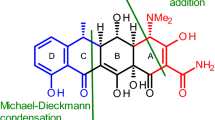
Abstract
Since the start of the 21st century, antibiotic drug discovery and development from natural products has experienced a certain renaissance. Currently, basic scientific research in chemistry and biology of natural products has finally borne fruit for natural product-derived antibiotics drug discovery. A batch of new antibiotic scaffolds were approved for commercial use, including oxazolidinones (linezolid, 2000), lipopeptides (daptomycin, 2003), and mutilins (retapamulin, 2007). Here, we reviewed the thiazolyl peptides (thiopeptides), an ever-expanding family of antibiotics produced by Gram-positive bacteria that have attracted the interest of many research groups thanks to their novel chemical structures and outstanding biological profiles. All members of this family of natural products share their central azole substituted nitrogen-containing six-membered ring and are classified into different series. Most of the thiopeptides show nanomolar potencies for a variety of Gram-positive bacterial strains, including methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant enterococci (VRE), and penicillin-resistant Streptococcus pneumonia (PRSP). They also show other interesting properties such as antiplasmodial and anticancer activities. The chemistry and biology of thiopeptides has gathered the attention of many research groups, who have carried out many efforts towards the study of their structure, biological function, and biosynthetic origin. Here we reviewed a total of 119 natural thiopeptides discovered during the last 50 years. Moreover, we summarized biological profiles, mechanisms of action, and biosynthesis of these thiopeptides.






























Similar content being viewed by others
References
Abe H, Kushida K, Shiobara Y, Kodama M (1988) The structures of sulfomycin I and berninamycin A. Tetrahedron Lett 29:1401–1404
Acker MG, Bowers AA, Walsh CT (2009) Generation of thiocillin variants by prepeptide gene replacement and in vivo processing by Bacillus cereus. J Am Chem Soc 131:17563–17565
Aminake MN, Schoof S, Sologub L, Leubner M, Kirschner M, Arndt HD, Pradel G (2011) Thiostrepton and derivatives exhibit antimalarial and gametocytocidal activity by dually targeting parasite proteasome and apicoplast. Antimicrob Agents Chemother 55:1338–1348
Anborgh PH, Parmeggiani A (1991) New antibiotic that acts specifically on the GTP-bound form of elongation factor Tu. EMBO J 10:779–784
Anderson B, Hodgkin DC, Viswamitra MA (1970) The structure of thiostrepton. Nature 225:233–235
Aoki M, Ohtsuka T, Itezono Y, Yokose K, Furihata K, Seto H (1991a) Structure of cyclothiazomycin, a unique polythiazole-containing peptide with renin inhibitory activity. Part 1. chemistry and partial structures of cyclothiazomycin. Tetrahedron Lett 32:217–220
Aoki M, Ohtsuka T, Itezono Y, Yokose K, Furihata K, Seto H (1991b) Structure of cyclothiazomycin, a unique polythiazole-containing peptide with renin inhibitory activity. Part 2. Total structure. Tetrahedron Lett 32:221–224
Aoki M, Ohtsuka T, Yamada M, Ohba Y, Yoshizaki H, Yasuno H, Sano T, Watanabe J, Yokose K, Seto H (1991c) Cyclothiazomycin, a novel polythiazole-containing peptide with renin inhibitory activity. Taxonomy, fermentation, isolation and physico-chemical characterization. J Antibiot 44:582–588
Arnison PG, Bibb MJ, Bierbaum G, Bowers AA, Bugni TS, Bulaj G, Camarero JA, Campopiano DJ, Challis GL, Clardy J (2013) Ribosomally synthesized and post-translationally modified peptide natural products: overview and recommendations for a universal nomenclature. Nat Prod Rep 30:108–160
Ashraf-Khorassani M, Taylor LT, Marr JGD (2000) Analysis of the sulfomycin component of alexomycin in animal feed by enhanced solvent extraction and supercritical fluid chromatography. J Biochem Biophys Methods 43:147–156
Aulakh VS, Ciufolini MA (2011) Total synthesis and complete structural assignment of thiocillin I. J Am Chem Soc 133:5900–5904
Bagley MC, Dale JW, Merritt EA, Xiong X (2005) Thiopeptide antibiotics. Chem Rev 105:685–714
Barot KP, Nikolova S, Ivanov I, Ghate MD (2013) Novel anticancer agents and targets: recent advances and future perspectives. Mini Rev Med Chem 13:1239–1255
Bechthold A, Floss HG (1994) Overexpression of the thiostrepton-resistance gene from Streptomyces azureus in Escherichia coli and characterization of recognition sites of the 23S rRNA a1067 2’-methyltransferase in the guanosine triphosphatase center of 23S ribosomal RNA. Eur J Biochem 224:431–437
Benazet F, Cartier JR (1980) Effect of nosiheptide as a feed additive in chicks on the quantity, duration, prevalence of excretion, and resistance to antibacterial agents of Salmonella typhimurium; on the proportion of Escherichia coli and other coliforms resistant to antibacterial agents. Poultry Sci 59:1405–1415
Benazet F, Cartier M, Florent J, Godard C, Jung G, Lunel J, Mancy D, Pascal C, Renaut J, Tarridec P, Theilleux J, Tissier R, Dubost M, Ninet L (1980) Nosiheptide, a sulfur-containing peptide antibiotic isolated from Streptomyces actuosus 40037. Experientia 36:414–416
Bennallack PR, Griffitts JS (2017) Elucidating and engineering thiopeptide biosynthesis. World J Microbiol Biotechnol 33:119
Bennett JE, Piggott WR, Utz JP, Emmons CW (1969) Assay of antifungal agent saramycetin (X-5079C) in patient body fluids. Am Rev Respir Dis 99:964–965
Berger J, Sternbach LH, Muller M, Lasala ER, Grunberg E, Goldberg MW (1962) X-5079C, a new polypeptide antifungal antibiotic active against systemic mycoses. Antimicrob Agents Chemother 1961:436–446
Bewley KD, Bennallack PR, Burlingame MA, Robison RA, Griffitts JS, Miller SM (2016) Capture of micrococcin biosynthetic intermediates reveals C-terminal processing as an obligatory step for in vivo maturation. Proc Natl Acad Sci USA 113:12450–12455
Bhat UG, Halasi M, Gartel AL (2009) Thiazole antibiotics target FoxM1 and induce apoptosis in human cancer cells. PLoS ONE 4:e5592
Bhat UG, Zipfel PA, Tyler DS, Gartel AL (2008) Novel anticancer compounds induce apoptosis in melanoma cells. Cell Cycle 7:1851–1855
Biskupiak JE, Meyers E, Gillum AM, Dean L, Trejo WH, Kirsch DR (1988) Neoberninamycin, a new antibiotic produced by Micrococcus luteus. J Antibiot 41:684–687
Bleich R, Watrous JD, Dorrestein PC, Bowersa AA, Shank EA (2015) Thiopeptide antibiotics stimulate biofilm formation in Bacillus subtilis Proc Natl Acad Sci USA 112(10):3086–3091
Bodanszky M (1963) The establishment of the identity of thiostrepton with thiactin(bryamycin). J Antibiot A 16:76–79
Boeck LD, Berry DM, Mertz FP, Wetzel RW (1992) A10255, a complex of novel growth-promoting thiopeptide antibiotics produced by a strain of Streptomyces gardneri. Taxonomy and fermentation studies. J Antibiot 45:1222–1230
Bond CS, Shaw MP, Alphey MS, Hunter WN (2001) Structure of the macrocycle thiostrepton solved using the anomalous dispersion contribution of sulfur. Acta Crystallogr D Biol Crystallogr 57:755–758
Bowers AA, Acker MG, Koglin A, Walsh CT (2010) Manipulation of thiocillin variants by prepeptide gene replacement: structure, conformation, and activity of heterocycle substitution mutants. J Am Chem Soc 132:7519–7527
Bowling BD, Doudican N, Manga P, Orlow SJ (2008) Inhibition of mitochondrial protein translation sensitizes melanoma cells to arsenic trioxide cytotoxicity via a reactive oxygen species dependent mechanism. Cancer Chemother Pharmacol 63:37–43
Bycroft BW, Gowland MS (1978) The structures of the highly modified peptide antibiotics micrococcin P1 and P2. J Chem Soc Chem Commun 6:256–258
Ca Edo Hernández LM, Romero Millán F, Fernández Medarde A, Fernández Chimeno RI, Hidalgo Villar JC, inventors; WO, assignee (2012) Peptides as bioactive compounds. Patent 2012/062906. 2011-11-11
Cameron DM, Thompson J, March PE, Dahlberg AE (2002) Initiation factor IF2, thiostrepton and micrococcin prevent the binding of elongation factor G to the Escherichia coli ribosome. J Mol Biol 319:27–35
Carnio MC, Höltzel A, Rudolf M, Henle T, Jung G, Scherer S (2000) The macrocyclic peptide antibiotic micrococcin P(1) is secreted by the food-borne bacterium Staphylococcus equorum WS 2733 and inhibits Listeria monocytogenes on soft cheese. Appl Environ Microbiol 66:2378–2384
Castro RJ, González HG, Santamaría Sánchez RI, Ca Edo LM (2002) Radamycin, a novel thiopeptide produced by Streptomyces sp. RSP9. II. Physico-chemical properties and structure determination. J Antibiot 55:391–395
Cha JY, Han S, Hong HJ, Cho H, Kim D, Kwon Y, Kwon SK, Crüsemann M, Bok LY, Kim JF, Giaever G, Nislow C, Moore BS, Thomashow LS, Weller DM, Kwak Y (2015) Microbial and biochemical basis of a Fusarium wilt-suppressive soil. ISME J 10:119–129
Chiu ML, Folcher M, Griffin P, Holt T, Klatt T, Thompson CJ (1996) Characterization of the covalent binding of thiostrepton to a thiostrepton-induced protein from Streptomyces lividans. Biochemistry 35:2332–2341
Chiu ML, Viollier PH, Katoh T, Jeremy J, Ramsden A, Thompson CJ (2001) Ligand-induced changes in the Streptomyces lividans TipAL protein imply an alternative mechanism of transcriptional activation for MERR-like proteins. Biochemistry 40:12950–12958
Clough B, Rangachari K, Strath M, Preiser PR, Wilson RJMI (1999) Antibiotic inhibitors of organellar protein synthesis in Plasmodium falciparum. Protist 150:189–195
Clough B, Strath M, Preiser P, Denny P, Wilson IR (1997) Thiostrepton binds to malarial plastid rRNA. FEBS Lett 406:123–125
Colombo L, Tavecchia P, Selva E, Gallo GG, Zerilli LF (1992) Contribution of mass spectrometric techniques to the structure elucidation of antibiotic GE2270A, a novel inhibitor of bacterial protein synthesis. J Mass Spectrom 27:219–225
Connolly TP, Regueiroren A, Leet JE, Springer DM, Goodrich J, Huang XS, Pucci MJ, Clark JM, Bronson JJ, Ueda Y (2005) Chemical conversion of nocathiacin I to nocathiacin II and a lactone analogue of glycothiohexide alpha. J Nat Prod 68:550–553
Cooper R, Truumees I, Barrett T, Patel M, Schwartz J, Puar M, Das P, Pramanik B (1990) Saramycetin, a thiazolyl peptide from a Streptomyces sp.: chemical characterization and molecular weight determination. J Antibiot 43:897–900
Cox CL, Tietz JI, Sokolowski K, Melby JO, Doroghazi JR, Mitchell DA (2014) Nucleophilic 1,4-additions for natural product discovery. ACS Chem Biol 9:2014–2022
Cromwell GL, Davis GW, Morrow WE, Primo RA, Rozeboom DW, Sims MD, Stanisiewski EP, Ho CH (1996) Efficacy of the antimicrobial compound U-82,127 as a growth promoter for growing-finishing pigs. J Anim Sci 74:1284–1287
Cromwell GL, Stahly TS, Jensen AH, Plumlee MP, Krider JL, Russett JC (1984a) Efficacy of thiopeptin as a growth promotant for growing barrows and gilts—a cooperative study. J Anim Sci 59:892–895
Cromwell GL, Stahly TS, Speer VC, O’Kelly R (1984b) Efficacy of nosiheptide as a growth promotant for growing-finishing swine—a cooperative study. J Anim Sci 59:1125–1128
Cron MJ, Whitehead DG, Hooper IR, Heinemann B, Lein J (1956) Bryamycin, a new antibiotic. Antibiot Chemother 6:63–67
Cundliffe E (1978) Mechanism of resistance to thiostrepton in the producing-organism Streptomyces azureus. Nature 272:792–795
Cundliffe E, Dixon P, Stark M, St Ffler G, Ehrlich R, St Fflermeilicke M, Cannon M (1979) Ribosomes in thiostrepton-resistant mutants of Bacillus megaterium lacking a single 50S subunit protein. J Mol Biol 132:235–252
Cundliffe E, Dixon PD (1975) Inhibition of ribosomal A site functions by sporangiomycin and micrococcin. Antimicrob Agents Chemother 8:1–4
Cundliffe E, Thompson J (1979) Ribose methylation and resistance to thiostrepton. Nature 278:859–861
Cundliffe E, Thompson J (1981a) The mode of action of nosiheptide (multhiomycin) and the mechanism of resistance in the producing organism. J Gen Microbiol 126:185–192
Cundliffe E, Thompson J (1981b) Concerning the mode of action of micrococcin upon bacterial protein synthesis. Eur J Biochem 118:47–52
Debono M, Molloy RM, Occolowitz JL, Paschal JW, Hunt AH, Michel KH, Martin JW (1992) The structures of A10255 B, -G and -J: new thiopeptide antibiotics produced by Streptomyces gardneri. J Org Chem 57:5200–5208
Donia MS, Cimermancic P, Schulze CJ, Brown LCW, Martin J, Mitreva M, Clardy J, Linington RG, Fischbach MA (2014) A systematic analysis of biosynthetic gene clusters in the human microbiome reveals a common family of antibiotics. Cell 158:1402–1414
Dunbar KL, Chekan JR, Cox CL, Burkhart BJ, Nair SK, Mitchell DA (2014) Discovery of a new ATP-binding motif involved in peptidic azoline biosynthesis. Nat Chem Biol 10:823–829
Dutcher JD, Vandeputte J (1954) Thiostrepton, a new antibiotic. II. Isolation and chemical characterization. Antibiot Ann 3:560–561
Egawa Y, Umino K, Tamura Y, Shimizu M, Kaneko K, Sakurazawa M, Awataguchi S, Okuda T (1969) Sulfomycins, a series of new sulfur-containing antibiotics. I. Isolation, purification and properties. J Antibiot 22:12–17
Endo T, Yonehara H (1978) Identity of multhiomycin with nosiheptide. J Antibiot 31:623–625
Engelhardt K, Degnes KF, Kemmler M, Bredholt H, Fj Rvik E, Klinkenberg G, Sletta HV, Ellingsen TE, Zotchev SB (2010a) Production of a new thiopeptide antibiotic, TP-1161, by a marine Nocardiopsis species. Appl Environ Microbiol 76:4969–4976
Engelhardt K, Degnes KF, Zotchev SB (2010b) Isolation and characterization of the gene cluster for biosynthesis of the thiopeptide antibiotic TP-1161. Appl Environ Microbiol 76:7093–7101
Etlinger JD, Goldberg AL (1977) A soluble ATP-dependent proteolytic system responsible for the degradation of abnormal proteins in reticulocytes. Proc Natl Acad Sci USA 74:54–58
Fabbretti A, He C, Gaspari E, Maffioli S, Brandi L, Spurio R, Sosio M, Jabes D, Donadio S (2015) A derivative of the thiopeptide GE2270A highly selective against Propionibacterium acnes. Antimicrob Agents Chemother 59:4560–4568
Favret ME, Paschal JW, Elzey TK, Boeck LD (1992) Biosynthesis of thiopeptide antibiotic A10255: incorporation of isotopically-labeled precursors. J Antibiot 45:1499–1511
Feng M, Cai Q, Shi X, Huang H, Zhou P, Guo X (2008) Recombinant high-density lipoprotein complex as a targeting system of nosiheptide to liver cells. J Drug Target 16:502–508
Ferrari P, Colombo L, Stella S, Selva E, Zerilli LF (1995) Antibiotic GE37468 A—a novel inhibitor of bacterial protein synthesis. II. Structure elucidation. J Antibiot 48:1304–1311
Ganguly AK, Girijavallabhan VM, Sarre O, inventors; WO, assignee (1987) Novel derivatives of the oligosaccharide antibiotic complex 13-384, their preparation and pharmaceutical compositions containing them. Patent 0241541. 1986-10-15
Gartel AL (2013) Thiazole antibiotics siomycin A and thiostrepton inhibit the transcriptional activity of FOXM1. Front Oncol 3:150
Gill DR, Owens FN, Fent RW, Fulton RK (1979) Thiopeptin and roughage level for feedlot steers. J Anim Sci 49:1145–1150
Goldstein BP, Berti M, Ripamonti F, Resconi A, Scotti R, Denaro M (1993) In vitro antimicrobial activity of a new antibiotic, MDL 62,879 (GE2270 A). Antimicrob Agents Chemother 37:741–745
González HG, Castro RJ, Ca Edo Hernández LM, Díaz M, Fernández-Abalos JM, Trujillano I, Santamaría RI (2002) Radamycin, a novel thiopeptide produced by Streptomyces sp. RSP9. I. Taxonomy, fermentation, isolation and biological activities. J Antibiot 55:383–390
Grunberg E, Berger J, Titsworth E (1961) Chemotherapeutic studies on a new antifungal agent, X-5079C, effective against Systemic mycoses. Am Rev Respir Dis 84:504–506
Guo X, Liu A, Hua H, Lu H, Zhang D, Lin Y, Sun Q, Zhu X, Yan G, Zhao F (2015) Siomycin a induces apoptosis in human lung adenocarcinoma A549 cells by suppressing the expression of FOXM1. Nat Prod Commun 10:1603–1606
Harms JM, Wilson DN, Schluenzen F, Connell SR, Stachelhaus T, Zaborowska Z, Spahn CM, Fucini P (2008) Translational regulation via L11: molecular switches on the ribosome turned on and off by thiostrepton and micrococcin. Mol Cell 30:26–38
Hashimoto M, Murakami T, Funahashi K, Tokunaga T, Nihei KI, Okuno T, Kimura T, Naoki H, Himeno H (2006) An RNA polymerase inhibitor, cyclothiazomycin B1, and its isomer. Bioorgan Med Chem 14:8259–8270
Hayashi S, Ozaki T, Asamizu S, Ikeda H, Omura S, Oku N, Igarashi Y, Tomoda H, Onaka H (2014) Genome mining reveals a minimum gene set for the biosynthesis of 32-membered macrocyclic thiopeptides lactazoles. Chem Biol 21:679–688
Heffron SE, Jurnak F (2000) Structure of an EF-Tu complex with a thiazolyl peptide antibiotic determined at 2.35 A resolution: atomic basis for GE2270A inhibition of EF-Tu. Biochemistry 39:37–45
Hegde NS, Sanders DA, Rodriguez RL, Balasubramanian S (2011) The transcription factor FOXM1 is a cellular target of the natural product thiostrepton. Nat Chem 3:725–731
Hensens OD, Albers-Sch Nberg G (1978) Total structure of the peptide antibiotic components of thiopeptin by 1H and 13C NMR spectroscopy. Tetrahedron Lett 19:3649–3652
Hensens OD, Alberssch Nberg G (1983) 13C NMR study of thiostrepton and thiopeptin components. J Antibiot 36:832–845
Hensens OD, Albers-Sch Nberg G, Anderson BF (1983) The solution conformation of the peptide antibiotic thiostrepton: a 1H NMR study. J Antibiot 36:799–813
Higashibayashi S, Kohno M, Goto T, Suzuki K, Mori T, Hashimoto K, Nakata M (2004) Synthetic studies on thiostrepton family of peptide antibiotics: synthesis of the pentapeptide segment containing dihydroxyisoleucine, thiazoline and dehydroamino acid. Tetrahedron Lett 35:3707–3712
Holmes DJ, Caso JL, Thompson CJ (1993) Autogenous transcriptional activation of a thiostrepton-induced gene in Streptomyces lividans. EMBO J 12:3183–3191
Hudson GA, Zhang Z, Jonathan I, Mitchell DA, Van Der Donk WA (2015) In vitro biosynthesis of the core scaffold of the thiopeptide thiomuracin. J Am Chem Soc 137:16012–16015
Imanaka H, Kohsaka M, Ikushima K, Miyairi N, Miyoshi T, inventors; US, assignee (1973) Antibiotic thiopeptin and process of production. Patent 3761587. 1971-02-23
Jayasuriya H, Herath K, Ondeyka JG, Zhang C, Zink DL, Brower M, Gailliot FP, Greene J, Birdsall G, Venugopal J, Ushio M, Burgess B, Russotti G, Walker A, Hesse M, Seeley A, Junker B, Connors N, Salazar O, Genilloud O, Liu A, Masurekar P, Barrett J, Singh SB (2007) Isolation and structure elucidation of thiazomycin—a potent thiazolyl peptide antibiotic from Amycolatopsis fastidiosa. J Antibiot 60:554–564
Jones RN, Chen YS, Marshall SA (1998) Comparative in vitro activity of alexomycin (U-82127) tested against Escherichia coli, Salmonella spp., and Enterococci of animal and human origin. Clin Microbiol Infect 4:601–603
Just-Baringo X, Albericio F, Lvarez M (2014a) Thiopeptide engineering: a multidisciplinary effort towards future drugs. Angew Chem 53:6602–6616
Just-Baringo X, Bruno P, Ottesen LK, Ca Edo LM, Albericio F, Álvarez M (2013) Total synthesis and stereochemical assignment of baringolin. Angew Chem Int Ed 52:7818–7821
Just-Baringo X, Bruno P, Pitart C, Vila J, Albericio F, álvarez M (2014b) Dissecting the structure of thiopeptides: assessment of thiazoline and tail moieties of baringolin and antibacterial activity optimization. J Med Chem 57:4185–4195
Kamigiri K, Watanabe M, Nagai K, Arao N, Suzumura K, Suzuki KI, Kurane R, Yamaoka M, Kawano Y, inventors; US, assignee (2006) Thiopeptide compounds. Patent 7030085. 2003-03-12
Keller-Juslen C, Kuhn M, King HD, inventors; US, assignee (1984) Antibiotics, pharmaceutical compositions and method of use. Patent 4478831. 1982-05-10
Kelly WL, Pan L, Li C (2009) Thiostrepton biosynthesis: prototype for a new family of bacteriocins. J Am Chem Soc 131:4327–4334
Kettenring J, Colombo L, Ferrari P, Tavecchia P, Nebuloni M, Vékey K, Gallo GG, Selva E (1991) Antibiotic GE2270A: a novel inhibitor of bacterial protein synthesis. II. Structure elucidation. J Antibiot 44:702–715
Kezar WW, Church DC (1979) Effect of thiopeptin and sodium bicarbonate on the prevention of lactic acidosis induced in sheep. J Anim Sci 49:1396–1402
Kim J, inventor WO, assignee (2002) Cancer therapeutic agent comprising thiopeptide with multiple thiazole rings
Kim SC, inventor WO, assignee (2002) Use of macrocyclic peptide compound as anticancer agents and method for diagnosing cancer. Patent 2001/102400. 2002-06-24
King A, Bethune L, Phillips I (1993) In vitro activity of MDL 62,879 (GE2270A) against aerobic Gram-positive and anaerobic bacteria. Antimicrob Agents Chemother 37:746–749
Kirschbaum J, Aszalos A (2010) Molecular weight of the antifungal antibiotic saramycetin. J Pharm Sci 56:410–411
Kodani S, Ninomiya A (2013) Isolation of new thiopeptide berninamycin E from Streptomyces atroolivaceus. Asian J Chem 25:490–492
Koerber-Plé K, Massiot G (1995) Total synthesis of nosiheptide. Synthesis of thiazole fragments. J Heterocyclic Chem 32:1309–1315
Kohno J, Kameda N, Nishio M, Kinumaki A, Komatsubara S (1996) The structures of sulfomycins II and III. J Antibiot 49:1063–1065
Kumar EKV, Kenia J, Mukhopadhyay T, Nadkarni SR (1999) Methylsulfomycin I, a new cyclic peptide antibiotic from a Streptomyces sp. HIL Y-9420704. J Nat Prod 62:1562–1564
Kwok JM, Myatt SS, Marson CM, Coombes RC, Constantinidou D, Lam EW (2008) Thiostrepton selectively targets breast cancer cells through inhibition of forkhead box M1 expression. Mol Cancer Ther 7:2022–2032
LaMarche MJ, Leeds JA, Amaral A, Brewer JT, Bushell SM, Deng G, Dewhurst JM, Ding J, Dzink-Fox JA, Gamber G et al. (2012) Discovery of LFF571: an investigational agent for Clostridium difficile infection. J Med Chem 55:2376–2387
LaMarche MJ, Leeds JA, Amaral K, Dzink-Fox JA, Gunderson K, Krastel P, Memmert K, Patane MA, Rann EM, Schmitt E, Tiamfook S, Wang B (2011) Antibacterial optimization of 4-aminothiazolyl analogues of the natural product GE2270 A: identification of the cycloalkylcarboxylic acids. J Med Chem 54:8099–8109
LaMarche MJ, Leeds JA, Dzink-Fox J, Gangl E, Krastel P, Neckermann G, Palestrant D, Patane MA, Rann EM, Tiamfook S, Yu D (2012) Antibiotic optimization and chemical structure stabilization of thiomuracin A. J Med Chem 55:6934–6941
LaMarche MJ, Leeds JA, Dzink-fox J, Gangl E, Krastel P, Neckermann G, Palestrant D, Patane MA, Rann EM, Tiamfook S, Yu D (2016) Antibiotic optimization and chemical structure stabilization of thiomuracin A. J Med Chem 55:6934–6941
Lau RCM, Rinehart KL (1994) Berninamycins B, C, and D, minor metabolites from Streptomyces bernensis. J Antibiot 47:1466–1472
Lee M, Yang J, Park S, Jo E, Kim HY, Bae YS, Windisch MP (2016) Micrococcin P1, a naturally occurring macrocyclic peptide inhibiting Hepatitis C virus entry in a pan-genotypic manner. Antivir Res 132:287–295
Leeds JA, Lamarche MJ, Brewer JT, Bushell SM, Deng G, Dewhurst JM, Dzink-Fox J, Gangl E, Jain A, Lee L (2011) In vitro and in vivo activities of novel, semisynthetic thiopeptide inhibitors of bacterial elongation factor Tu. Antimicrob. Agents Chemother 55:5277–5283
Leet JE, Ax HA, Gustavson DR, Brown DM, Turner L, Brown K, Li W, Lam KS, inventors; US, assignee (2001) Nocathiacin antibiotics. Patent 6218398. 1999-07-13
Leet JE, Li W, Ax HA, Matson JA, Huang S, Huang R, Cantone JL, Drexler D, Dalterio RA, Lam KS (2003) Nocathiacins, new thiazolyl peptide antibiotics from Nocardia sp. II. isolation, characterization, and structure determination. J Antibiot 34:232–242
Li C, Kelly WL (2010) Recent advances in thiopeptide antibiotic biosynthesis. Nat Prod Rep 27:153–164
Li W, Leet JE, Ax HA, Gustavson DR, Brown DM, Turner L, Brown K, Clark J, Yang H, Fung-Tomc J, Lam KS (2003) Nocathiacins, new thiazolyl peptide antibiotics from Nocardia sp. I. Taxonomy, fermentation and biological activities. J Antibiot 56:226–231
Liao R, Duan L, Lei C, Pan H, Ding Y, Zhang Q, Chen D, Shen B, Yu Y, Liu W (2009) Thiopeptide biosynthesis featuring ribosomally synthesized precursor peptides and conserved posttranslational modifications. Chem Biol 16:141–147
Liesch JM, McMillan JA, Pandey RC, Paul IC, Rinehart Jr KL, Reusser F (1976a) Letter: Berninamycin. I. The structure of berninamycinic acid. J Am Chem Soc 98:299–300
Liesch JM, Millington DS, Pandey RC, Rinehart Jr KL (1976b) Berninamycin. 2. Products of acidic hydrolysis, methanolysis, and acetolysis of berninamycin A1. J Am Chem Soc 98:8237–8249
Liesch JM, Rinehart Jr. KL (1977) Berninamycin. 3. Total structure of berninamycin A1,2. J Am Chem Soc 99:1645–1646
Lociuro S, Tavecchia P, Marzorati E, Landini P, Goldstein BP, Denarod M, Ciabatti R (1997) Antimicrobial activities of chemically modified thiazolyl peptide antibiotic MDL 62,879 (GE2270A). J. Antibiot 50:344–349
Luo X, Zambaldo C, Liu T, Zhang Y, Xuan W, Wang C, Wang C, Reed SA, Yang PY, Wang RE, Tsotne Javahishvili T, Schultz PG, Young TS (2016) Recombinant thiopeptides containing noncanonical amino acids. Proc Natl Acad Sci USA 113:3615–3620
Mahajan GB, George SD, Ranadive PV, Mishra PDS, Panshikar RM, Sawant SN, Krishna S, Sivakumar M, Pari K, inventors; US, assignee (2007) Novel antibacterial compounds. Patent 2007/119201. 2007-04-10
Mak JYW, Xu W, Fairlie DP (2015) Thiazoles in peptides and peptidomimetics. Peptidomimetics 48:235–266
Malcolmsona SJ, Young TS, Ruby JG, Skewes-Cox P, Walsh CT (2013) The posttranslational modification cascade to the thiopeptide berninamycin generates linear forms and altered macrocyclic scaffolds. Proc Natl Acad Sci USA 110:8483–8488
Mankin AS, Leviev I, Garrett RA (1994) Cross-hypersensitivity effects of mutations in 23S rRNA yield insight into aminoacyl-tRNA binding. J Mol Biol 244:151–157
Marinelli F, Gastaldo L, Toppo G, Quarta C (1996) Antibiotic GE37468A—a new inhibitor of bacterial protein synthesis. III. strain and fermentation study. J Antibiot 49:880–885
Markham NP, Wells AQ, Heatley NG, Florey HW (1951) The effect on experimental tuberculosis of the intravenous injection of micrococcin. Br J Exp Pathol 32:25–33
Marshall SA, Jones RN (1999) Spectrum and antimicrobial activity of alexomycin (PNU-82, 127), a peptide compound projected for use in animal health. Diagn Microbiol Infect Dis 33:181–186
Martín J, Sousa TDS, Crespo G, Palomo S, González I, Tormo JR, De La Cruz M, Anderson M, Hill RT, Vicente F, Genilloud O, Reyes F (2013) Kocurin, the true structure of PM181104, an anti-methicillin-resistant Staphylococcus aureus (MRSA) thiazolyl peptide from the marine-derived bacterium Kocuria palustris. Mar Drugs 11:387–398
Matsumoto M, Kawamura Y, Yasuda Y, Tanimoto T, Matsumoto K, Yoshida T, Shoji J (1989) Isolation and characterization of thioxamycin. J Antibiot 42:1465–1469
Mcconkey GA, Rogers MJ, Mccutchan TF (1997) Inhibition of Plasmodium falciparum protein synthesis. Targeting the plastid-like organelle with thiostrepton. J Biol Chem 272:2046–2049
Mine K, Miyairi N, Takano N, Mori S, Watanabe N (1972) Thiopeptin, a new feed-additive antibiotic: biological studies and field trials. Antimicrob Agents Chemother 1:496–503
Miyairi N, Miyoshi T, Aoki H, Kosaka M, Ikushima H, Kunugita K, Sakai H, Imanaka H (1970) Studies on thiopeptin antibiotics. I. Characteristics of thiopeptin B. J Antibiot 23:113–119
Mizuhara N, Kuroda M, Ogita A, Tanaka T, Usuki Y, Fujita K (2011) Antifungal thiopeptide cyclothiazomycin B1 exhibits growth inhibition accompanying morphological changes via binding to fungal cell wall chitin. Bioorg Med Chem 19:5300–5310
Mocek U, Beale JM, Floss HG (1989) Reexamination of the 1H and 13C NMR spectral assignments of thiostrepton. J Antibiot 42:1649–1652
Mocek U, Zeng Z, O’Hagan D, Zhou P, Fan LDG, Beale JM, Floss HG (1993) Biosynthesis of the modified peptide antibiotic thiostrepton in Streptomyces azureus and Streptomyces laurentii. J Am Chem Soc 115:4338–4341
Modolell J, Cabrer B, Parmeggiani A, Vazquez D(1971) Relationship between elongation factor I- and elongation factor II-dependent guanosine triphosphatase activities of ribosomes. Inhibition of both activities by ricin Proc Natl Acad Sci USA 68:1796–1800
Möhrle VG, Tieleman LN, Kraal B (1997) Elongation factor Tu1 of the antibiotic GE2270A producer Planobispora rosea has an unexpected resistance profile against EF-Tu targeted antibiotics. Biochem Biophys Res Commun 230:320–326
Morris RP, Leeds JA, Naegeli HU, Oberer L, Memmert K, Weber E, Lamarche MJ, Parker CN, Burrer N, Esterow S, Hein AE, Schmitt EK, Krastel P (2009) Ribosomally synthesized thiopeptide antibiotics targeting elongation factor Tu. J Am Chem Soc 131:5946–5955
Muir LA, Duquette PF, Rickes EL, Smith GE (1980a) Thiopeptin for the prevention of ovine lactic acidosis induced by diet change. J Anim Sci 51:1182–1188
Muir LA, Rickes EL, Duquette PF, Smith GE (1980b) Control of wheat-induced lactic acidosis in sheep by thiopeptin and related antibiotics. J Anim Sci 50:547–553
Muir LA, Rickes EL, Duquette PF, Smith GE (1981) Prevention of induced lactic acidosis in cattle by thiopeptin. J Anim Sci 52:635–643
Mukai A, Fukai T, Hoshino Y, Yazawa K, Harada K, Mikami Y (2009) Nocardithiocin, a novel thiopeptide antibiotic, produced by pathogenic Nocardia pseudobrasiliensis IFM 0757. J Antibiot 62:613–619
Mullane K, Lee C, Bressler A, Buitrago M, Weiss K, Dabovic K, Praestgaard J, Leeds JA, Blais J, Pertel P (2014) A multi-center, randomized clinical trial to compare the safety and efficacy of LFF571 and vancomycin for Clostridium difficile infections. Antimicrob Agents Chemother 59:1435–1440
Murakami T, Holt TG, Thompson CJ (1989) Thiostrepton-induced gene expression in Streptomyces lividans. J Bacteriol 171:1459–1466
Muraoka Y, Iinuma H, Takeuchi T, Okonogi T, inventors; US, assignee (2000) Amide derivative of amythiamicin. Patent 6043217. 1997-08-20
Myers CL, Hang PC, Ng G, Yuen J, Honek JF (2010) Semi-synthetic analogues of thiostrepton delimit the critical nature of tail region modifications in the control of protein biosynthesis and antibacterial activity. Bioorg Med Chem 18:4231–4237
Nagai K, Kamigiri K, Arao N, Suzumura K, Kawano Y, Yamaoka M, Zhang H, Watanabe M, Suzuki K (2003) YM-266183 and YM-266184, novel thiopeptide antibiotics produced by Bacillus cereus isolated from a marine sponge. I. taxonomy, fermentation, isolation, physico-chemical properties and biological properties. J Antibiot 56:123–128
Nagaraja TG, Avery TB, Bartley EE, Roof SK, Dayton AD (1982) Effect of lasalocid, monensin and thiopeptin on lactic acidosis in cattle. J Anim Sci 54:649–658
Naidu BN, Sorenson ME, Hudyma T, Zheng X, Zhang Y, Bronson JJ, Pucci MJ, Clark JM, Ueda Y (2004) Synthesis and antibacterial activity of O-substituted nocathiacin I derivatives. Bioorg Med Chem Lett 14:3743–3746
Naidu BN, Sorenson ME, Matiskella JD, Li W, Sausker JB, Zhang Y, Connolly TP, Lam KS, Bronson JJ, Pucci MJ (2006) Synthesis and antibacterial activity of nocathiacin I analogues. Bioorg Med Chem Lett 16:3545–3549
Naidu BN, Sorenson ME, Zhang Y, Kim OK, Matiskella JD, Wichtowski JA, Connolly TP, Li W, Lam KS, Bronson JJ, Pucci MJ, Clark JM, Ueda Y (2004) Nocathiacin I analogues: synthesis, in vitro and in vivo biological activity of novel semi-synthetic thiazolyl peptide antibiotics. Bioorg Med Chem Lett 14:5573–5577
Nakano I, Joshi K, Visnyei K, Hu B, Watanabe M, Lam D, Wexler E, Saigusa K, Nakamura Y, Dan RL, Mischel PS, Viapiano M, Kornblum HI (2011) Siomycin A targets brain tumor stem cells partially through a MELK-mediated pathway. NeuroOncol 13:622–634
Newman DJ, Cragg GM (2016) Natural products as sources of new drugs from 1981 to 2014. J Nat Prod 79:629–661
Nicolaou KC, Zak M, Rahimipour S, Estrada AA, Lee SH, O’Brate A, Giannakakou P, Ghadiri MR (2005) Discovery of a biologically active thiostrepton fragment. J Am Chem Soc 127:15042–15044
Ninomiya A, Kodani S (2011) Isolation and identification of berninamycin a from Streptomyces atroolivaceus. J Nat Prod 7:163–167
Nishimura H (1961) Siomycin, a new thiostrepton-like antibiotic. J Antibiot A 14:255–263
Northcote PT, Siegel M, Borders DB, Lee MD (1994a) Glycothiohexide alpha, a novel antibiotic produced by “Sebekia” sp., LL-14E605. III. Structural elucidation. J Antibiot 47:901–908
Northcote PT, Williams D, Siegel M, Borders DB, Maiese WM, Lee MD (1994b) Glycothiohexides, novel antibiotics produced by Sebekia sp LL-14E605 II isolation and physical–chemical characterization. J Antibiot 47:894–900
Nowak-Jary J, Andruszkiewicz R (2009) Antifungal activity of thionated analogues of NVA-FMDP and LYS-NVA-FMDP. Pol J Microbiol 58:295–299
Palomo S, González I, De La Cruz M, Martín J, Tormo JR, Anderson M, Hill RT, Vicente F, Reyes F, Genilloud O (2013) Sponge-derived Kocuria and Micrococcus spp. as sources of the new thiazolyl peptide antibiotic kocurin. Mar Drugs 11:1071–1086
Pellegrino RM, inventor EP, assignee (1984) Berninamycin A as a growth permittant. Patent 0112233. 1983-12-05
Porse BT, Cundliffe E, Garrett RA (1999) The antibiotic micrococcin acts on protein L11 at the ribosomal GTPase centre. J Mol Biol 287:33–45
Porse BT, Leviev I, Mankin AS, Garrett RA (1998) The antibiotic thiostrepton inhibits a functional transition within protein L11 at the ribosomal GTPase centre. J Mol Biol 276:391–404
Puar MS, Chan TM, Hegde V, Patel M, Bartner P, Ng KJ, Pramanik BN, Macfarlane RD (1998) Sch 40832: a novel thiostrepton from Micromonospora carbonacea. J Antibiot 51:221–224
Puar MS, Ganguly AK, Afonso A, Brambilla R, Mangiaracina P, Sarre O, Macfarlane RD (1981) Sch 18640. A new thiostrepton-type antibiotic. J Am Chem Soc 41:1220–1225
Pucci MJ, Bronson JJ, Barrett JF, Denbleyker KL, Discotto LF, Fungtomc JC, Ueda Y (2004) Antimicrobial evaluation of nocathiacins, a thiazole peptide class of antibiotics. Antimicrob Agents Chemother 48:3697–3701
Qiao S, Lamore SD, Cabello CM, Lesson JL, Mu Oz-Rodriguez JL, Wondrak GT (2012) Thiostrepton is an inducer of oxidative and proteotoxic stress that impairs viability of human melanoma cells but not primary melanocytes. Biochem Pharmacol 83:1229–1240
Radhakrishnan SK, Bhat UG, Hughes DE, Wang IC, Costa RH, Gartel AL (2006) Identification of a chemical inhibitor of the oncogenic transcription factor forkhead box M1. Cancer Res 66:9731–9735
Reusser F (1969) Mode of action of berninamycin. an inhibitor of protein biosynthesis. Biochemistry 8:3303–3308
Reynolds JM, Kamal EB, Jens B, Arthur G, Ben MC (2007) Antimalarial activity of the anticancer and proteasome inhibitor bortezomib and its analog ZL3B. BMC Clin Pharmacol 7:13
Rodnina MV, Savelsbergh A, Matassova NB, Katunin VI, Semenkov YP, Wintermeyer W (1999) Thiostrepton inhibits the turnover but not the GTPase of elongation factor G on the ribosome. Proc Natl Acad Sci USA 96:9586–9590
Rogers MJ, Bukhman YV, Mccutchan TF, Draper DE (1997) Interaction of thiostrepton with an RNA fragment derived from the plastid-encoded ribosomal RNA of the malaria parasite. RNA 3:815–820
Rosendahl G, Douthwaite S (1993) Ribosomal proteins L11 and L10.(L12)4 and the antibiotic thiostrepton interact with overlapping regions of the 23S rRNA backbone in the ribosomal GTPase centre. J Mol Biol 234:1013–1020
Rosendahl G, Douthwaite S (1994) The antibiotics micrococcin and thiostrepton interact directly with 23S rRNA nucleotides 1067A and 1095A. Nucleic Acids Res 22:357–363
Sajid I, Shaaban KA, Frauendorf H, Hasnain S, Laatsch H (2008) Val-geninthiocin: a thiopeptide antibiotic produced by Streptomyces sp. RSF18. J Biotechnol 136:75–98
Sajid I, Shaaban KA, Frauendorf H, Hasnain S, Laatscha H (2014) Val-geninthiocin: structure elucidation and MSN fragmentation of thiopeptide antibiotics produced by Streptomyces sp. RSF18. Z Naturforsch B 63:1223–1230
Sakai K, Komaki H, Gonoi T (2015) Identification and functional analysis of the nocardithiocin gene cluster in Nocardia pseudobrasiliensis. PLoS ONE 10:e143264
Sasaki T, Otani T, Matsumoto H, Unemi N, Hamada M, Takeuchi T, Hori M (1998) MJ347-81F4 A & B, novel antibiotics from Amycolatopsis sp.: taxonomic characteristics, fermentation, and antimicrobial activity. J Antibiot 51:715–721
Schlitzer M (2007) Malaria chemotherapeutics part I: history of antimalarial drug development, currently used therapeutics, and drugs in clinical development. ChemMedChem 2:944–986
Schoof S, Pradel G, Aminake MN, Ellinger B, Baumann S, Potowski M, Najajreh Y, Kirschner M, Arndt HD (2010) Antiplasmodial thiostrepton derivatives: proteasome inhibitors with a dual mode of action. Angew Chem Int Ed 49:3317–3321
Selva E, Beretta G, Montanini N, Saddler GS, Gastaldo L, Ferrari P, Lorenzetti R, Landini P, Ripamonti F, Goldstein BP, Berti M, Montanaro L, Denaro M (1991) Antibiotic GE2270A: a novel inhibitor of bacterial protein synthesis. I. Isolation and characterization. J Antibiot 44:693–701
Selva E, Ferrari P, Kurz M, Tavecchia P, Colombo L, Stella S, Restelli E, Goldstein BP, Ripamonti F, Denaro M (1995) Components of the GE2270 complex produced by Planobispora rosea ATCC 53773. J Antibiot 48:1039–1042
Sharma I, Sullivan M, Mccutchan TF (2015) The in vitro anti-malarial activity of novel semi synthetic nocathiacin I. Antibiotics. Antimicrob Agents Chemother 59:3174–3179
Shimanaka K, Iinuma H, Hamada M, Ikeno S, Tsuchiya KS, Arita M, Hori M (1995a) Novel antibiotics, amythiamicins. IV. A mutation in the elongation factor Tu gene in a resistant mutant of B. subtilis. J Antibiot 48:182–184
Shimanaka K, Kinoshita N, Iinuma H, Hamada M, Takeuchi T (1994a) Novel antibiotics, amythiamicins. I. Taxonomy, fermentation, isolation, physico-chemical properties, and antimicrobial activity. J Antibiot 47:668–674
Shimanaka K, Takahashi Y, Iinuma H, Naganawa H, Takeuchi T (1994b) Novel antibiotics, amythiamicins. III. Structure elucidations of amythiamicins A, B and C. J Antibiot 26:1153–1159
Shimanaka K, Takahashi Y, Iinuma H, Naganawa H, Takeuchi T (1995b) Novel antibiotics, amythiamicins. II. Structure elucidations of amythiamicins D. J Antibiot 26:1145–1152
Shoji J, Hinoo H, Wakisaka Y, Koizumi K, Mayama M, Matsuura S, Matsumoto K (1976) Isolation of three new antibiotics, thiocillins I, II and III, related to micrococcin P. studies on antibiotics from the genus Bacillus. VIII. J Antibiot 29:366–374
Shoji J, Kato T, Yoshimura Y, Tori K (1981) Structural studies on thiocillins I, II and III (studies on antibiotics from the genus Bacillus XXIX). J Antibiot 34:1126–1136
Si TI (1948) Micrococcin, an antibacterial substance formed by a strain of micrococcus. Br J Exp Pathol 29:473–481
Singh SB, Occi J, Jayasuriya H, Herath K, Motyl M, Dorso K, Gill C, Hickey E, Overbye KM, Barrett JF, Masurekar P (2007) Antibacterial evaluations of thiazomycin—a potent thiazolyl peptide antibiotic from Amycolatopsis fastidiosa. J Antibiot 60:565–571
Singh SB, Xu L, Meinke PT, Kurepina N, Kreiswirth BN, Olsen DB, Young K (2017) Thiazomycin, nocathiacin and analogs show strong activity against clinical strains of drug-resistant Mycobacterium tuberculosis. J Antibiot 70:671–674
Steinberg DA, Bernan VS, Montenegro DA, Abbanat DR, Pearce CJ, Korshalla JD, Jacobus NV, Petersen PJ, Mroczenskiwildey MJ, Maiese WM, Greenstein M (1994) Glycothiohexides, novel antibiotics produced by “Sebekia” sp. LL-14E605. I. taxonomy, fermentation and biological evaluation. J Antibiot 47:887–893
Stella S, Montanini N, Lemonnier FJ, Colombo L, Selva E, Denaro M, inventors; US, assignee (1997) Antibiotics GE37468A, B and C. Patent 5618724. 1996-05-16
Stella S, Montanini N, Monnier FL, Ferrari P, Colombo L, Landini P, Ciciliato I, Goldstein B, Denaro M, Selva E (1995) Antibiotic GE37468A—a new inhibitor of bacterial protein synthesis. I. Isolation and characterization. J Antibiot 48:780–786
Stutz MW, Johnson SL, Judith FR, Muir LA (1983) Effect of the antibiotic thiopeptin on Clostridium perfringens and growth and feed efficiency of broiler chicks. Poultry Sci 62:1633–1638
Sullivan M, Li J, Kumar S, Rogers MJ, Mccutchan TF (2000) Effects of interruption of apicoplast function on malaria infection, development, and transmission. Mol Biochem Parasitol 109:17–23
Suzumura K, Yokoi T, Funatsu M, Nagai K, Tanaka K, Zhang H, Suzuki K (2003) YM-266183 and YM-266184, novel thiopeptide antibiotics produced by Bacillus cereus isolated from a marine sponge II. Structure elucidation. J Antibiot 56:129–134
Takagi M, Motohashi K, Nagai A, Hashimoto J, Shinya K (2010) JBIR-83 and JBIR-84, new promothiocin derivatives, isolated from Streptomyces sp. RI19. J Antibiot 63:405–408
Tanaka T, Endo T, Shimazu A, Yoshida R, Suzuki Y, Otake N, Yonehara H (1970a) A new antibiotic, multhiomycin. J Antibiot 23:231–237
Tanaka T, Sakaguchi K, Yonehara H (1970b) On the mode of action of multhiomycin. I. Effects of multhiomycin on macromolecular syntheses. J Antibiot 23:401–407
Tanaka T, Sakaguchi K, Yonehara H (1971a) Inhibition by multhiomycin of T factor- and GTP-dependent binding of phenylalanyl-tRNA to ribosomes and GTP hydrolysis associated with it. J Biochem 69:1127–1130
Tanaka T, Sakaguchi K, Yonehara H (1971b) On the mode of action of multhiomycin. II. Effects of multhiomycin on Phe-tRNA binding to ribosomes and on other steps in protein synthesis. J Antibiot 24:537–542
Tavecchia P, Gentili P, Kurz M, Sottani C, Bonfichi R, Lociuro S, Selva E (1994) Revised structure of the antibiotic GE2270A. J Antibiot 47:1564–1567
Tavecchia P, Gentili P, Kurz M, Sottani C, Bonfichi R, Selva E, Lociuro S, Restelli E, Ciabatti R (1995) Degradation studies of antibiotic MDL 62,879 (GE2270A) and revision of the structure. Tetrahedron Lett 51:4867–4869
Thiemann JE, Coronelli C, Pagani H, Beretta G, Tamoni G, Arioli V (1968) Antibiotic production by new form-genera of the actinomycetales. I. Sporangiomycin, an antibacterial agent isolated from Planomonospora parontospora var. antibiotica var. nov. J Antibiot 21:525–531
Thompson J, Cundliffe E (1980) Resistance to thiostrepton, siomycin, and sporangiomycin in actinomycetes that produce them. J Bacteriol 142:455–461
Tokura K, Hayashi K, Okabe K, Tori K, Yoshimura Y, Mayama M, Matsuura S, Otsuka H (1981) Water-soluble siomycin-A derivatives. Preparation, chemical structures and biological properties of half-Esters of the peptide antibiotic. J Antibiot 34:800–810
Tori K, Tokura K, Okabe K, Ebata M, Otsuka H, Lukacs G (1976) Carbon-13 NMR studies of peptide antibiotics, thiostrepton and siomycin A: the structure relationship. Tetrahedron Lett 17:185–188
Tori K, Tokura K, Yoshimura Y, Terui Y, Okabe K, Otsuka H, Matsushita K, Inagaki F, Miyazawa T (1981) Structures of siomycin-B and -C and thiostrepton-B determined by NMR spectroscopy and carbon-13 signal assignments of siomycins, thiostreptons, and thiopeptin-B. J Antibiot 34:124–129
Trejo WH, Dean LD, Pluscec J, Meyers E, Brown WE (1977) Streptomyces laurentii, a new species producing thiostrepton. J Antibiot 30:639–643
Ueno M, Furukawa S, Abe F, Ushioda M, Fujine K, Johki S, Hatori H, Ueda H (2004) Suppressive effect of antibiotic siomycin on antibody production. J Antibiot 57:590–596
Uppoor GB, Marianna H, Andrei LG (2013) Evaluation of the effects of the thiopeptides on a panel of human cancer cell lines. PLoS ONE 4:e5922
Waitz JA, Patel MG, Marquez JA, Kalyanpur MG, Horan AC, inventors; US, assignee (1986) Antibiotic 13-384 complex from Micromonospora carbonacea var africana. Patent 4597968. 1984-06-21
Wang J, Lin Z, Bai X, Tao J, Liu W (2019) Optimal design of thiostrepton-derived thiopeptide antibiotics and their potential application against oral pathogens. Org Chem Front 2:1–15
Wang J, Yu Y, Tang K, Liu W, He X, Huang X, Deng Z (2010) Identification and analysis of the biosynthetic gene cluster encoding the thiopeptide antibiotic cyclothiazomycin in streptomyces hygroscopicus 10-22. Appl Environ Microbiol 76:2335–2344
Wang S, Zheng Q, Wang J, Zhao Z, Li Q, Yu Y, Wanga R, Liu W (2015) Target-oriented design and biosynthesis of thiostrepton-derived thiopeptide antibiotics with improved pharmaceutical properties. Org Chem Front 2:106–109
Wang S, Zheng X, Pan Q, Chen Y (2016) Mutagenesis of precursor peptide for the generation of nosiheptide analogues. RSC Adv 6:94643–94650
Wang S, Zhou S, Liu W (2013) Opportunities and challenges from current investigations into the biosynthetic logic of nosiheptide-represented thiopeptide antibiotics. Curr Opin Chem Biol 17:626–634
Watanabe S (1972) Interaction of siomycin with the acceptor site of Escherichia coli ribosomes. J Mol Biol 67:443–457
Watanabe S, Tanaka K (1971) Effect of siomycin on the acceptor site of Escherichia coli ribosomes. Biochem Biophys Res Commun 45:728
Weinstein MJ, Wagman GH, Marquez JA, Testa RT, inventors; US, assignee (1978) Antibiotic complex from Micromonospora arborensis. Patent 4078056. 1976-02-02
Wenying LI, Leet J, Huang X, Lam KS, Regueiro-Ren A, inventors; US, assignee (2002) Nocathiacin antibiotics prepared by biotransformation or chemical methods. Patent 013834. 2001-08-01
Wever WJ, Bogart JW, Baccile JA, Chan AN, Schroeder FC, Bowers AA (2015) Chemoenzymatic synthesis of thiazolyl peptide natural products featuring an enzyme-catalyzed formal [4 + 2] cycloaddition. J Am Chem Soc 137:3494–3497
Wierstra I (2013) The transcription factor FOXM1 (forkhead box M1): proliferation-specific expression, transcription factor function, target genes, mouse models, and normal biological roles. Adv Cancer Res 118:397–398
Xing Y, Draper DE (1996) Cooperative interactions of RNA and thiostrepton antibiotic with two domains of ribosomal protein L11. Biochemistry 35:1581–1588
Yeo WH, Kim SK, Kim SS, Yu SH, Park EK (1994a) Isolation, physico-chemical properties, and biological activity of new thiopeptide antibiotics, kimorexins. J Microbiol Biotechnol 4:349–353
Yeo WH, Kim SK, Kim SS, Yu SH, Park EK (1994b) Taxonomy and fermentation of Kitasatospora kimorexae producing new thiopeptide antibiotics, kimorexins. J Microbiol Biotechnol 4:354–359
Yoshimura Y, Koenuma M, Matsumoto K, Tori K, Terui Y (1988) NMR studies of chromomycins, olivomycins, and their derivatives. J Antibiot 41:53–67
Young TS, Dorrestein PC, Walsh CT (2012) Codon randomization for rapid exploration of chemical space in thiopeptide antibiotic variants. Chem Biol 19:1600–1610
Yun BS, Fujita KI, Furihata K, Seto H (2001) Absolute stereochemistry and solution conformation of promothiocins. Tetrahedron Lett 57:9683–9687
Yun BS, Hidaka T, Furihata K, Seto H (1994a) Microbial metabolites with tipA promoter inducing activity. II. geninthiocin, a novel thiopeptide produced by Streptomyces sp. DD84. J Antibiot 47:969–975
Yun BS, Hidaka T, Furihata K, Seto H (1994b) Microbial metabolites with tipA promoter inducing activity. III. thioxamycin and its novel derivative, thioactin, two thiopeptides produced by Streptomyces sp. DP94. J Antibiot 47:1541–1545
Yun BS, Hidaka T, Furihata K, Seto H (1994c) Promothiocins A and B novel thiopeptides with a tipA promoter inducing activity produced by Streptomyces sp. Sf2741. J Antibiot 47:510–514
Yun BS, Hidaka T, Furihata K, Seto H (1994d) Thiotipin, a novel thiopeptide with a tipa promoter inducing activity produced by Streptomyces sp. DT31. Tetrahedron 50:11659–11664
Yun BS, Seto H (1995) Promoinducin, a novel thiopeptide produced by Streptomyces sp. SF2741. Biosci Biotechnol Biochem 59:876–880
Zhang C, Herath K, Jayasuriya H, Ondeyka JG, Zink DL, Occi J, Birdsall G, Venugopal J, Ushio M, Burgess B, Masurekar P, Barrett JF, Singh SB (2009) Thiazomycins, thiazolyl peptide antibiotics from Amycolatopsis fastidiosa. J Nat Prod 72:841–847
Zhang C, Occi J, Masurekar P, Barrett JF, Zink DL, Smith S, Onishi R, Ha S, Salazar O, Genilloud O, Basilio A, Vicente F, Gill C, Hickey EJ, Dorso K, Motyl M, Singh SB (2008a) Isolation, structure, and antibacterial activity of philipimycin, a thiazolyl peptide discovered from Actinoplanes philippinensis MA7347. J Am Chem Soc 130:12102–12110
Zhang C, Zink DL, Ushio M, Burgess B, Onishi R, Masurekar P, Barrett JF, Singh SB (2008b) Isolation, structure, and antibacterial activity of thiazomycin A, a potent thiazolyl peptide antibiotic from Amycolatopsis fastidiosa. Bioorg Med Chem 16:8818–8823
Zhang F, Kelly WL (2015) Saturation mutagenesis of TsrA Ala4 unveils a highly mutable residue of thiostrepton A. ACS Chem Biol 10:998–1009
Zhang F, Li C, Kelly WL (2016) Thiostrepton variants containing a contracted quinaldic acid macrocycle result from mutagenesis of the second residue. ACS Chem Biol 11:415–424
Zuurmond AM, Martien de Graaf J, Olsthoorn-Tieleman LN, van Duyl BY, Mörhle VG, Jurnak F, Mesters JR, Hilgenfeld R, Kraal B (2000) GE2270A-resistant mutations in elongation factor Tu allow productive aminoacyl-tRNA binding to EF-TuGTP·GE2270A complexes. J Mol Biol 304:995–1005
Acknowledgements
This work was supported by Minjiang Scholar Project grants from Fujian Provincial Government and Fujian Agriculture and Forestry University (Grant no. 114-118360030).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Shen, X., Mustafa, M., Chen, Y. et al. Natural thiopeptides as a privileged scaffold for drug discovery and therapeutic development. Med Chem Res 28, 1063–1098 (2019). https://doi.org/10.1007/s00044-019-02361-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-019-02361-1