Abstract
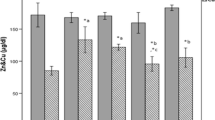
Zinc exerts a wide range of important biological roles. The present study was carried out to investigate the effects of zinc threoninate chelate in blood glucose levels, lipid peroxidation, activities of antioxidant defense systems and nitrite concentration, and histology of the pancreas in diabetic rats. Wistar rats were intravenously injected with a single dose of streptozotocin to induce diabetes. Then, diabetic rats were administrated orally with zinc threoninate chelate (3, 6, and 9 mg/kg body weight) once daily for 7 weeks. Fasting blood glucose was monitored weekly. At the end of the experimental period, the diabetic rats were killed, and levels of serum insulin, malondialdehyde, and nitric oxide, activities of glutathione peroxidase, total superoxide dismutase, copper/zinc-superoxide dismutase, and nitric oxide synthase were determined; pancreas was examined histopathologically as well. Zinc threoninate chelate significantly reduced the blood glucose levels and significantly increased the serum insulin levels in diabetic rats. In addition, zinc threoninate chelate caused a significant increase in activities of antioxidant enzymes and significant decrease in nitrite concentration and malondialdehyde formation in the pancreas and serum of diabetic rats. These biochemical observations were supplemented by histopathological examination of the pancreas. These results suggested that the antidiabetic effect of zinc threoninate chelate may be related to its antioxidative stress ability in diabetic rats.


Similar content being viewed by others
Abbreviations
- ZTC:
-
Zinc threoninate chelate
- STZ:
-
Streptozotocin
- Cu/Zn-SOD:
-
Copper/zinc-superoxide dismutase
- FBG:
-
Fasting blood glucose
- GPx:
-
Glutathione peroxidase
- MDA:
-
Malondialdehyde
- NOS:
-
Nitric oxide synthase
- SOD:
-
Superoxide dismutase
References
Jemai H, El Feki A, Sayadi S (2009) Antidiabetic and antioxidant effects of hydroxytyrosol and oleuropein from olive leaves in alloxan-diabetic rats. J Agri Food Chem 57:8798–8804
Li WL, Zheng HC, Bukuru J, De Kimpe N (2004) Natural medicines used in the traditional Chinese medical system for therapy of diabetes mellitus. J Ethnopharmacol 92:1–21
Nazıroğlu M, Dikici D, Dursun Ş (2012) Role of oxidative stress and Ca2+ signaling on molecular pathways of neuropathic pain in diabetes: focus on TRP channels. Neurochem Res 37:2065–2075. doi:10.1007/s11064-012-0850-x
Nakhaee A, Bokaeian M, Saravani M, Farhangi A, Akbarzadeh A (2009) Attenuation of oxidative stress in streptozotocin-induced diabetic rats by Eucalyptus globulus. Indian J Clin Biochem 24:419–425
Maritim AC, Sanders RA, Watkins JB III (2003) Diabetes, oxidative stress, and antioxidants: a review. J Biochem Mol Toxicol 17:24–38
Fatmi W, Kechrid Z, Nazıroğlu M, Flores-Arce M (2013) Selenium supplementation modulates zinc levels and antioxidant values in blood and tissues of diabetic rats fed zinc-deficient diet. Biol Trace Elem Res 152:243–250
Scott DA (1934) CCXI. Crystalline insulin. Biochem J 8:1592–1602
Beletate V, El Dib R, Atallah A (2007) Zinc supplementation for the prevention of type 2 diabetes mellitus. Cochrane Database Syst Rev 1: Art. No
Marreiro DN, Geloneze B, Tambascia MA, Lerário AC, Halpern A, Cozzolino SMF (2006) Effect of zinc supplementation on serum leptin levels and insulin resistance of obese women. Biol Trace Elem Res 112:109–118
Özcelik D, Nazıroğlu M, Tunçdemir M, Çelik Ö, Öztürk M, Flores-Arce M (2012) Zinc supplementation attenuates metallothionein and oxidative stress changes in kidney of streptozotocin-induced diabetic rats. Biol Trace Elem Res 150:342–349
Rojas L, McDowell L, Martin F, Wilkinson N, Johnson A, Njeru C (1996) Relative bioavailability of zinc methionine and two inorganic zinc sources fed to cattle. J Trace Elem Med Bio 10:205–209
Chien XX, Zafra-Stone S, Bagchi M, Bagchi D (2006) Bioavailability, antioxidant and immune-enhancing properties of zinc methionine. Biofactors 27:231–244
Lönnerdal B (2000) Dietary factors influencing zinc absorption. J Nutr 130:1378S–1383S
Hu XB, Gong Y, Li L, Nie SP, Wang YX, Xie MY (2010) Safety evaluation of zinc threoninate chelate. Int J Toxicol 29:372–379
Hu XB, Qiao LN, Gong Y, Wang YX, Nie SP, Xie MY (2012) Synthesis and characterization of zinc threoninate. Sci Tech Food Industry 24:355–357, (In Chinese)
Gong D, Lu J, Chen X, Choong SY, Zhang S, Chan YK, Glyn-Jones S, Gamble GD, Phillips AR, Cooper GJ (2006) Molecular changes evoked by triethylenetetramine treatment in the extracellular matrix of the heart and aorta in diabetic rats. Mol Pharmacol 70:2045–2051
Zhu KX, Nie SP, Li C, Lin SL, Xing MM, Li WJ, Gong D, Xie MY (2013) A newly identified polysaccharide from Ganoderma atrum attenuates hyperglycemia and hyperlipidemia. Int J Biol Macromol 57:142–150
Adachi Y, Yoshikawa Y, Sakurai H (2007) Antidiabetic zinc (II)-N-acetyl-L-cysteine complex: evaluations of in vitro insulinomimetic and in vivo blood glucose-lowering activities. Biofactors 29:213–223
Tang Y, Yang Q, Lu J, Zhang X, Suen D, Tan Y, Jin L, Xiao J, Xie R, Rane M (2010) Zinc supplementation partially prevents renal pathological changes in diabetic rats. J Nutr Biochem 21:237–246
Paglia DE, Valentine WN (1967) Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J Lab Clin Med 70:158–169
Zafari AM, Ushio-Fukai M, Akers M, Yin Q, Shah A, Harrison DG, Taylor WR, Griendling KK (1998) Role of NADH/NADPH oxidase-derived H2O2 in angiotensin II-induced vascular hypertrophy. Hypertension 32:488–495
Bredt DS, Snyder SH (1990) Isolation of nitric oxide synthetase, a calmodulin-requiring enzyme. Proc Natl Acad Sci USA 87:682–685
Gavino V, Miller J, Ikharebha S, Milo G, Cornwell D (1981) Effect of polyunsaturated fatty acids and antioxidants on lipid peroxidation in tissue cultures. J Lipid Res 22:763–769
Turgut M, Erdogan S, Ergin K, Serter M (2007) Melatonin ameliorates blood–brain barrier permeability, glutathione, and nitric oxide levels in the choroid plexus of the infantile rats with kaolin-induced hydrocephalus. Brain Res 1175:117–125
Sivakumar S, Subramanian SP (2009) Pancreatic tissue protective nature of D-Pinitol studied in streptozotocin-mediated oxidative stress in experimental diabetic rats. Eur J Pharmacol 622:65–70
Wang JL, Zhang J, Zhao BT, Wu YQ, Wang C, Wang YP (2010) Structural features and hypoglycaemic effects of Cynomorium songaricum polysaccharides on STZ-induced rats. Food Chem 120:443–451
Ohkuwa T, Sato Y, Naoi M (1995) Hydroxyl radical formation in diabetic rats induced by streptozotocin. Life Sci 56:1789–1798
Bolkent S, Yanardag R, Mutlu O (2009) The influence of zinc supplementation on the pancreas of streptozotocin-diabetic rats. Dig Dis Sci 54:2583–2587
Li C, Zhu KX, Nie SP, Hu XB, Huang DF, Xie MY (2011) Protective effect of zinc threonine against liver injury in diabetic rats. Food Sci 32:198–200 (In Chinses)
Salgueiro MJ, Krebs N, Zubillaga MB, Weill R, Postaire E, Lysionek AE, Caro RA, De Paoli T, Hager A, Boccio J (2001) Zinc and diabetes mellitus. Biol Trace Elem Res 81:215–228
Tobia MH, Zdanowicz MM, Wingertzahn MA, McHeffey-Atkinson B, Slonim AE, Wapnir RA (1998) The role of dietary zinc in modifying the onset and severity of spontaneous diabetes in the BB Wistar rat. Mol Genet Metab 63:205–213
Mustafa AG, Nawfal NA, Saad HA (2006) Effects of treatment with zinc sulphate on the oxidative stress state during chronic exposure to lead in humans. AJPS 3:16–21
Naziroğlu M, Çay M (2001) Protective role of intraperitoneally administered vitamin E and selenium on the antioxidative defense mechanisms in rats with diabetes induced by streptozotocin. Biol Trace Elem Res 79:149–159
Duzguner V, Kaya S (2007) Effect of zinc on the lipid peroxidation and the antioxidant defense systems of the alloxan-induced diabetic rabbits. Free Radical Bio Med 42:1481–1486
Ugochukwu NH, Figgers CL (2007) Attenuation of plasma dyslipidemia and oxidative damage by dietary caloric restriction in streptozotocin-induced diabetic rats. Chem-Biol Interact 169:32–41
Gunasekara P, Hettiarachchi M, Liyanage C, Lekamwasam S (2011) Effects of zinc and multimineral vitamin supplementation on glycemic and lipid control in adult diabetes. Diabetes Metab Syndr Obes 4:53–60
Corbett JA, McDaniel ML (1992) Does nitric oxide mediate autoimmune destruction of β-cells?: possible therapeutic interventions in IDDM. Diabetes 41:897–903
Tanaka Y, Shimizu H, Sato N, Mori M, Shimomura Y (1995) Involvement of spontaneous nitric oxide production in the diabetogenic action of streptozotocin. Pharmacology 50:69–73
Shen H, Oesterling E, Stromberg A, Toborek M, MacDonald R, Hennig B (2008) Zinc deficiency induces vascular pro-inflammatory parameters associated with NF-κB and PPAR Signaling. J Am Coll Nutr 27:577–587
Gandhi GR, Ignacimuthu S, Paulraj MG (2011) Solanum torvum Swartz. fruit containing phenolic compounds shows antidiabetic and antioxidant effects in streptozotocin induced diabetic rats. Food Chem Toxicol 49:2725–2733
Acknowledgment
This study was supported by the National Nature Science Foundation of China (31071532), the Program for New Century Excellent Talents in University (NCET-12-0749), and Training Project of Young Scientists of Jiangxi Province (Stars of Jinggang). We appreciate the help of the Fourth Affiliated Hospital, Nanchang University with serum insulin assay.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Zhu, K., Nie, S., Li, C. et al. Antidiabetic and Pancreas-Protective Effects of Zinc Threoninate Chelate in Diabetic Rats may be Associated with its Antioxidative Stress Ability. Biol Trace Elem Res 153, 291–298 (2013). https://doi.org/10.1007/s12011-013-9675-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-013-9675-y