Abstract
Plastics are widely used for various applications. Once discarded, it is commonly known that they represent a high environmental threat due to their slow degradation; for this reason, there is an imminent need to replace these products with eco-friendlier ones. In the present work, four bacterial polyhydroxybutyrate (PHB) producers, two consortia, and two isolated strains were successfully recovered from the facilities of a paper-manufacturing industry. Spectroscopic studies of the biopolymers obtained from these bacteria corroborated their PHB production capabilities, ranging from 4.04 ± 0.16 to 23.82 ± 3.39 g/L. The characterization of the isolate that presented the highest production yield initially coded as E22 led to the identification of a Klebsiella pneumoniae strain, which, compared with other PHA bacterial producers reported to date, could be considered with high production potential. The strain E22 was grown in 5 different media prepared from fruit peel residues of banana, orange, papaya, watermelon, and melon, to determine its growth and PHA production capabilities in these low-cost media. The results obtained show different bacterial growth yields among the media tested, although PHB production yields and productivities were similar in all these low-cost media. Cellular accumulation of the biopolymer was higher in watermelon peel medium (8.4 × 10−10 g/CFU). These results reveal the potential of K. pneumoniae E22 for PHB production applications and establish encouraging alternatives to be broader explored regarding low-cost media that could enhance the scale-up of bacterial PHA production processes.

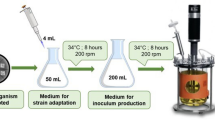
Graphical abstract






Similar content being viewed by others
References
Al-Salem SM, Lettieri P, Baeyens J (2010) The valorization of plastic solid waste (psw) by primary to quaternary routes: from re-use to energy and chemicals. Prog Energ Combust 36:103–129. https://doi.org/10.1016/j.pecs.2009.09.001.2010
Andrady AL, Neal MA (2009) Applications and societal benefits of plastics. Philos Trans R Soc Lond Ser B Biol Sci 364:1977–1984. https://doi.org/10.1098/rstb.2008.0304
Devasahayam S, Raman RKS, Chennakesavulu K, Bhattacharya S (2019) Plastics-villain or hero? Polymers and recycled polymers in mineral and metallurgical processing-a review. Materials 12:1–36. https://doi.org/10.3390/ma12040655
Sukan A, Roy I, Keshavarz T (2015) Dual production of biopolymers from bacteria. Carbohydr Polym 1:47–51. https://doi.org/10.1016/j.carbpol.2015.03.001
Šprajcar M, Horvat P, Krzan A (2012) Biopolymers and bioplastics–plastics aligned with nature. European Regional Development Fund. https://www.umsicht.fraunhofer.de/content/dam/umsicht/de/ dokumente/ueber-uns/nationale-infostelle-nachhaltige-kunststoffe/biopolymers-bioplastics-brochure-for-teachers.pdf. Accessed 14 February 2020
Serwanska-Leja K, Lewandowicz G (2010) Polymer biodegradation and biodegradable polymers-a review. Pol J Environ Stud 19:255–266. https://pdfs.semanticscholar.org/72a6/fb58bf5809b42af5861af6dd2541941e7858.pdf?_ga=2.69194769.1238613553.1588290677-1430679523.1583797552. Accessed 14 February 2020
Krueger CL, Radetski CM, Bendia AG, Oliveira IM, Castro-Silva MA, Rambo CR, Antonio RV, Lima AOS (2012) Bioconversion of cassava starch by-product into Bacillus and related bacteria polyhydroxyalkanoates. Electron J Biotechnol 15:1–13. https://doi.org/10.2225/vol15-issue3-fulltext-6
Sinha K, Rathore P (2015) Study of polyhydroxybutyrate producing Bacillus sp. isolated from soil. Res J Recent Sci 4:61–69. http://www.isca.in/rjrs/archive/v4/iIYSC-2015/12.ISCA-IYSC-2015-3BS-09.pdf. Accessed 18 February 2020
Mohapatra S, Maity S, Dash HR, Das S, Pattnaik S, Rath CC, Samantaray D (2017) Bacillus and biopolymer: prospects and challenges. Biochem Biophys Rep 12:206–213. https://doi.org/10.1016/j.bbrep.2017.10.001
Shah KR (2014) Optimization and production of polyhydroxybutarate (PHB) by Bacillus subtilis G1S1 from soil. Int J Curr Microbiol App Sci 3:377–387. https://www.ijcmas.com/vol-3-5/K.R.Shah.pdf. Accessed 9 March 2020
Apparao U, Krishnaswamy VG (2015) Production of polyhydroxyalkanoate (PHA) by a moderately halotolerant bacterium Klebsiella pneumoniae U1 isolated from rubber plantation area. Int J Environ Biorem Biodegrad 3:54–61. https://doi.org/10.12691/ijebb-3-2-3
Chen BY, Shiau TJ, Wei YH, Chen WM (2012) Feasibility study on polyhydroxybutyrate production of dye-decolorizing bacteria using dye and amine-bearing cultures. J Taiwan Inst Chem Eng 43:241–245. https://doi.org/10.1016/j.jtice.2011.09.001
Eiamsa-ard P (2017) Isolation and identification of polyhydroxyalkanoate (PHA) producing bacteria from food industrial wastewater by using fluorometric screening. Journal of KMUTNB 27:771–781. https://doi.org/10.14416/j.kmutnb.2017.11.004
Menaga M, Felix S, Gopalakannan A (2019) Screening of a biodegradable polymer, polyhydroxy butyrate (PHB) accumulating bacteria from the biofloc based gift tilapia ponds. Aquaculture 2019 - meeting abstract; World Aquaculture Society. https://www.was.org/meetings/ShowAbstract.aspx?Id= 136449. Accessed 9 March 2020
Narayankumar S, Krishnaswamy VG (2017) Characterization and optimization studies on poly[(R)-3-hydroxybutyrate] producing moderately halotolerant bacterial strains isolated from saline environment. J Biodivers Manage Forestry 6:3. https://doi.org/10.4172/2327-4417.1000181
Peña C, Castillo T, García A, Millán M, Segura D (2014) Biotechnological strategies to improve production of microbial poly-(3-hydroxybutyrate): a review of recent research work. Microb Biotechnol 7:278–293. https://doi.org/10.1111/1751-7915.12129.4
Pillai AB, Kumar AJ, Kumarapillai H (2018) Enhanced production of poly (3-hydroxybutyrate) in recombinant Escherichia coli and EDTA-microwave-assisted cell lysis for polymer recovery. AMB Express 8:1–15. https://doi.org/10.1186/s13568-018-0672-6
Sadaf N, Kamthane DC (2019) PHB production from bacteria isolated from oil contaminated soil. Bioscience Discovery 10:103-107. http://biosciencediscovery.com/Vol%2010%20No%202/Nausheen103-107.pdf. Accessed 9 March 2020
Tufail S, Munir S, Jamil N (2017) Variation analysis of bacterial polyhudroxyalkanoates production using saturated and unsaturated hydrocarbons. Braz J Microbiol 48:629–636. https://doi.org/10.1016/j.bjm.2017.02.008
Zhang H, Obias V, Gonyer K, Dennis D (1994) Production of polyhydroxyalkanoates in sucrose-utilizing recombinant Escherichia coli and Klebsiella strains. Appl Environ Microbiol 60:1198–1205. https://aem.asm.org/content/aem/60/4/1198.full.pdf. Accessed 19 February 2020
Kourmentza C, Plácido J, Venetsaneas N, Burniol-Figols A, Varrone C, Gavala HN, Reis MAM (2017) Recent advances and challenges towards sustainable polyhydroxyalkanoate (PHA) production. Bioengineering (Basel) 4:1–43. https://doi.org/10.3390/bioengineering4020055
Guerra-Blanco P, Cortes O, Poznyak T, Chairez I, García-Peña EI (2018) Polyhydroxyalkanoates (PHA) production by photoheterotrophic microbial consortia: effect of culture conditions over microbial population and biopolymer yield and composition. Eur Polym J 98:94–104. https://doi.org/10.1016/j.eurpolymj.2017.11.007
Nielsen C, Rahman A, Rehman AU, Walsh MK, Miller CD (2017) Food waste conversion to microbial polyhydroxyalkanoates. Microb Biotechnol 10:1338–1352. https://doi.org/10.1111/1751-7915.12776
Baird RB, Eaton AD, Rice EW (2001) Standard methods for the examination of water and wastewater. American Public Health Association. https://www.academia.edu/38769108/Standard_Methods_For_the_Examination_of_Water_and_Wastewater_23nd_edition. Accessed 15 May 2017
Bhuwal AK, Singh G, Aggarwal NK, Goyal V, Yadab A (2013) Isolation and screening of polyhydroxyalkanoates producing bacteria from pulp, paper, and cardboard industry wastes. Int J Biomater 2013:1–10. https://doi.org/10.1155/2013/752821
Rivas-Castillo AM, Valdez-Calderón A, Rojas-Avelizapa NG (2019) Culture medium to promote the production of biopolymers (in Spanish). Patent application no. 3895 submitted to the Ministry of Economy in 29 November 2019 for its presentation at the Mexican Institute of Industrial Property (IMPI)
Amutha R, Kavusik T, Sudha A (2017) Analysis of bioactive compounds in citrus fruit peels. International Journal of Scientific Research and Review 6:19–27. https://www.researchgate.net/publication/323551324_ANALYSIS_OF_BIOACTIVE_COMPOUNDS_IN_CITRUS_FRUIT_PEELS. Accessed 14 June 2019
Gusakov AV, Kondratyeva EG, Sinitsyn AP (2011) Comparison of two methods for assaying reducing sugars in the determination of carbohydrase activities. Int J Anal Chem 2011:283658–283654. https://doi.org/10.1155/2011/283658
Jones DB (1941) Factors for converting percentages of nitrogen in foods and feeds into percentages of protein. US Department of Agriculture - Circular No 183. https://www.ars.usda.gov/ARSUserFiles/80400525/Data/Classics/cir183.pdf. Accessed 14 June 2019
Mariotti F, Tomé D, Mirand PP (2008) Converting nitrogen into protein-beyond 6.25 and Jones Factors. Crit Rev Food Sci Nutr 48:177–184. https://doi.org/10.1080/10408390701279749
Hungund B, Shyama VS, Patwhardan P, Saleh AM (2013) Production of polyhydroxyalkanoate from Paenibacillus durus BV-1 isolated from oil mill soil. J Microb Biochem Technol 5:13–17. https://doi.org/10.4172/1948-5948.1000092
Sánchez-Moreno SA, Marín-Montoya MA, Mora-Martínez AL, Yepes-Pérez MS (2012) Identification of bacteria that produce polyhydroxyalcanoates (PHAs) in soils contaminated with residues of fique (in Spanish). Revista Colombiana de Biotecnología 16:89-100. https://revistas.unal.edu.co/ index.php/biotecnología/article/view/37286/39379. Accessed 26 May 2019
Thapa C, Shakya P, Shrestha R, Pal S, Manandhar P (2018) Isolation of polyhydroxybutyrate (PHB) producing bacteria, optimization of culture conditions for PHB production, extraction and characterization of PHB. Nepal Journal of Biotechnology 6:62–68. https://www.researchgate.net/publication/330403719_Isolation_of_Polyhydroxybutyrate_PHB_Producing_Bacteria_Optimization_of_Culture_Conditions_for_PHB_production_Extraction_and_Characterization_of_PHB. Accessed 5 August 2020
Kaur G, Roy I (2015) Strategies for large-scale production of polyhydroxyalkanoates. Chem Biochem Eng Q 29:157–172. https://doi.org/10.15255/CABEQ.2014.2255
Pincus DH (2005) Microbial identification using the bioMériueux VITEK® 2 system. In: Miller MJ (ed) Encyclopedia of rapid microbiological methods. PDA, Barcelona, pp. 1–32
Miller JH (1972) Experiments in molecular genetics. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York
Cutting SM, Vander Horn PB (1990) Genetic analysis. In: Harwood CR, Cutting SM (eds) Molecular biological methods for Bacillus. John Wiley & Sons, Sussex, pp 27–74
Ratul S, Farrance CE, Verghese B, Hong S, Donofrio RS (2013) Klebsiella michiganensis sp. nov., a new bacterium isolated from a tooth brush holder. Curr Microbiol 66:72–78. https://doi.org/10.1007/s00284-012-0245-x
Shakib P, Kalani MT, Ramazanzadeh R, Ahmadi A, Rouhi S (2018) Molecular detection of virulence genes in Klebsiella pneumoniae clinical isolates from Kurdistan Province, Iran. Biomedical Research and Therapy 5:2581–2589. http://www.bmrat.org/index.php/BMRAT/article/view/467/919. Accessed 5 June 2018
Ben M, Mato T, Lopez A, Vila M, Kennes C, Veiga MC (2011) Bioplastic production using wood mill effluents as feedstock. Water Sci Technol 63:1196–1202. https://doi.org/10.2166/wst.2011.358
Al-Rowaihi IS, Paillier A, Rasul S, Karan R, Grötzinger SW, Takanabe K, Eppinger J (2018) Poly (3-hydroxybutyrate) production in an integrated electromicrobial setup: investigation under stress inducing conditions. PLoS One 13:1–13. https://doi.org/10.1371/journal.pone.0196079
Toczyłowska-Mamińska R (2017) Limits and perspectives of pulp and paper industry wastewater treatment–a review. Renew Sust Energ Rev 78:764–772. https://doi.org/10.1016/j.rser.2017.05.021
Ghribi M, Meddeb-Mouelhi F, Beauregard M (2016) Microbial diversity in various types of paper mill sludge: identification of enzyme activities with potential industrial applications. Springerplus 5:1–14. https://doi.org/10.1186/s40064-016-3147-8
Karigar CS, Rao SS (2011) Role of microbial enzymes in the bioremediation of pollutants: a review. Enzyme Res 2011:1–11. https://doi.org/10.4061/2011/805187
Singh P, Parmar N (2011) Isolation and characterization of two novel polyhydroxybutyrate (PHB)-producing bacteria. Afr J Biotechnol 10:4907–4919. Accessed 7 November 2017
Flores-Vásquez ADP, Idrogo E-III, Carreño-Farfán CR (2018) Yield of polyhydroxyalcanoates (PHA) in halophilic microorganisms isolated from salt flats (in Spanish). Revista Peruana de Biología 25:153–160. https://doi.org/10.15381/rpb.v25i2.14249
Ashby RD, Solaiman DKY, Strahan GD (2011) Efficient utilization of crude glycerol as fermentation substrate in the synthesis of poly (3-hydroxybutyrate) biopolymers. J Am Oil Chem Soc 88:949–959. https://doi.org/10.1007/s11746-011-1755-6
Silva AF, Taciro MK, Michelin-Ramos ME, Carter JM, Pradella JGC, Gomez JGC (2004) Poly-3-hydroxybutyrate (P3HB) production by bacteria from xylose, glucose and sugarcane bagasse hydrolysate. J Ind Microbiol Biotechnol 31:245–254. https://doi.org/10.1007/s10295-004-0136-7
Zhu C, Nomura CT, Perrotta JA, Stipanovic AJ, Nakas JP (2010) Production and characterization of poly-3-hydroxybutyrate from biodiesel-glycerol by Burkholderia cepacia ATCC 17759. Biotechnol Prog 26:424–430. https://doi.org/10.1002/btpr.355
Li R, Jiang Y, Wang X, Yang J, Gao Y, Zi X, Zhang X, Gao H, Hu N (2013) Psychrotrophic Pseudomonas mandelii CBS-1 produces high levels of poly-β-hydroxybutyrate. Springerplus 2:1–7. https://doi.org/10.1186/2193-1801-2-335
Oliveira FC, Dias ML, Castilho LR, Freire FMG (2007) Characterization of poly (3-hydroxybutyrate) produced by Cupriavidus necator in solid-state fermentation. Bioresour Technol 98:633–638. https://doi.org/10.1016/j.biortech.2006.02.022
Kiran GS, Lipton AN, Anitha SPK, Cruz-Suárez LE, Arasu MV, Choi KC, Selvin J, Al-Dhabi NA (2014) Antiadhesive activity of poly-hydroxy butyrate biopolymer from a marine Brevibacterium casei MSI04 against shrimp pathogenic vibrios. Microb Cell Factories 13:1–12. https://doi.org/10.1186/s12934-014-0114-3
Mohapatra S, Sarkar B, Samantaray DP, Daware A, Maity S, Pattnaik S, Bhattacharjee S (2017) Bioconversion of fish solid waste into PHB using Bacillus subtilis based submerged fermentation process. Environ Technol 38:3201–3208. https://doi.org/10.1080/09593330.2017.1291759
Egli T (2015) Microbial growth and physiology: a call for better craftsmanship. Front Microbiol 6:1–12. https://doi.org/10.3389/fmicb.2015.00287
Penkhrue W, Jendrossek D, Khanongnuch C, Pathom-aree W, Aizawa T, Behrens RL, Lumyong S (2020) Response surface method for polyhydroxybutyrate (PHB) bioplastic accumulation in Bacillus drentensis BP17 using pineapple peel. PLoS One 15:e0230443. https://doi.org/10.1371/journal.pone.0230443
Batool N, Shakir HA, Qazi JI (2015) Comparative growth potential of different Bacillus species on fruit peels. Punjab University Journal of Zoology 30:25-29. http://pu.edu.pk/images/journal/zology/ PDF-FILES/6-Comparative%20growth%20potential%20of%20different%20Bacillus%20species%20 on% 20fruit%20peels.pdf. Accessed 12 June 2019
Lyte M (1997) Induction of gram-negative bacterial growth by neurochemical containing banana (Musa x paradisiaca) extracts. FEMS Microbiol Lett 154:245–250. https://doi.org/10.1111/j.1574-6968.1997.tb12651.x
Torrado AM, Cortés S, Salgado JM, Max B, Rodríguez N, Bibbins BP, Converti A, Domínguez JM (2011) Citric acid production from orange peel wastes by solid- state fermentation. Braz J Microbiol 42:394–409. https://doi.org/10.1590/S1517-83822011000100049
Golden DA, Rhodehamel EJ, Kautter DA (1993) Growth of Salmonella spp. in cantaloupe, watermelon, and honeydew melons. J Food Prot 56:194–196. https://doi.org/10.4315/0362-028X-56.3.194
Blin C, Passet V, Touchon M, Rocha EPC, Brisse S (2017) Metabolic diversity of the emerging pathogenic lineages of Klebsiella pneumoniae. Environ Microbiol 19:1881–1898. https://doi.org/10.1111/1462-2920.13689
Lu X, Ji G, Zong H, Zhuge B (2016) The role of budABC on 1,3-propanediol and 2,3-butanediol production from glycerol in Klebsiella pneumoniae CICIM B0057. Bioengineered 7:439–444. https://doi.org/10.1080/21655979.2016.1169355
Sun S, Zhang H, Lu S, Lai C, Liu H, Zhu H (2016) The metabolic flux regulation of Klebsiella pneumoniae based on quorum sensing system. Sci Rep 6:1–9. https://doi.org/10.1038/srep38725
Afzal AM, Rasool MH, Waseem M, Aslam M (2017) Assessment of heavy metal tolerance and biosorptive potential of Klebsiella variicola isolated from industrial effluents. AMB Express 7:1–9. https://doi.org/10.1186/s13568-017-0482-2
Aransiola EF, Ige OA, Ehinmitola EO, Lakoyun SK (2017) Heavy metals bioremediation potential of Klebsiella species isolated from diesel polluted soil. Afr J Bitechnol 16:1098–1105. https://doi.org/10.5897/AJB2016.15823
Muñoz AJ, Espinola F, Ruiz E (2015) Removal of Pb(II) in a packed-bed column by a Klebsiella sp. 3S1 biofilm supported on porous ceramic Raschig rings. J Ind Eng Chem 40:118–127. https://doi.org/10.1016/j.jiec.2016.06.012
Muñoz AJ, Espinola F, Ruiz ER (2017) Removal of heavy metals by Klebsiella sp. 3S1. Kinetics, equilibrium and interaction mechanisms of Zn (II) biosorption. J Chem Technol Biotechnol 93:1213–1511. https://doi.org/10.1002/jctb.5503
Cardona-Echavarría AC, Mora-Martínez AL, Marín-Montoya M (2013) Molecular identification of bacteria that produce polyhydroxyalcanoates in dairy subproducts and sugar cane (in Spanish). Revista Facultad Nacional de Agronomía Medellín 66:7129-7140. https://revistas.unal.edu.co/index.php/refame/ article/view/41230/42790. Accessed 5 November 2018
Fathima N, Krishnaswamy VG (2016) Optimization of poly-β-hydroxybutyrate production by halotolerant bacterial strains isolated from saline environment. Int J Bioassays 5.8:4775-4781. https://www.doi.org/2F10.21746%2Fijbio.2016.08.0011
Favaro L, Basaglia M, Casella S (2018) Improving polyhydroxyalkanoate production from inexpensive carbon sources by genetic approaches: a review. Biofuels Bioprod Biorefin 13:208–227. https://doi.org/10.1002/bbb.1944
Mitrea L, Vodnar DC (2019) Klebsiella pneumoniae—a useful pathogenic strain for biotechnological purposes: diols biosynthesis under controlled and uncontrolled pH levels. Pathogens 8:1–10. https://doi.org/10.3390/pathogens8040293
Acknowledgments
We deeply thank Prof. Gilberto José López de la Mora, Coordinator of the Scientific Illustration Program of the Universidad Tecnológica de la Zona Metropolitana del Valle de México, for his valuable assistance in the elaboration of the graphical abstract. Biochemical characterization of strain E22 was performed in Acosta Group—Laboratories, in Tizayuca, Hgo., Mexico.
Funding
This project was supported by the budget from the Annual Operational Program assigned to the Universidad Tecnológica de la Zona Metropolitana del Valle de México for Scientific, Technological, and Educational Research Projects, item numbers 251001 and 255001 for years 2017 and 2018.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 121 kb)
Rights and permissions
About this article
Cite this article
Valdez-Calderón, A., Barraza-Salas, M., Quezada-Cruz, M. et al. Production of polyhydroxybutyrate (PHB) by a novel Klebsiella pneumoniae strain using low-cost media from fruit peel residues. Biomass Conv. Bioref. 12, 4925–4938 (2022). https://doi.org/10.1007/s13399-020-01147-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13399-020-01147-5