Abstract
Key message
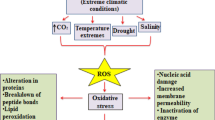
The study focuses on the interaction between reactive oxygen species and hormones that regulate the programmed cell death in plants of Melissa officinalis exposed to ozone.
Abstract
Interaction between hormone and redox signaling pathways has been investigated in ozone-stressed (200 ppb, 5 h) lemon balm to verify if the response resembles the biotic defense reactions. In comparison to controls, plants exhibited foliar injury and the cell death was induced by (1) biphasic production of hydrogen peroxide and superoxide radical; (2) hormonal regulation of ozone-induced lesion formation with a significant production of ethylene, salicylic, jasmonic and abscisic acid; (3) ozone degradation to reactive oxygen species and their detoxification by some enzymatic (such as superoxide dismutase) and non-enzymatic antioxidant systems (such as ascorbic acid, glutathione and carotenoids), that worked in cooperation without providing a defense against free radicals (such as confirmed by the modification of the antioxidant properties of leaf tissue). This integrated view showed that reactive oxygen species interact with hormonal signaling pathway regulating cell death and the sensitivity of lemon balm to ozone.









Similar content being viewed by others
References
Able AJ, Guest DI, Sutherland MW (1998) Use of a new tetrazolium-based assay to study the production of superoxide radicals by tobacco cell cultures challenged with avirulent zoospores of Phytophthora parasitica var nicotianae. Plant Physiol 117:491–499
Ahlfors R, Lång S, Overmyer K, Jaspers P, Brosché M, Tauriainen A, Kollist H, Tuominen H, Belles-Boix E, Piippo M, Inzé D, Palva ET, Kangasjärvi J (2004) Arabidopsis radical-induced cell death1 belongs to the WWE protein–protein interaction domain protein family and modulates abscisic acid, ethylene, and methyl jasmonate responses. Plant Cell 16:1925–1937
Alscher RG, Erturk N, Heath SL (2002) Role of superoxide dismutases (SODs) in controlling oxidative stress in plants. J Exp Bot 53:1331–1341
Bartoli CG, Casalongué CA, Simontacchi M, Marquez-Garcia B, Foyer CH (2013) Interactions between hormone and redox signalling pathways in the control of growth and cross tolerance to stress. Environ Exp Bot 94:73–88
Bent AF, Innes R, Ecker J, Staskawicz B (1992) Disease development in ethylene-insensitive Arabidopsis thaliana infected with virulent and avirulent Pseudomonas and Xanthomonas pathogens. Mol Plant Microbe Interact 5:372–378
Blomster T, Salojärvi J, Sipari N, Brosché M, Ahlfors R, Keinanen M, Overmyer K, Kangasjärvi J (2011) Apoplastic reactive oxygen species transiently decrease auxin signalling and cause stress-induced morphogenic response in Arabidopsis. Plant Physiol 157:1866–1883
Burden RS, Taylor HF (1970) The structure and chemical transformations of xanthoxin. Tetrahedron Lett 47:4071–4074
Castagna A, Ederli L, Pasqualini S, Mensuali-Sodi A, Baldan B, Donnini S, Ranieri A (2007) The tomato ethylene receptor LE-ETR3 (NR) is not involved in mediating ozone sensitivity: casual relationships among ethylene emission, oxidative burst and tissue damage. New Phytol 174:342–356
Chen Z, Gallie DR (2004) The ascorbic acid redox state controls guard cell signalling and stomatal movement. Plant Cell 16:1143–1162
Cheng H, Zhang Q, Guo D (2013) Genes that respond to H2O2 are also evoked under light in Arabidopsis. Mol Plant 6:226–228
Ciompi S, Castagna A, Ranieri A, Nali C, Lorenzini G, Soldatini FG (1997) CO2 assimilation, xanthophyll cycle pigments and PSII efficiency in pumpkin plants as affected by ozone fumigation. Physiol Plant 101:881–889
Collins A (2001) Carotenoids and genomic stability. Mutat Res 475:1–28
Conklin PL, Barth C (2004) Ascorbic acid, a familiar small molecule intertwined in the response of plants to ozone, pathogens, and the onset of senescence. Plant Cell Environ 27:959–970
Cooke MS, Evans MD, Dizdaroglu M, Lunec J (2003) Oxidative DNA damage: mechanisms, mutation and disease. FASEB J 17:1195–1214
Corral-Aguayo RD, Yahia EM, Carrillo-Lopez A, Gonzalez-Aguilar G (2008) Correlation between some nutritional components and the total antioxidant capacity measured with six different assays in eight horticultural crops. Food Nutr Bull 26:248–355
Cui X, Luan S (2012) A new wave of hormone research: crosstalk mechanisms. Mol Plant 5:959–960
Davison AW, Barnes JD (1998) Effects of ozone on wild plants. New Phytol 139:135–151
Devoto A, Turner JG (2005) Jasmonate-regulated Arabidopsis stress signalling network. Physiol Plant 123:161–172
Di Baccio D, Ederli L, Marabottini R, Badiani M, Francini A, Nali C, Antonelli M, Santangelo E, Sebastiani L, Pasqualini S (2012) Similar foliar lesions but opposite hormonal patterns in a tomato mutant impaired in ethylene perception and its near isogenic wild type challenged with ozone. Environ Exp Bot 75:286–297
Diara C, Castagna A, Baldan B, Mensuali Sodi A, Sahr T, Langebartels C, Sebastiani L, Ranieri A (2005) Differences in the kinetics and scale of signalling molecule production modulate the ozone sensitivity of hybrid poplar clones: the roles of H2O2, ethylene and salicylic acid. New Phytol 168:351–364
Durner J, Shah J, Klessig DF (1997) Salicylic acid and disease resistance in plants. Trends Plant Sci 2:162–165
Francini A, Nali C, Pellegrini E, Lorenzini G (2008) Characterization and isolation of some genes of the shikimate pathway in sensitive and resistant Centaurea jacea plants after ozone exposure. Environ Pollut 151:272–279
Fuhrer J, Booker FL (2003) Ecological issues related to ozone: agricultural issues. Environ Int 29:141–154
Gill SS, Tuteja N (2010) Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol Biochem 48:909–930
Grantz DA, Vu HB (2012) Root and shoot gas exchange respond additively to moderate ozone and methyl jasmonate without induction of ethylene: ethylene is induced at higher O3 concentrations. J Exp Bot 63:4303–4313
Grossmann K (2003) Mediation of herbicide effects by hormone interactions. J Plant Growth Regul 22:109–122
Heath RL (2008) Modification of the biochemical pathways of plants induced by ozone: what are the varied routes of change? Environ Pollut 155:453–463
Jiang M, Zhang J (2002) Water stress-induced abscisic acid accumulation triggers the increased generation of reactive oxygen species and up-regulates the activities of antioxidant enzymes in maize leaves. J Exp Bot 53:2401–2410
Kangasjärvi J, Talvinen J, Utrianen M, Karjalainen R (1994) Plant defense system induced by ozone. Plant Cell Environ 17:783–794
Kangasjärvi J, Jaspers P, Kollist H (2005) Signalling and cell death in ozone-exposed plants. Plant Cell Environ 28:1021–1036
Koch JR, Creelman RA, Eshita SM, Seskar M, Mullet JE, Davis KR (2000) Ozone sensitivity in hybrid poplar correlates with insensitivity to both salicylic acid and jasmonic acid. The role of programmed cell death in lesion formation. Plant Physiol 123:487–496
Kohler B, Hills A, Blatt MR (2003) Control of guard cell ion channels by hydrogen peroxide and abscisic acid indicates their action through alternate signalling pathways. Plant Physiol 131:385–388
Kohli A, Sreenivasulu N, Lakshmanan P, Kumar PP (2013) The phytohormone crosstalk paradigm takes center stage in understanding how plants respond to abiotic stress. Plant Cell Rep 32:945–957
Kramell R, Miersch O, Schneider G, Wasternack C (1999) Liquid chromatography of jasmonic acid amine conjugates. Chromatographia 49:42–46
Krupa S, McGrath MT, Andersen CP, Booker FL, Burkey KO, Chappelka AH, Chevone BI, Pell EJ, Zilinskas BA (2000) Ambient ozone and plant health. Plant Dis 85:4–12
Langebartels C, Kerner K, Leonardi S, Schraudner M, Trost M, Heller W, Sandermann H Jr (1991) Biochemical plant responses to ozone I. Differential induction of polyamine and ethylene biosynthesis in tobacco. Plant Physiol 95:882–889
Lee H, Léon L, Raskin I (1995) Biosynthesis and metabolism of salicylic acid. Prot Natl Acad Sci USA 92:4076–4079
Lomonte C, Sgherri C, Baker AJM, Kolev SD, Navari-Izzo F (2010) Antioxidative response of Atriplex codonocarpa to mercury. Environ Exp Bot 69:9–16
Mandal S, Kumar Das R, Mishra S (2011) Differential occurrence of oxidative burst and oxidative mechanism in compatible and incompatible interaction of Solanum lycopersicum and Ralstonia solanacearum. Plant Physiol Biochem 49:117–123
May MJ, Vernoux T, Leaver C, Montagu MV, Inzé D (1998) Glutathione homeostasis in plants: implications for environmental sensing and plant development. J Exp Bot 49:649–667
Mensuali Sodi A, Panizza M, Tognoni F (1992) Quantification of ethylene losses in different container-seal systems and comparison of biotic and abiotic contributions to ethylene accumulation in cultured tissues. Physiol Plant 84:472–476
Meyer AJ (2008) The integration of glutathione homeostasis and redox signalling. J Plant Physiol 165:1390–1403
Montillet JL, Chamnongpol S, Rustérucci C, Dat J, van de Cotte B, Agnel JP, Battesti C, Inzé D, Van Breusegem F, Triantaphylides C (2005) Fatty acid hydroperoxides and H2O2 in the execution of hypersensitive cell death in tobacco leaves. Plant Physiol 138:1516–1526
Mudd JB (1997) Biochemical basis for the toxicity of ozone. In: Yunus M, Iqbal M (eds) Plant response to air pollution. Wiley, New York, pp 267–284
Mur LAJ, Lloyd AJ, Cristescu SM, Harren FJM, Hall MA, Smith AR (2009) Biphasic ethylene production during the hypersensitive response in Arabidopsis. Plant Signal Behav 4:610–613
Ou B, Hampsch-Woodill M, Flanagan JA, Prior RL (2002) High-throughput assay of oxygen radical absorbance capacity (ORAC) using a multichannel liquid handling system coupled with a microplate fluorescence reader in 96-well format. J Agric Food Chem 50:4437–4444
Overmyer K, Tuomainen H, Kettunen R, Betz C, Langebartels C, Sandermann H Jr, Kangasjärvi J (2000) The ozone-sensitive Arabidopsis rcd1 mutant reveals opposite roles for ethylene and jasmonate signalling pathways in regulating superoxide-dependent cell death. Plant Cell 12:1849–1862
Overmyer K, Brosché M, Kangasjärvi J (2003) Reactive oxygen species and hormonal control of cell death. Trends Plant Sci 8:335–342
Overmyer K, Brosché M, Pellinen R, Kuittinen T, Tuominen H, Ahlfors R, Keinanen M, Saarma M, Scheel D, Kangasjärvi J (2005) Ozone-induced programmed cell death in the Arabidopsis radical-induced cell death1 mutant. Plant Physiol 137:1092–1104
Overmyer K, Kollist H, Tuominen H, Betz C, Langebartels C, Wingsle G, Kangasjärvi S, Brader G, Mullineaux P, Kangasjärvi J (2008) Complex phenotypic profiles leading to ozone sensitivity in Arabidopsis thaliana mutants. Plant Cell Environ 31:1237–1249
Pääkkönen E, Holopainen T, Kärenlampi L (1995) Effects of ozone on birch (Betula pendula Roth.) clones. Water Air Soil Pollut 85:1331–1336
Pasqualini S, Della Torre G, Ferranti F, Ederli L, Piccioni C, Reale L, Antonielli M (2002) Salicylic acid modulates ozone-induced hypersensitive cell death in tobacco plants. Physiol Plant 115:204–212
Pastori GM, Kiddle G, Antoniw J, Bernard S, Veljovic-Jovanovic S, Verrier PJ, Noctor G, Foyer CH (2003) Leaf vitamin C contents modulate plant defense transcripts and regulate genes that control development through hormone signalling. Plant Cell 15:939–951
Pei ZM, Murata Y, Benning G, Thomine S, Klusener B, Allen GJ, Grill E, Schroeder JI (2000) Calcium channels activated by hydrogen peroxide mediate abscisic acid signalling in guard cells. Nature 406:731–734
Peleg Z, Blumwald E (2011) Hormone balance and abiotic stress tolerance in crop plants. Curr Opin Plant Biol 14:290–295
Pellegrini E, Carucci MG, Campanella A, Lorenzini G, Nali C (2011) Ozone stress in Melissa officinalis plants assessed by photosynthetic function. Environ Exp Bot 73:94–101
Pellegrini E, Cioni PL, Francini A, Lorenzini G, Nali C, Flamini G (2012) Volatile emission patterns in poplar clones varying in response to ozone. J Chem Ecol 38:924–932
Puckette M, Weng H, Mahalingam R (2007) Physiological and biochemical responses of acute ozone-induced oxidative stress in Medicago truncatula. Plant Physiol Biochem 45:70–79
Quan LJ, Zhang B, Shi WW, Li HY (2008) Hydrogen peroxide in plants: a versatile molecule of the reactive oxygen species network. J Integr Plant Biol 50:2–18
Ranieri A, D’Urso G, Nali C, Lorenzini G, Soldatini GF (1996) Ozone stimulates apoplastic antioxidant systems in pumpkin leaves. Physiol Plant 97:381–387
Ranieri A, Serini R, Castagna A, Nali C, Baldan B, Lorenzini G, Soldatini GF (2000) Differential sensitivity to ozone in two poplar clones. Analysis of thylakoid pigment-protein complexes. Physiol Plant 110:181–188
Rao MV, Koch JE, Davis KR (2000) Ozone: a tool for probing programmed cell death in plants. Plant Mol Biol 44:345–358
Rao MV, Lee HI, Davis KR (2002) Ozone-induced ethylene production is dependent on salicylic acid, and both salicylic acid and ethylene act in concert to regulate ozone-induced cell death. Plant J 32:447–456
Romero-Puertas MC, Palma JM, Gomez M, Del Rio LA, Sandalio LM (2002) Cadmium causes the oxidative modification of proteins in pea plants. Plant Cell Environ 25:677–686
Roychoudhury A, Paul S, Basu S (2013) Cross-talk between abscisic acid-dependent and abscisic acid-independent pathways during abiotic stress. Plant Cell Rep 32:985–1006
Santino A, Taurino M, De Domenico S, Bonsegna S, Poltronieri P, Pastor V, Flors V (2013) Jasmonate signalling in plant development and defense response to multiple (a)biotic stress. Plant Cell Rep 32:1085–1098
Sgherri CLM, Navari-Izzo F (1995) Sunflower seedlings subjected to increasing water deficit stress: oxidative stress and defence mechanisms. Physiol Plant 93:25–30
Shin R, Berg RH, Schachtman DP (2005) Reactive oxygen species and root hairs in Arabidopsis root response to nitrogen, phosphorus and potassium deficiency. Plant Cell Physiol 46:1350–1357
Smirnoff N (1996) The function and metabolism of ascorbic acid in plants. Ann Bot 78:661–669
Thaler JS, Humphrey PT, Whiteman NK (2012) Evolution of jasmonate and salicylate signal crosstalk. Trends Plant Sci 17:260–270
Tománková K, Luhová L, Petřivalský M, Peč P, Lebeda A (2006) Biochemical aspects of reactive oxygen species formation in the interaction between Lycopersicon spp. and Oidium neolycopersici. Physiol Mol Plant Pathol 68:22–32
Ton J, Flors V, Mauch-Mani B (2009) The multifaceted role of ABA in disease resistance. Trends Plant Sci 14:310–317
Trivellini A, Ferrante A, Vernieri P, Serra G (2011) Effects of abscisic acid on ethylene biosynthesis and perception in Hibiscus rosa-sinensis L. flower development. J Exp Bot 62:5437–5452
Tuomainen J, Pellinen R, Roy S, Kiiskinen M, Eloranta T, Karjalainen R, Kangasjärvi J (1996) Ozone affects birch (Betula pendula Roth) phenylpropanoid, polyamine and active oxygen detoxifying pathways at biochemical and gene expression level. J Plant Physiol 148:179–188
Tuomainen H, Overmyer K, Keinänen M, Kollist H, Kangasjärvi J (2004) Mutual antagonism of ethylene and jasmonic acid regulates ozone-induced spreading cell death in Arabidopsis. Plant J 39:59–69
Vahala J, Keinänen M, Schützendübel A, Polle A, Kangasjärvi J (2003) Differential effects of elevated ozone on two hybrid aspen genotypes predisposed to chronic ozone fumigation. Role of ethylene and salicylic acid. Plant Physiol 132:196–205
Vijayan P, Shockey J, Levesque CA, Cook RJ, Browse J (1998) A role for jasmonate in pathogen defense of Arabidopsis. Proc Natl Acad Sci USA 95:7209–7214
Wagg S, Mills G, Hayes F, Wikinson S, Copper D, Davies WJ (2012) Reduced soil water availability did not protect two competing-grassland species from the negative effects of increasing background ozone. Environ Pollut 165:91–99
Walker-Simmons M (1987) ABA levels and sensitivity in developing wheat embryos of sprouting resistant and susceptible cultivars. Plant Physiol 84:61–66
Wang S, Jiao HJ, Faust M (1991) Changes in ascorbate, glutathione and related enzyme activities during thidiazuron-induced bud break apple. Physiol Plant 82:231–236
Wang F, Cui X, Sun Y, Dong CH (2013) Ethylene signaling and regulation in plant growth and stress responses. Plant Cell Rep 32:1099–1109
Wilkinson S, Davies WJ (2009) Drought, ozone, ABA and ethylene: new insights from cell to plant to community. Plant Cell Environ 33:510–525
Xiong L, Yang Y (2003) Disease resistance and abiotic stress tolerance in rice are inversely modulated by an abscisic acid inducible mitogen-activated protein kinase. Plant Cell 15:745–759
Yamaguchi-Shinozaki K, Shinozaki K (2006) Transcriptional regulatory networks in cellular responses and tolerance to dehydration and cold stresses. Annu Rev Plant Biol 7:781–803
Zawoznik MS, Groppa MD, Tomaro ML, Benavides MP (2007) Endogenous salicylic acid potentiates cadmium-induced oxidative stress in Arabidopsis thaliana. Plant Sci 173:190–197
Zhang J, Kirkham MB (1994) Drought-stress-induced changes in activities of superoxide dismutase, catalase, and peroxidase in wheat species. Plant Cell Physiol 35:783–791
Zhang X, Zhang L, Dong F, Gao J, Galbraith DW, Song CP (2001) Hydrogen peroxide is involved in abscisic acid-induced stomatal closure in Vicia faba. Plant Physiol 126:1438–1448
Acknowledgments
We gratefully acknowledge Dr. Giulia Fabiani and Dr. María José Díaz for their support during the biochemical analyses.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by A. Feher.
Rights and permissions
About this article
Cite this article
Pellegrini, E., Trivellini, A., Campanella, A. et al. Signaling molecules and cell death in Melissa officinalis plants exposed to ozone. Plant Cell Rep 32, 1965–1980 (2013). https://doi.org/10.1007/s00299-013-1508-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00299-013-1508-0