The intensive research which penicillin has stimulated may bring forth others as good, or even better, or the chemists may be able to modify the penicillin molecule so that its power is increased or its limitations are removed. There is still plenty of scope for further advance” [1]. While Fleming’s prediction that there is scope for further advance in the manufacture of new antimicrobials undoubtedly held true in the first half of the twentieth century, the same cannot be said for now [2]. Hence, it is of paramount importance that we conserve what we have and need to protect those new antibiotics from misuse when they do become available. With the increasing antimicrobial resistance and reduced antibiotic development, there is an urgent call for better use of currently available antimicrobial agents.
Antimicrobial use in the intensive care unit
Due to the extremely vulnerable nature of critically ill patients admitted to an intensive care unit (ICU), and the frequent use of invasive procedures, the ICU is the epicenter of infections [3]. Early and appropriate antimicrobial administration is paramount in critically ill patients with suspected or confirmed infection and sepsis although the timing of initial administration has become debatable [4].
Time saves lives but more haste less speed
Initial results from studies that have been conducted from the Surviving Sepsis Campaign suggested that in-hospital mortality risk increased linearly for every hour delay before the administration of the first antimicrobial dose [5, 6], and the first antimicrobial dose should be administered within 1 h after the onset of hypotension in sepsis. Such studies, however, were retrospective over a considerable time period in various ICUs without any resuscitation or antimicrobial protocols. Recent data suggest a more liberal time frame of within 3 h for the first dose without compromising outcome. Although controversial, this may make physiological sense for, following the first few hours of resuscitation, improved hemodynamics should allow optimal delivery of the drug, whereas if administered during the phase of hypoperfusion, there may be suboptimal pharmacokinetics and dynamics.
Appropriate dosing and dosing strategies of antimicrobial agents in the ICU
Critically ill patients also present with profound physiological dysfunction, altering the pharmacokinetics of the administered antimicrobials, and failure to achieve target serum concentration is not uncommon [7]. Appropriate dosing of especially the first dose is of paramount importance. Most often there is a need to administer a loading dose of the antimicrobial agent as these patients have an increased volume of distribution and conventional antimicrobial dosing results in subtherapeutic antimicrobial serum concentration. The dosing strategy for antibiotics should take into account the mode of action of the drug and individual patient characteristics that influence pharmacodynamics and pharmacokinetic factors, in order to maximize bacterial killing, prevent the development of antimicrobial resistance, and avoid concentration-related adverse drug reactions [5]. Hence, correct dosing and dosing intervals of antimicrobials become a huge challenge in an attempt to treat infections within an ICU successfully.
The antimicrobial effects observed for concentration-dependent agents such as aminoglycosides, colistin, quinolones, and vancomycin are maximal when the free drug peak concentration in a dosing interval exceeds the minimum inhibitory concentration (MIC) by 8–10 times (Cmax/MIC > 8–10). This has been translated into single daily or extended interval dosing for aminoglycosides [8].
For time-dependent agents such as beta-lactams, the killing effect is almost entirely related to the time for which levels in tissue and plasma exceed the MIC of the offending pathogen (fT>MIC). Penicillin and monobactams are reported to require at least 50–60 % fT>MIC, cephalosporins need a 60–70 % fT>MIC, whereas carbapenems require a 40 % fT>MIC [9]. Time-dependent antibiotics lack a post-antibiotic effect, and, once the levels fall below the MIC, bacterial growth resumes, resulting in treatment failure and bacterial resistance. Hence, dosing regimens for beta-lactams are being re-evaluated, and at least for critically ill patients with resistant pathogens, extended or continuous dosing is recommended [10]. There has been improved therapeutic outcomes with modifying antibiotic delivery in order to improve the probability of obtaining fT>MIC.
For concentration- and time-dependent agents such as quinolones, daptomycin, glycopeptides, tigecycline, and linezolid, the antimicrobial effect is defined by the area under the curve (AUC) of free drug over a 24-h period over the MIC [5]. For example, contemporary vancomycin dosing regimens target an AUC/MIC ≥ 400 for serious methicillin-resistant Staphylococcus infections [11].
Selection of antimicrobial agent—know the indigenous flora of your ICU
Due to the complexity of patients admitted to an ICU, managing possible infections become extremely difficult. In an attempt to properly manage patients, clinicians often find themselves causing direct harm such as inappropriate initial therapy, or indirect harm such as prolonged antibiotic exposure resulting in the emergence of multidrug-resistant pathogens with their subsequent spread within the ICU [12]. Unnecessary administration of antimicrobial therapy not only impacts on the individual patient but also on those patients in the same ICU environment. When faced with an extremely vulnerable patient with a possible life-threatening infection, it can be very daunting even for the most experienced intensivist. The possibility of “what if “becomes the driving force behind why most ICU clinicians adopt the use of ultra-broad spectrum antimicrobials to cover “all possible pathogens.” Kollef makes a salient statement that the empiric choice of antimicrobials should cover the most likely pathogens endemic to the specific location [13]. It is important to note that the statement does not say all pathogens, and prescriptions cannot be based on uncommon organisms unless the situation dictates the need [2]. Regular surveillance enables clinicians to make a more informed decision with regard to empiric antimicrobial choices. Knowledge of inherent flora and their antimicrobial susceptibility patterns are crucial in this regard. Surgical source control in patients with an obvious infectious site is imperative. Antimicrobial therapy alone will be ineffective for a surgical source but works synergistically with source control strategies that aim to physically remove an infectious nidus.
De-escalation strategies
Most ICUs adopt a de-escalation strategy which involves administration of ultra-broad spectrum antimicrobials as initial empiric antimicrobial therapy. This strategy is meant to provide coverage for the vast majority of the most likely pathogens. Thereafter, this initial antibiotic treatment is modified based on microbiological analysis of appropriate specimens. Such empiric therapy should, however, always be modified once the agent of infection is identified or discontinued altogether if the diagnosis of infection becomes unlikely [14]. De-escalation of antibiotic therapy can be thought of as a strategy to balance the need to provide adequate initial antibiotic treatment of high-risk patients with the avoidance of unnecessary antibiotic utilization, which promotes resistance [14]. Unfortunately, more often than not, clinicians are not willing to modify initial empiric therapy based on the premise that “the patient has responded so why not continue.” This “resistant behaviour/refusal to comply” on the part of clinicians has assisted in the propagation of antimicrobial resistance.
Combination antimicrobial agents
Combination antimicrobial therapy involves the administration of usually two antimicrobial agents from different classes, in order to achieve a synergistic effect. Over the years, the topic of combination therapy has been controversial and contested. Most studies evaluating the benefit of combination therapy have not shown a survival benefit, the only exception being Pseudomonas bacteremia and carbapenemase-producing Klebsiella pneumoniae bacteremia [15–19]. However, in critically ill patients, combination therapy may be appropriate for empiric treatment, especially in cases where infections due to resistant organisms are suspected. This increases coverage of pathogens. Patient risk factors for colonization or infection with multidrug-resistant pathogens should be taken into account, including recent antibiotic use and hospitalization, prolonged hospital stay, dialysis, and the presence of invasive devices [14].
Duration
Biomarkers such as procalcitonin (PCT) have been used by many ICUs in the initiation and de-escalation of antimicrobial therapy. PCT has some diagnostic and prognostic utility in the management of critically ill patients, since it is elevated in patients with severe sepsis, septic shock, and bacteraemia, and its decline is associated with better prognosis [20]. PCT-based algorithms are associated with shorter duration of antibiotic treatment without compromising ICU outcomes [21].
Provided that the infection source is controlled, short antibiotic courses (<7 days) are sufficient for most infections in the critically ill patient, with a few exceptions [22]. More specifically as demonstrated by Pugh et al., shorter (3–8 days) rather than longer (10–21 days) antimicrobial courses have been shown to be equally efficacious in the treatment of ventilator-associated pneumonia (VAP). In patients with VAP not due to non-lactose fermenting Gram negative organisms, a short, fixed course of 7 or 8 days of antibiotic therapy appears not to increase the risk of adverse clinical outcomes, and may reduce the emergence of resistant organisms, compared to a prolonged course of 10–15 days [23].
Colonization versus infection
It is also vital to differentiate colonization from true infection. This is commonly seen with Acinetobacter. Most Acinetobacter species are inherently resistant to all but a few antimicrobials, and embarking on therapy would necessitate the use of ultra-broad spectrum antimicrobial agents and the risk of treating colonization rather than true infection, which in turn would lead to emergence of other MDR organisms [2]. A recent study conducted in a TICU in Durban, South Africa revealed that although MDR Acinetobacter baumannii was the most common organism isolated in late VAP, the vast majority of patients did not receive antimicrobial therapy for the MDR A. baumannii [24]. Owing to its natural multidrug resistance, low virulence, and pathogenicity, the unit’s policy is not to treat MDR Acinetobacter unless it is the sole pathogen in possible VAP that fails to improve. Of the 43 patients in whom MDR A. baumannii was cultured, only six were offered therapy, mainly in the form of inhaled/nebulized amikacin. There was one fatality due to uncontrolled retroperitoneal fasciitis following a fecal leak [24]. Based on these data, we suggest that treating all Acinetobacter isolates is unnecessary, and will promote bacterial resistance. In addition, Acinetobacter has the ability to develop resistance even during directed therapy [25], and the isolation of subpopulations that are resistant to colistin is of serious concern. Although ethically debatable, this practice conforms to the principle of social justice over autonomy and complements the view of Mendelson et al. [26] that inappropriate and unnecessary prescriptions of broad-spectrum antimicrobials may ultimately result in an era of untreatable bacterial infections.
Antimicrobial stewardship and surveillance
So how then does one optimize antimicrobial therapy and practice judicious use of antimicrobials without compromising the health and safety of the patient? The answer lies in antimicrobial stewardship programs (ASPs) as this may provide a possible solution to the increasing problem of multidrug-resistant organisms.
ASPs aim to optimize appropriate antibiotic treatment while minimizing antibiotic resistance. Antimicrobial stewardship is a multifaceted approach primarily aimed to ensure that infections are treated in a timely manner with appropriate and effective empiric regimens, that the use of unnecessary antibiotics is minimized, and that complications associated with the use of antimicrobial agents are avoided [12]. Surveillance is also a key element in combating this problem of multidrug-resistant pathogens. Studies have demonstrated that knowledge of flora, local resistance patterns, and an antimicrobial policy that optimizes the choice, dose, and duration of AQtherapy can be of assistance for availing correct empiric antimicrobial choices [2].
ASPs are initiated by an antimicrobial stewardship team at a healthcare institution. Members that make up this multidisciplinary team include intensivists (e.g., trauma surgeons trained in intensive care), infectious disease specialists, microbiologists, pharmacists, hospital management, and infection control nurses, along with the treating surgeons in the case of trauma patients. The presence of surgeons are important since surgical sepsis may require operative source-control, minimally invasive procedures, or local wound debridement as part of the appropriate care of the trauma patient. Local and national protocols and guidelines should be established, with input from all stakeholders, providing basic concepts and practices that will facilitate the implementation and success of ASPs in the ICU, as well as throughout the hospital [27]. The impact of the ASP should be regularly evaluated to determine its overall effectiveness and need for updating [27].
ASPs should be integrated into an overall program aimed at reducing antimicrobial resistance and its impact on patient outcomes. This should go hand in hand with good infection-control practices. Infection control and hand hygiene play a pivotal with ASPs in combating multidrug-resistant organisms and the spread thereof. With regard to initial empiric antimicrobial therapy in a given ICU, patient location and population play respective roles. A study conducted at a level-1 trauma intensive care unit (TICU) in Durban, South Africa [2] has demonstrated that the population admitted to the TICU was fairly unique compared to other ICU (Medical and Surgical ICU) populations. Patients admitted to the TICU were younger individuals without pre-existing medical co-morbidities. The majority of the patients were antibiotic naive having never received any prior antimicrobial therapy for pre-existing illnesses and were only admitted to the TICU by virtue of their serious life-threatening injuries that warranted mechanical ventilation. It was found that in the majority of patients who presented with nosocomial infection, the offending pathogen was community acquired or a pathogen that was susceptible to narrower spectrum agents. There was no need to employ the use of ultra-broad antimicrobial therapy in the vast majority of patients even at the time of starting empiric therapy. The unit employs an antimicrobial policy that is informed by regular surveillance carried out in the TICU. The study demonstrated that the choice of empiric antimicrobials was correct in more than 90 % of instances, indicating that frequent surveillance and stewardship promote a more rational and restricted policy of antimicrobial use and an infrequent resort to ultra-broad spectrum antimicrobial therapy [2]. The study suggests that in this unique patient population admitted specifically to a TICU who have had no prior antimicrobial therapy, blanket cover with ultra-broad spectrum antimicrobial therapy is rarely indicated in the presence of an active surveillance program and empiric antimicrobial policy. Can this strategy be adopted in other exclusive TICUs? Geographical location and local resistance patterns need to be considered before employing such a strategy. Based on this study, it raises the question in view of the the fact that patients admitted to the TICU are different with regard to flora, having no prior medical co-morbidities and no prior antimicrobial exposure: can this unique population of patients be managed differently from other ICU patient populations?
Conclusion
The issue of antimicrobial resistance can no longer be ignored. It is time that healthcare professionals make a concerted effort to address the issue of multidrug-resistant organisms and inappropriate antimicrobial prescribing. This review provides a guide on antimicrobial use in the ICU and also provides scientific evidence that narrower spectrum antimicrobials may be used in specific patient populations. The use of empiric broad-spectrum antimicrobials may not be necessary in all patients admitted to the ICU. This coupled with antimicrobial stewardship and surveillance may provide a solution to the problem of AMR. Failure to address this colossal problem responsibly and swiftly may result in a post-antibiotic era where even simple infections may be untreatable.
References
Fleming A (1946) History and development of penicillin. In: Fleming A (ed) Penicillin. Butterworth and Co, London, pp 1–23
Ramsamy Y, Muckart DJJ, Han KSS (2013) Microbiological surveillance and antimicrobial stewardship minimise the need for ultrabroad-spectrum combination therapy for treatment of nosocomial infections in a trauma intensive care unit: an audit of an evidence-based empiric antimicrobial policy. S Afr Med J 103(6):371–376
Brusselaers N, Vogelaers D, Blot S (2011) The rising problem of antimicrobial resistance in the intensive care unit. Ann Intensive Care 1(47):1–7
Sterling SA, Miller WR, Pryor J et al (2015) The impact of timing of antibiotics on outcomes in severe sepsis and septic shock: a systematic review and meta-analysis. Crit Care Med 43:1907–1915
Kumar A, Roberts D, Wood KE et al (2006) Duration of hypotension before initiation of effective antimicrobial therapy is the critical determinant of survival in human septic shock. Crit Care Med 34(6):1589–1596
Ferrer R, Martin-Loeches I, Phillips G et al (2014) Empiric antibiotic treatment reduces mortality in severe sepsis and septic shock from the first hour: results from a guideline-based performance improvement program. Crit Care Med 42(8):1749–1755
Roberts JA, Lipman J (2009) Pharmacokinetic issues for antibiotics in the critically ill patient. Crit Care Med 37(3):51–840
Barza M, Ioannidis JP, Cappelleri JC et al (1996) Single or multiple daily doses of aminoglycosides: a meta-analysis. BMJ 312:338–345
Craig WA (1998) Pharmacokinetic/pharmacodynamic parameters: rationale for antibacterial dosing of mice and men. Clin Infect Dis 26(1):1–10
Dulhunty JM, Roberts JA, Davis JS et al (2013) Continuous infusion of beta-lactam antibiotics in severe sepsis: a multicenter double-blind, randomized controlled trial. Clin Infect Dis 56(2):236–244
Liu C, Bayer A, Cosgrove SE et al (2011) Clinical practice guidelines by the infectious diseases society of america for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children: executive summary. Clin Infect Dis 52(3):285–292
Kollef MH, Micek ST (2012) Antimicrobial stewardship programs: mandatory for all ICUs. Crit Care 16(179):1–2
Kollef MH (2001) Hospital-acquired pneumonia and de-escalation of antimicrobial treatment. Crit Care Med 29:1473–1475
Kollef MH (2001) Optimizing antibiotic therapy in the intensive care unit setting. Crit Care 2001(5):189–195
Hilf M, Yu VL, Sharp J et al (1989) Antibiotic therapy for Pseudomonas aeruginosa bacteremia: outcome correlations in a prospective study of 200 patients. Am J Med 87(5):540–546
Paul M, Benuri-Silbiger I, Soares-Weiser K et al (2004) Beta lactam monotherapy versus beta lactam-aminoglycoside combination therapy for sepsis in immunocompetent patients: systematic review and meta-analysis of randomized trials. BMJ 328(7441):668–672
Leibovici L, Paul M, Poznanski O et al (1997) Monotherapy versus beta-lactam-aminoglycoside combination treatment for Gram-negative bacteremia: a prospective, observational study. Antimicrob Agents Chemother 41(5):1127–1133
Safdar N, Handelsman J, Maki DG (2004) Does combination antimicrobial therapy reduce mortality in Gram-negative bacteraemia? a meta-analysis. Lancet Infect Dis 4(8):519–527
Daikos GL, Tsaousi S, Tzouvelekis LS et al (2014) Carbapenemase-producing Klebsiella pneumoniae bloodstream infections: lowering mortality by antibiotic combination schemes and the role of carbapenems. Antimicrob Agents Chemother 58(4):2322–2328
Reinhart K, Meisner M (2011) Biomarkers in the critically ill patient: procalcitonin. Crit Care Clin 27(2):253–263
Schuetz P, Briel M, Christ-Crain M et al (2012) Procalcitonin to guide initiation and duration of antibiotic treatment in acute respiratory infections: an individual patient data meta-analysis. Clin Infect Dis 55(5):62–651
Lipman J, Boots R (2009) A new paradigm for treating infections: “go hard and go home”. Crit Care Resusc 11(4):276–281
Pugh R, Grant C, Cooke RP, Dempsey G (2015) Short-course versus prolonged-course antibiotic therapy for hospital-acquired pneumonia in critically ill adults. Cochrane Database Syst Rev 24(8):CD007577. doi:10.1002/14651858.CD007577.pub3
Ramsamy Y, Muckart DJJ, Bruce JL, Hardcastle TC et al (2016) Empirical antimicrobial therapy for probable v. directed therapy for possible ventilator-associated pneumonia in critically injured patients. S Afr Med J 106(2):196–200
Murray CK, Hospenthal DR (2008) Acinetobacter infection in the ICU. Crit Care Clin 24:237–248
Brink A, Coetzee J, Clay C et al (2012) The spread of carbapenem-resistant Enterobacteriaceae in South Africa: risk factors for acquisition and prevention. S Afr J Med 102:599–601
Society for Healthcare Epidemiology of America, Infectious Diseases Society of America, Pediatric Infectious Diseases Society (2012) Policy statement on antimicrobial stewardship by the Society for Healthcare Epidemiology of America (SHEA), the Infectious Diseases Society of America (IDSA), and the Pediatric Infectious Diseases Society (PIDS). Infect Control Hosp Epidemiol 33:322–327
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ramsamy, Y., Hardcastle, T.C. & Muckart, D.J.J. Surviving Sepsis in the Intensive Care Unit: The Challenge of Antimicrobial Resistance and the Trauma Patient. World J Surg 41, 1165–1169 (2017). https://doi.org/10.1007/s00268-016-3531-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-016-3531-0