Abstract
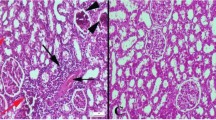
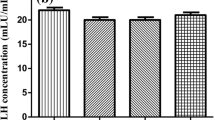
Infertility affects 15% of global population. This study was designed to search out the most effective dose of chloroform fraction of hydro-ethanolic extract of Hygrophila auriculata seed to ameliorate cyproterone acetate (CPA)-treated male subfertility. The rats were made subfertile by CPA at the dose of 2.5 mg/100gm body weight for 45 days. The male subfertility represented by low sperm concentration, less motile, less viable, and less hypo osmotic tail swelled spermatozoa in CPA-treated group. Serum LH, FSH, and testosterone levels were significantly decreased in CPA-treated group in respect to control. Androgenic key enzyme Δ5,3β-HSD, 17β-HSD activities and gene expression pattern were also decreased significantly in respect to control. These antispermatogenic and antiandrogenic activities of CPA were significantly recovered after the treatment of Hygrophila auriculata at the dose of 2.5 mg, 5mg, and 10 mg/100gm body weight. CPA also generate oxidative free radical that indicated by altered catalase, superoxide dismutase, and peroxidase activities and protein expression pattern along with conjugated diene and thiobarbituric acid reactive substance levels in testis. Expression pattern of Bax and Bcl2 genes were deviated from control after CPA treatment. Significant diminution of body weight, organo-somatic indices, and SGOT, SGPT activities were observed in CPA-treated group. All these biomarkers significantly recovered towards control after the treatment of Hygrophila auriculata at different doses. More significant recovery was observed in 5 mg and 10 mg of chloroform fraction-treated group and 5 mg dose, i.e., the minimum therapeutic dose to recover the CPA-induced subfertility.










Similar content being viewed by others
References
Kumar N, Singh A. Trends of male factor infertility, an important cause of infertility: a review of literature. J Hum Reprod Sci. 2015;8:191–6. https://doi.org/10.4103/0974-1208.170370.
Paul D, Mallick C, Ali KM, Nandi DK, Ghosh D. Duration dependent effect of hydro-ethanolic extract of leaf of S. hernandifolia and root of A. aspera on testicular androgenic and gametogenic activity: an approach for male herbal contraceptive development - Open Access Library. Int J of Appl Nat Prod. 2009;2:1–10. https://www.oalib.com/paper/2590093#.Yh5s2KtBy5c
Benagiano G, Carrara S, Filippi V. Sex and reproduction: an evolving relationship. Hum Reprod Update. 2009;16:96–107. https://doi.org/10.1093/humupd/dmp028.
Singh V, Nettemu S, Nettem S, Hosadurga R, Nayak S. Oral health and erectile dysfunction. J of Hum Reprod Sci. 2017;10:162. https://doi.org/10.4103/jhrs.jhrs_87_17.
Tripathy A, Ghosh A, Dey A, Pakhira BP, Ghosh D. Attenuation of the cyproterone acetate-induced testicular hypofunction by a novel nutraceutical lycopene: a genomic approach. Andrologia. 2016;49:e12709. https://doi.org/10.1111/and.12709.
Nantia EA, Moundipa PF, Monsees TK, Carreau S. Medicinal plants as potential male anti-infertility agents: a review. Basic Clin Androl. 2009;19:148–58. https://doi.org/10.1007/s12610-009-0030-2.
Palermo G, Joris H, Derde M-P, Camus M, Devroey P, Van Steirteghem A. Sperm characteristics and outcome of human assisted fertilization by subzonal insemination and intracytoplasmic sperm injection. Int J of Gynecol Obstet. 1994;44:93–3. https://doi.org/10.1016/0020-7292(94)90035-3.
Van Steirteghem A, Tournaye H, Van der Elst J, Verheyen G, Liebaers I, Devroey P. Intracytoplasmic sperm injection three years after the birth of the first ICSI child. Hum Reprod. 1995;10:2527–8.
Neumann F. Pharmacology and potential use of cyproterone acetate. HormMetab Res. 1977;9:1–13. https://doi.org/10.1055/s-0028-1093574.
Ghosh C, Mallick C. Protective effect of ethanolic extract of Hygrophila auriculata seeds in cyproterone acetate-induced sexual dysfunction in male albino rats. Andrologia. 2019;52 https://doi.org/10.1111/and.13482.
Chauhan NS, Sharma V, Dixit VK. Effect of Asteracantha longifolia seeds on the sexual behavior of male rats. Nat Prod Res. 2011;25:1423–31. https://doi.org/10.1080/14786410802588493.
Nadkarni AK. Dr. KM Nadkarni’s Indian materiamedica: with Ayurvedic, Unani-Tibbi, Siddha, allopathic, homeopathic, naturopathic & home remedies, appendices & indexes. India: Popular Prakashan; 1996.
MadhavBahadurKarki RJ, Asia I. Tribal folk medicinal plant resources of South Asia: report of the South Asia conference on tribal folk medicinal plant resources, September 4-6, 1996, Tirupati, India. Canada: Medicinal and Aromatic Plants Program In Asia, International Development Research Centre; 1999.
Beentje HJ, Watve A, Molur S. Hygrophila schulli. IUCN Red List of Threatened Species. 2016. https://indiabiodiversity.org/species/show/229999
On C. Health Implication of Obesity. Guide for the care and use of laboratory animals. United States: Dept. of Health and Human Services, Public Health Service, National Insititutes of Health; 1985.
World Health Organization, editor. WHO laboratory manual for the examination and processing of human semen. 5th ed. World Health Organization, Cop; 2010.
Ramu S, Jeyendran RS. The hypo-osmotic swelling test for evaluation of sperm membrane integrity. Methods Mol Biol. 2012;927:21–5. https://doi.org/10.1007/978-1-62703-038-0_3.
Jarabak J, Adams JA, Williams-Ashman HG, Talalay P. Purification of a 17β-hydroxysteroid dehydrogenase of human placenta and studies on its transhydrogenase function. J of Biol Chem. 1962;237:345–57. https://doi.org/10.1016/s0021-9258(18)93926-8.
Talalay P. [69] Hydroxysteroid dehydrogenases: hydroxysteroid + DPN + (TPN+) ⇄Ketosteroid DPNH (TPNH) + H+. Methods Enzymol. 1962;5:512–26. https://doi.org/10.1016/S0076-6879(62)05269-6.
Karvonen MJ, Malm M. Colorimetric determination of fructose with indol. Scand J Clin Lab Invest. 1955;7:305–7. https://doi.org/10.3109/00365515509134669.
Rat T(testosterone) ELISA kit. FineTest ELISA kit. Wuhan Fine Biotech Co., Ltd. Published August 26, 2019. https://www.fn-test.com/content/uploads/product/manuals/elisa/ER1462.pdf Accessed January 2, 2021.
Rat LH(luteinizing hormone) ELISA kit. FineTest ELISA kit. Wuhan Fine Biotech Co., Ltd. Published August 26, 2019. https://www.fn-test.com/content/uploads/product/manuals/elisa/ER1123.pdf Accessed January 2, 2021.
Rat FSH(follicle stimulating hormone) ELISA kit. FineTest ELISA Kit. Wuhan Fine Biotech Co., Ltd. Published August 26, 2019. https://www.fn-test.com/content/uploads/product/manuals/elisa/ER0960.pdf Accessed January 2, 2021.
Sinha AK. Colorimetric assay of catalase. Anal Biochem. 1972;47:389–94. https://doi.org/10.1016/0003-2697(72)90132-7.
Maehly AC. The assay of catalases and peroxidases. In Glick D (ed.) Methods of Biochemical Analysis. 1954:357–424. https://doi.org/10.1002/9780470110171.ch14.
Kakkar P, Das B, Viswanathan PN. A modified spectrophotometric assay of superoxide dismutase. Indian J Biochem Biophys. 1984;21(2):130–2. http://nopr.niscair.res.in/handle/123456789/19932
Weydert CJ, Cullen JJ. Measurement of superoxide dismutase, catalase and glutathione peroxidase in cultured cells and tissue. Nat Protoc. 2009;5:51–66. https://doi.org/10.1038/nprot.2009.197.
Castro D, Contreras LM, Kurz L, Wilkesman J. Detection of guaiacol peroxidase on electrophoretic gels. Methods Mol Biol. 2017;1626:199–204. https://doi.org/10.1007/978-1-4939-7111-4_18.
Devasagayam TPA, Boloor KK, Ramasarma T. Methods for estimating lipid peroxidation: an analysis of merits and demerits. Indian J Biochem Biophys. 2003;40:300–8. http://nopr.niscair.res.in/handle/123456789/3805
Allain CC, Poon LS, Chan CSG, Richmond W, Fu PC. Enzymatic determination of total serum cholesterol. Clin Chem. 1974;20:470–5. https://doi.org/10.1093/clinchem/20.4.470.
Reitman S, Frankel S. A colorimetric method for the determination of serum glutamic oxalacetic and glutamic pyruvic transaminases. Am J ClinPathol. 1957;28:56–63. https://doi.org/10.1093/ajcp/28.1.56.
Slaoui M, Fiette L. Histopathology procedures: from tissue sampling to histopathological evaluation. Methods Mol Biol. 2010;691:69–82. https://doi.org/10.1007/978-1-60761-849-2_4.
Sokal RR, Rohle FJ. Introduction to analysis of variance. New York: Biometry. WH Freeman and Company; 1997. p. 179–206.
Ali J, Ansari S, Kotta S. Exploring scientifically proven herbal aphrodisiacs. Pharmacogn Rev. 2013;7:1–10. https://doi.org/10.4103/0973-7847.112832.
Wakelin SH. In: Wakelin SH, Maibach HI, Archer CB, editors. Systemic drug treatment in dermatology: a handbook. Manson Publishing; 2002.
Jameson JL, De Groot LJ. Endocrinology: adult and pediatric e-book. 7th ed. Saunders; 2015.
Moltz L, Römmler A, Post K, Schwartz U, Hammerstein J. Medium dose cyproterone acetate (CPA): effects on hormone secretion and on spermatogenesis in men. Contraception. 1980;21:393–413. https://doi.org/10.1016/s0010-7824(80)80017-5.
Turcu A, Smith JM, Auchus R, Rainey WE. Adrenal androgens and androgen precursors—definition, synthesis, regulation and physiologic actions. Compr Physiol. 2014;4:1369–81. https://doi.org/10.1002/cphy.c140006.
Ramaswamy S, Weinbauer GF. Endocrine control of spermatogenesis: role of FSH and LH/ testosterone. Spermatogenesis. 2014;4:e996025. https://doi.org/10.1080/21565562.2014.996025.
Malarvizhi D, Mathur PP. Effects of cisplatin on testicular functions in rats. Indian J Exp Biol. 1996;34:995–8. https://europepmc.org/article/med/9055653
Mallick C, Bera TK, Ali KM, Chatterjee K, Ghosh D. Diabetes-induced testicular disorders vis-a-vis germ cell apoptosis in albino rat: remedial effect of hexane fraction of root of Musa paradisiaca and leaf of Coccinia indica. J Health Sci. 2010;56:641–54. https://doi.org/10.1248/jhs.56.641.
Ahmed M, Al-Daghri N, Alokail M, Hussain T. Potential changes in rat spermatogenesis and sperm parameters after inhalation of Boswellia papyrifera and Boswellia carterii incense. Int J Environ Res Public Health. 2013;10:830–44. https://doi.org/10.3390/ijerph10030830.
Ray S, Chatterjee K, De D, Ghosh D. Bioefficacy of hydromethanolic extract of tuber of Chlorophytum borivilianum (Safed Musli) for the management of male infertility in cyproterone acetate-treated albino rats. Andrologia. 2013;46:659–71. https://doi.org/10.1111/and.12133.
Shpakov AO, Ryzhov JR, Bakhtyukov AA, Derkach KV. The regulation of the male hypothalamic-pituitary-gonadal axis and testosterone production by adipokines. AdvTesto Act. 2018:25–57. https://doi.org/10.5772/intechopen.76321.
Jana K, Jana N, De DK, Guha SK. Ethanol induces mouse spermatogenic cell apoptosis in vivo through over-expression of Fas/Fas-L, p53, and caspase-3 along with cytochrome c translocation and glutathione depletion. Mol Reprod Dev. 2010;77:820–33. https://doi.org/10.1002/mrd.21227.
Dare BJ, Chukwu RO, Oyewopo O, et al. Histological integrity of the testis of adult Wistar rats (Rattus novergicus) treated with Garcinia kola. Reprod Syst Sex Disord. 2012(1):4. https://doi.org/10.4172/2161-038x.1000113.
Acknowledgements
We are thankful to Vidyasagar University authority for providing us research facilities to conduct this work.
Author information
Authors and Affiliations
Contributions
CG: Investigation; data curation; formal analysis; software; writing, original draft; writing, review and editing.
RM: Data curation; formal analysis; writing—review and editing.
AR: Data curation; formal analysis; writing—review and editing.
CM: Conceptualization; data curation; investigation; methodology; software; supervision; writing—review and editing.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ghosh, C., Maity, R., Roy, A. et al. Dose-Dependent Protective Effect of Hygrophila auriculata Seeds on Cyproterone Acetate-Induced Testicular Dysfunction. Reprod. Sci. 30, 3359–3371 (2023). https://doi.org/10.1007/s43032-023-01279-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-023-01279-9