Abstract
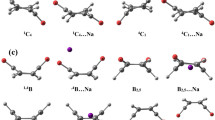
It is generally accepted that the structural characteristics of a molecule determine its physical and electrochemical properties. In this study, the conformations and some electrochemical properties of various boronates were investigated through computational study using density functional theory (DFT) with the Becke’s three-parameter hybrid method utilizing the Lee–Young–Parr correlation functional (B3LYP). After initial energy optimization using Møller–Plesset perturbation theory (MP2), the conformational preferences and energetics were investigated using DFT calculations and the 6-31G(d,p) basis set in vacuo. The calculations and first results show that the ethyleneoxyboronates can be expected to perform well as redox shuttles, and boron-based redox shuttles can contribute to overcharge protection and safer batteries. HOMO–LUMO energy differences also indicate higher reactivities of the boronates, contributing to better solid electrolyte interphase formation.
Similar content being viewed by others
References
Chen Z.H., Amine K.: Functional electrolyte additive for lithium-ion batteries. Electrochem. Commun. 9, 703–707 (2007)
Xiao Q.C., Liu H.Y., Xia Q.L., Xiao Q.Z., Lei G.T., Li Z.H.: A nanocomposite polymer electrolyte with high-temperature stability for rechargeable lithium batteries. Arab. J. Sci. Eng. 39, 6651–6657 (2014)
Moshuchak L.M., Bulinski M., Lamanna W.M., Wang R.L., Dahn J.R.: Direct comparison of 2,5-di-tert-butyl-1,4-dimethoxybenzene and 4-tert-butyl-1,2-dimethoxybenzene as redox shuttles in LiFePO4-based Li-ion cells. Electrochem. Commun. 9, 1497–1501 (2007)
Zhang S.S.: A review on electrolyte additives for lithium-ion batteries. J. Power Sources 162, 1379–1394 (2006)
Xia Q., Wang B., Wu Y.P., Luo H.J., Zhao S.Y., van Ree T.: Phenyl tris-2-methoxydiethoxysilane as an additive to PC-based electrolytes for lithium-ion batteries. J. Power Sources 180, 602–606 (2008)
Zhang H.P., Xia Q., Wang B., Yang L.C., Wu Y.P., Sun D.L., Gan C.L., Luo H.J., Bebeda A.W., van Ree T.: Vinyl-tris-(methoxydiethoxy)silane as an effective and ecofriendly flame retardant for electrolytes in lithium ion batteries. Electrochem. Comm. 11, 526–529 (2009)
Li L.L., Li L., Wang B., Liu L.L., Wu Y.P., van Ree T., Thavhiwa K.A.: Methyl phenyl bis-methoxydiethoxysilane as bi-functional additive to propylene carbonate-based electrolyte for lithium ion batteries. Electrochim. Acta 56, 4858–4864 (2011)
Wang B., Qu Q.T., Xia Q., Wu Y.P., Li X., Gan C.L., van Ree T.: Effects of 3,5-bis(trifluoromethyl)benzeneboronic acid as an additive on electrochemical performance of propylene carbonate-based electrolytes for lithium ion batteries. Electrochim. Acta 54, 816–820 (2008)
Xiao L.F., Ai X.P., Cao Y.L., Wang Y.D., Yang H.X.: A composite polymer membrane with reversible overcharge protection mechanism for lithium ion batteries. Electrochem. Commun. 7, 589–592 (2005)
Wang R.L., Buhrmester C., Dahn J.R.: Calculations of oxidation potentials of redox shuttle additives for Li-ion cells. J. Electrochem. Soc. 153, A445–A449 (2006)
Frisch M.J., Trucks G.W.: Gaussian 03, Revision D.01. Gaussian, Inc., Wallingford (2004)
Dennington R., Keith T., Millam J.: GaussView, Version 4.1. Semichem Inc., Shawnee Mission (2006)
Ufheil J., Baertsch M.C., Wursig A., Novak P.: Acetone as oxidative decomposition product in propylene carbonate containing battery electrolyte. Electrochim. Acta 50, 1733–1738 (2005)
Author information
Authors and Affiliations
Corresponding author
Additional information
Based on 10th International Conference on Novel Materials and their Synthesis (NMS) – China.
Rights and permissions
About this article
Cite this article
Bebeda, A.W., van Ree, T. Conformational Preferences and Electrochemical Performance of Ethyleneoxy Phenylboronate Electrolyte Additives. Arab J Sci Eng 40, 2841–2851 (2015). https://doi.org/10.1007/s13369-015-1669-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13369-015-1669-y