Abstract
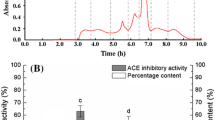
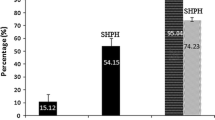
R-phycoerythrin prepared from red algae (Bangia fusco-purpurea) was hydrolyzed by pepsin followed by trypsin to produce angiotensin I-converting enzyme (ACE) inhibitory peptides. The IC50 of the hydrolysate of R-phycoerythrin (HRPE) was 191.1 ± 4.1 μg/mL, and the molecular weight of most products (89.9 %) was below 2 kDa. After sequential gel permeation and reversed-phase chromatography steps to purify the hydrolysate, two peptides with the sequences of ALLAGDPSVLEDR and VVGGTGPVDEWGIAGAR were obtained and their IC50 values were 57.2 ± 5.0 and 66.2 ± 4.2 μg/mL, respectively. The ALLAGDPSVLEDR and VVGGTGPVDEWGIAGAR peptides were derived from the β- and α-subunit of R-phycoerythrin from Polysiphonia urceolata, with a 92.3 % (12/13) and 94.1 % (16/17) match, respectively. Both peptides were resistant to digestion by proteinases common in the gastrointestinal tract. Therefore, the identified novel peptides derived from R-phycoerythrin may be used as potential nutraceuticals for development of functional foods.





Similar content being viewed by others
References
Eriksson U, Danilczyk U, Penninger JM (2002) Just the beginning: novel functions for angiotensin-converting enzymes. Curr Biol 12:R745–R752
Riordan JF (2003) Angiotensin I-converting enzyme and its relatives. Genome Biol 4:225
FitzGerald RJ, Murray BA, Walsh GJ (2004) Hypotensive peptides from milk proteins. J Nutr 134:980–988
Cian RE, Garzón AG, Ancona DB, Guerrero LC, Drago SR (2015) Hydrolyzates from Pyropia columbina seaweed have antiplatelet aggregation, antioxidant and ACE inhibitory peptides which maintain bioactivity after simulated gastrointestinal digestion. LWT-Food Sci Technol 64:881–888
White BL, Sanders TH, Davis JP (2014) Potential ACE-inhibitory activity and nanoLC-MS/MS sequencing of peptides derived from aflatoxin contaminated peanut meal. LWT-Food Sci Technol 56:537–542
Lee SJ, Kim YS, Kim SE, Kim EK, Hwang JW, Park TK et al (2012) Purification and characterization of a novel angiotensin I-converting enzyme inhibitory peptide derived from an enzymatic hydrolysate of duck skin byproducts. J Agric Food Chem 60:10035–10040
Wu Q, Cai QF, Tao ZP, Sun LC, Shen JD, Zhang LJ et al (2015) Purification and characterization of a novel angiotensin I-converting enzyme inhibitory peptide derived from abalone (Haliotis discus hannai Ino) gonads. Eur Food Res Technol 240:137–145
Ahn CB, Jeon YJ, Kim YT, Je JY (2012) Angiotensin I converting enzyme (ACE) inhibitory peptides from salmon byproduct protein hydrolysate by Alcalase hydrolysis. Process Biochem 47:2240–2245
Sheih IC, Fang TJ, Wu TK (2009) Isolation and characterisation of a novel angiotensin I-converting enzyme (ACE) inhibitory peptide from the algae protein waste. Food Chem 115:279–284
Vermeirssen V, Camp JV, Verstraete W (2004) Bioavailability of angiotensin I-converting enzyme inhibitory peptides. Br J Nutr 92:357–366
Hultin HO (1994) Oxidation of lipids in seafood. In: Shahidi F, Botta JR (eds) Seafoods: chemistry, processing technology and quality. Blackie Academic & Professional, Glasgow, pp 49–74
You LJ, Zhao MM, Regenstein JM, Ren JY (2010) Changes in the antioxidant activity of loach (Misgurnus anguillicaudatus) protein hydrolysates during a simulated gastrointestinal digestion. Food Chem 120:810–816
Pérez-Vega JA, Olivera-Castillo L, Gómez-Ruiz J, Hernández-Ledesma B (2013) Release of multifunctional peptides by gastrointestinal digestion of sea cucumber (Isostichopus badionotus). J Funct Foods 5:869–877
Hao W, Xu N, Xue S, Hong Y, Zhou C (2015) Hydrolysis and purification of ACE inhibitory peptides from the marine microalga Isochrysis galbana. J Appl Phycol 27:351–361
Furuta T, Miyabe Y, Yasui H, Kinoshita Y, Kishimura H (2016) Angiotensin I-converting enzyme (ACE) inhibitory peptides derived from phycobiliproteins of dulse Palmaria palmata. Mar Drugs 14:32. doi:10.3390/md14020032
Sato M, Hosokawa T, Yamaguchi T, Nakano T, Muramoto K, Kahara T et al (2002) Angiotensin I-converting enzyme inhibitory peptides derived from wakame (Undaria pinnatifida) and their antihypertensive effect in spontaneously hypertensive rats. J Agric Food Chem 50:6245–6252
Senthilkumar N, Kurinjimalar C, Thangam R, Suresh V, Kavitha G, Gunasekaran P et al (2013) Further studies and biological activities of macromolecular protein R-phycoerythrin from Portieria hornemannii. Int J Biol Macromol 62:107–116
Cano-Europa E, Ortiz-Butrón R, Gallardo-Casas CA, Blas-Valdivia V, Pineda-Reynoso M, Olvera-Ramírez R et al (2010) Phycobiliproteins from Pseudanabaena tenuis rich in C-phycoerythrin protect against HgCl2-caused oxidative stress and cellular damage in the kidney. J Appl Phycol 22:495–501
Wu Q, Fu XP, Sun LC, Zhang Q, Liu GM, Cao MJ et al (2015) Effects of physicochemical factors and in vitro gastrointestinal digestion on antioxidant activity of R-phycoerythrin from red algae (Bangia fusco-purpurea). Int J Food Sci Technol 50:1445–1551
Shahidi F, Naczk M, Hall D, Synowiecki J (1992) Insensitivity of the amino acids of canola and rapeseed to methanol-ammonia extraction and commercial processing. Food Chem 44:283–285
Lee YB, Elliott JG, Rickansrud DA, Hagberg EYC (1978) Predicting protein efficiency ratio by the chemical determination of connective tissue content in meat. J Food Sci 43:1359–1362
Cushman D, Cheung H (1971) Spectrophotometric assay and properties of the angiotensin-converting enzyme of rabbit lung. Biochem Pharmacol 20:1637–1648
US Pharmacopoeia (1995) The National Formulary. Simulated gastric fluid and simulated intestinal fluid, TS. In: The United States Pharmacopeia 23, The National Formulary 18, The United States Pharmacopeial Convention, Inc., Rockville, MD, 2053
Schägger H (2006) Tricine-SDS-PAGE. Nat Protoc 1:16–22
Galland-Irmouli AV, Pons L, Lucon M, Villaume C, Mrabet NT, Gueant JL et al (2000) One-step purification of R-phycoerythrin from the red macroalga Palmaria palmata using preparative polyacrylamide gel electrophoresis. J Chromatogr B 739:117–123
Chiou TK, Lai MM (2002) Comparison of taste components in cooked meats of small abalone fed different diets. Fish Sci 68:388–394
Ahn CB, Kim JG, Je JY (2014) Purification and antioxidant properties of octapeptide from salmon byproduct protein hydrolysate by gastrointestinal digestion. Food Chem 147:78–83
Sun LP, Zhang YF, Zhuang YL (2013) Antiphotoaging effect and purification of an antioxidant peptide from tilapia (Oreochromis niloticus) gelatin peptides. J Funct Foods 5:154–162
Boschin G, Scigliuolo GM, Resta D, Arnoldi A (2014) ACE-inhibitory activity of enzymatic protein hydrolysates from lupin and other legumes. Food Chem 145:34–40
Nikoo M, Benjakul S (2015) Potential application of seafood-derived peptides as biofunctional ingredients, antioxidant-cryoprotectant: a review. J Funct Foods 19:753–764
Intarasirisawat R, Benjakul S, Wu J, Visessanguan W (2013) Isolation of antioxidative and ACE inhibitory peptides from protein hydrolysate of skipjack (Katsuwana pelamis) roe. J Funct Foods 5:1854–1862
Asooden A, Yazdi MM, Chamani J (2012) Purification and characterisation of angiotensin I converting enzyme inhibitory peptides from lysozyme hydrolysates. Food Chem 131:291–295
Cheung HS, Wang FL, Ondetti MA, Sabo EF, Cushman DW (1980) Binding of peptide substrates and inhibitors of angiotensin-converting enzyme. Importance of the COOH-terminal dipeptide sequence. J Biol Chem 255:401–407
Matthews BW (2009) Racemic crystallography-easy crystals and easy structures: what’s not to like? Protein Sci 18:1135–1138
Ahn J, Cao MJ, Yu YQ, Engen JR (2013) Accessing the reproducibility and specificity of pepsin and other aspartic proteases. BBA-Proteins Proteom 1834:1222–1229
Guo C, Cao MJ, Liu GM, Lin XS, Hara K, Su WJ (2007) Purification, characterization, and cDNA cloning of a myofibril-bound serine proteinase from the skeletal muscle of crucian carp (Carassius auratus). J Agr Food Chem 55:1510–1516
Acknowledgments
This study was sponsored by the National Natural Scientific Foundation of China (Nos. 31271838, 31471640), the Key Project of the Ministry of Science and Technology of China (2012BAD38B09), the Public Science and Technology Research Fund Project of Ocean (201305015-3), and the Science and Technology Bureau of Xiamen (3502Z20133020). We thank Professor Qi-Xin Zhong in the University of Tennessee for critical reviewing of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Compliance with ethics requirements
This article does not contain any studies with human or animal subjects.
Rights and permissions
About this article
Cite this article
Wu, Q., Cai, QF., Yoshida, A. et al. Purification and characterization of two novel angiotensin I-converting enzyme inhibitory peptides derived from R-phycoerythrin of red algae (Bangia fusco-purpurea). Eur Food Res Technol 243, 779–789 (2017). https://doi.org/10.1007/s00217-016-2792-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00217-016-2792-z