Abstract
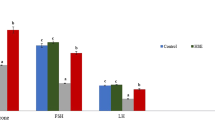
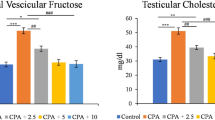
Cultivated wild ginseng has a variety of pharmacological effects. The aim of this study was to investigate the effects of cultivated wild ginseng extracts (CWGE) on apoptosis. CWGE showed the ability to protect hormone production (testosterone and progesterone from in vitro results) in TM3 Leydig and TM4 Sertoli cells damaged by Bisphenol A (BPA). CWGE also showed the ability to protect production of testosterone (7.24%, P<0.05), plasma luteinizing hormone (LH) (32.67%, P<0.05), and follicle-stimulating hormone (FSH) (37.34%, P<0.05) compared to the control and BPA-only groups. Reduction of apoptotic protein expression and induction of anti-apoptotic protein expression was observed in cells and rats pretreated with CWGE. These proteins are expressed through the ERK and p38 signalling pathways. In addition, CWGE might be a useful herbal medicine of cellular defense agents as our results in cells and animals.
Similar content being viewed by others
References
World Health Organization. Infertility/subfertility. Extract from Meeting to develop a global consensus on preconception care to reduce maternal and childhood mortality and morbidity. http://www.who.int/reproduc tivehealth/publications/infertility/978924150500 0_ annex3/en/ (2013).
Guzick, D. S. et al. Sperm morphology, motility, and concentration in fertile and infertile men. N Engl J Med 345:1388–1393 (2001).
Irvine, D. S. Epidemiology and aetiology of male infertility. Hum Reprod 13:33–44 (1998).
Paasch, U. et al. Semen quality in sub-fertile range for a significant proportion of young men from the general German population: a co-ordinated, controlled study of 791 men from Hamburg and Leipzig. Int J Androl 31:93–102 (2008).
Carlsen, E., Giwercman, A., Keiding, N. & Skakkebaek, N. E. Evidence for deacreasing quality of semen during past 50 years. Br Med J 305:609–613 (1992).
Vandenberg, L. N. et al. Bisphenol-A and the great divide: a review of controversies in the field of endocrine disruption. Endocr Rev 30:75–95 (2009).
Patisaul, H. B. & Adewale, H. B. Long-term effects of environmental endocrine disruptors on reproductive physiology and behavior. Front Behav Neurosci 3:10 (2009).
Richter, C. A. et al. In vivo effects of bisphenol A in laboratory rodent studies. Reprod Toxicol 24:199–224 (2007).
Al-Hiyasat, A. S., Darmani, H. & Elbetieha, A. M. Effects of bisphenol A on adult male mouse fertility. Eur J Oral Sci 110:163–167 (2002).
Takahashi, O. & Oishi, S. Testicular toxicity of dietary 2,2-bis(4-hydroxyphenyl)propane (bisphenol A) in F344 rats. Arch Toxicol 75:42–51 (2001).
Toyama, Y. et al. Adverse effects of bisphenol A to spermiogenesis in mice and rats. Arch Histol Cytol 67:373–381 (2004).
Toyama, Y. & Yuasa, S. Effects of neonatal administration of 17beta-estradiol, beta-estradiol 3-benzoate, or bisphenol A on mouse and rat spermatogenesis. Reprod Toxicol 19:181–188 (2004).
Li, M. W., Mruk, D. D., Lee, W. M. & Cheng, C. Y. Disruption of the blood-testis barrier integrity by bisphenol A in vitro: is this a suitable model for studying blood-testis barrier dynamics? Int J Biochem Cell Biol 41:2302–2314 (2009).
Farabollini, F. et al. Effects of perinatal exposure to bisphenol A on sociosexual behavior of female and male rats. Environ Health Perspect 110:409–414 (2002).
Gartner, A., Boag, P. P. & Blackwell, T. K. Germline survival and apoptosis. Wormbook 4:1–20 (2008).
Yu, Q. & Wu, J. Involvement of cyclins in mammalian spermatogenesis. Mol Cell Biochem 315:17–24 (2008).
Saikumar, P. et al. Apoptosis: definition, mechanisms, and relevance to disease. Am J Med 107:489–506 (1999).
Print, C. G. & Loveland, K. L. Germ cell suicide: new insights into apoptosis during spermatogenesis. Bioessays 22:423–430 (2000).
Tesarik, J., Greco, E., Cohen-Bacrie, P. & Mendoza, C. Germ cell apoptosis in men with complete and incomplete spermiogenesis failure. Mol Hum Reprod 4:757–762 (1998).
Gupta, S. Molecular signaling in death receptor and mitochondrial pathways of apoptosis. Int J Oncol 22:15–20 (2003).
Danial, N. N. & Korsmeyer, S. J. Cell death: critical control points. Cell 116:205–219 (2004).
Lee, S. T. et al. Panax ginseng enhances cognitive performance in Alzheimer disease. Alzheimer Dis Assoc Disord 22:222–226 (2008).
Spelman, K. et al. Modulation of cytokine expression by traditional medicines: a review of herbal immunomodulators. Altern Med Rev 11:128–150 (2006).
Wang, A. et al. Determination of major ginsenosides in Panax quinquefolius (American ginseng) using highperformance liquid chromatography. Phytochem Anal 16:272–277 (2005).
Christensen, L. P. Ginsenosides chemistry, biosynthesis, analysis, and potential health effects. Adv Food Nutr Res 55:1–99 (2009).
Nakamura, D. et al. Bisphenol A may cause testosterone reduction by adversely affecting both testis and pituitary systems similar to estradiol. Toxicol Lett 194:16–25 (2010).
Peretza, J. & Flawsb, J. A. Bisphenol A down-regulates rate-limiting Cyp11a1 to acutely inhibit steroidogenesis in cultured mouse antral follicles. Toxicol Appl Pharmacol 271:249–256 (2013).
Wang, Q. et al. Mitochondrial signaling pathway is also involved in bisphenol A induced germ cell apoptosis in testes. Toxicol Lett 199:129–135 (2010).
Shafi, G. et al. Induction of apoptosis in HeLa cells by chloroform fraction of seed extracts of Nigella sativa. Cancer Cell Int 9:29 (2009).
Pommier, Y. et al. Apoptosis defects and chemotherapy resistance: molecular interaction maps and networks. Oncogene 23:2934–2949 (2004).
Alshatwi, A. A. et al. Fenugreek induced apoptosis in breast cancer MCF-7 cells mediated independently by fas receptor change. Asian Pac J Cancer Prev 14:5783–5788 (2013).
Choi, H. S. et al. Ethanol extract of paeonia suffruticosa Andrews (PSE) induced AGS human gastric cancer cell apoptosis via fas-dependent apoptosis and MDM2–p53 pathways. J Biomed Sci 19:82 (2012).
Al-Hiyasat, A. S., Darmani, H. & Elbetieha, A. M. Effects of bisphenol A on adult male mouse fertility. Eur J Oral Sci 110:163–167 (2002).
Jiang, G. Z. & Li, J. C. Protective effects of ginsenoside Rg1 against colistin sulfate-induced neurotoxicity in PC12 cells. Cell Mol Neurobiol 34:167–172 (2014).
Wang, L. et al. Protective effects of ginsenosides against bisphenol A-induced cytotoxicity in 15P1 Sertoli cells via extracellular signal-regulated kinase 1/2 signalling and antioxidant mechanisms. Basic Clin Pharmacol Toxicol 111:42–49 (2012).
Park, W. H. et al. Cytoprotective effect of Panax ginseng on gallic acid-induced toxicity in TM3 mouse Leydig cells. Fitoterapia 78:577–579 (2007).
de Kretser, D. M. et al. Spermatogenesis. Hum Reprod 13:1–8 (1998).
Li, M. W., Mruk, D. D. & Cheng, C. Y. Mitogen-activated protein kinases in male reproductive function. Trends Mol Med 15:159–168 (2009).
Thuillier, R., Manku, G., Wang, Y. & Culty, M. Changes in MAPK pathway in neonatal and adult testis following fetal estrogen exposure and effects on rat testicular cells. Microsc Res Tech 72:773–786 (2009).
Boockfor, F. R. & Blake, C. A. Chronic administration of 4-tert-octylphenol to adult male rats causes shrinkage of the testes and male accessory sex organs, disrupts spermatogenesis, and increases the incidence of sperm deformities. Biol Reprod 57:267–277 (1997).
Wisniewski, P. et al. Adult exposure to bisphenol A (BPA) in Wistar rats reduces sperm quality with disruption of the hypothalamic-pituitary-testicular axis. Toxicology 329:1–9 (2015).
Salvati, G. Effects of Panax Ginseng C.A. Meyer saponins on male fertility. Panminerva Med 38:249–254 (1996).
Rao, L. & White, E. Bcl-2 and the ICE family of apoptotic regulators: making a connection. Curr Opin Genet Dev 7:52–58 (1997).
Shimizu, S. et al. Bcl-2 expression prevents activation of the ICE protease cascade. Oncogene 12:2251–2257 (1996).
Shi, J., Fujieda, H., Kokubo, Y. & Wake, K. Apoptosis of neutrophils and their elimination by Kupffer cells in rat liver. Hepatology 24:1256–1263 (1996).
Spreafico, R. et al. In situ labeling of apoptotic cell death in the cerebral cortex and thalamus of rats during development. J Comp Neurol 363:281–295 (1995).
Valverde, F., Lopez-Mascaraque, L., Santacana, M. & De Carlos, J. A. Persistence of early-generated neurons in the rodent subplate: assessment of cell death in neocortex during the early postnatal period. J Neurosci 15:5014–5024 (1995).
Dobrzynska, M. M. & Radzikowska, J. Genotoxicity and reproductive toxicity of bisphenol A and X-ray/ bisphenol A combination in male mice. Drug Chem Toxicol 36:19–26 (2013).
Chitra, K. C., Latchoumycandane, C. & Mathur, P. P. Induction of oxidative stress by bisphenol A in the epididymal sperm of rats. Toxicology 185:119–127 (2003).
Ok, S., Kang, J. S. & Kim, K. M. Simultaneous analysis method for polar and non-polar ginsenosides in cultivated wild ginseng by reversed-phase HPLC-CAD. J Life Sci 26:247–252 (2016).
EPA (U.S. Environmental Protection Agency), Bisphenol A, CASRN 80–05-7, IRIS, Integrated Risk Information System, on-line. Available on the Internet at http://www.epa.gov/iris/ (1993).
Kim, K. M., Seo, J. L. & Kang, J. S. Decursin and decursinol angelate affect spermatogenesis in the adult rat at oral administration. Mol Cell Toxicol 10:83–89 (2014).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Ok, S., Kang, J.S. & Kim, K.M. Cultivated wild ginseng extracts upregulate the anti-apoptosis systems in cells and mice induced by bisphenol A. Mol. Cell. Toxicol. 13, 73–82 (2017). https://doi.org/10.1007/s13273-017-0008-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13273-017-0008-7