Abstract
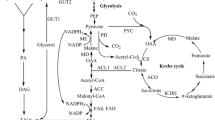
The growth of wild type strain Yarrowia lipolytica VKM Y-2373 and its mutant Y. lipolytica NG40/UV7 as well as the biosynthesis of citric and isocitric acid on rapeseed oil were studied. It was indicated that the initial step of assimilation of rapeseed oil in the yeast Y. lipolytica is its hydrolysis by extracellular lipases with the formation of glycerol and fatty acids, which appear in the medium in the phase of active growth. The concentrations of these metabolites were changed insignificantly upon further cultivation. Lipase and the key enzymes of glycerol metabolism (glycerol kinase) and the glyoxylate cycle responsible for the metabolism of fatty acids (isocitrate lyase and malate synthase) are induced just at the beginning of the growth phase and remain active in the course of further cultivation. These results, taken together, suggest that glycerol and fatty acids available in the medium do not suppress the metabolism of each other. Citric acid production and a ratio between citric and isocitric acids depended on the strain used. It was revealed that the wild strain produced almost equal amounts of citric and isocitric acids while the mutant produced only citric acid (175 g/L) with a yield of 1.5 g of CA per g of oil.






Similar content being viewed by others
References
Anastassiadis S, Morgunov IG, Kamzolova SV, Finogenova TV (2008) Citric acid production patent review. Recent Pat Biotechnol 2:107–123
Papanikolaou S, Galiotou M, Blanchard F, Komaitis M, Marc I, Aggelis G (2006) Influence of glucose and saturated free-fatty acid mixtures on citric acid and lipid production by Yarrowia lipolytica. Curr Microbiol 52:134–139
Rymowicz W, Rywińska A, Żarowska B, Juszczyk P (2006) Citric acid production from raw glycerol by acetate mutants of Yarrowia lipolytica. Chem Pap 60:391–395. doi:10.2478/s11696-006-0071
Papanikolaou S, Aggelis G (2010) Yarrowia lipolytica: a model microorganism used for the production of tailor-made lipids. Eur J Lipid Sci Technol 112:639–654
Zvyagintseva IS (1971) Lipase activity of some yeasts. Microbiology (Moscow) 1:16–20
Ota Y, Oikawa S, Morimoto Y, Minoda Y (1982) Purification and some properties of cell-bound lipase from Saccharomycopsis lipolytica. Agric Biol Chem 46:2885–2893
Pignede G, Wang HJ, Fidalej F, Seman M, Gailaardin C, Nicaud JM (2000) Autocloning and amplification of LIP2 in Yarrowia lipolytica. Appl Environ Microbiol 66:3283–3289
Kamzolova SV, Morgunov IG, Aurich A, Perevoznikova OA, Shishkanova NV, Stottmeister U, Finogenova TV (2005) Lipase secretion and citric acid production in Yarrowia lipolytica yeast grown on animal and vegetable fat. Food Technol Biotechnol 43(2):113–122
Förster A, Jacobs K, Juretzek J, Mauersberger S, Barth G (2007) Overexpression of the ICL1 gene changes the product ration of citric acid production by Yarrowia lipolytica. Appl Microbiol Biotechnol 77(4):681–689
Gandhi NN (1997) Applications of lipase. J Am Oil Chem Soc 74:621–634
Lodder J (1970) The yeast. A taxonomic study. North Holland, Amsterdam
Aloulou A, Rodriguez JA, Puccinelli D, Mouz N, Leclaire J, Leblond Y, Carrière F (2007) Purification and biochemical characterization of the LIP2 lipase from Yarrowia lipolytica. Biochim Biophys Acta 1771:228–237
Desfougères T, Haddouche R, Fudalej F, Neuvéglise C, Nicaud JM (2010) SOA genes encode proteins controlling lipase expression in response to triacylglycerol utilization in the yeast Yarrowia lipolytica. FEMS Yeast Res 10:93–103
Burkholder PR, McVeigh J, Moyer D (1944) Studies on some growth factors of yeasts. J Bacteriol 48:385–391
Bradford MM (1976) Rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Ermakova IT, Morgunov IG (1987) Pathways of glycerol metabolism in Yarrowia (Candida) lipolytica yeasts. Mikrobiologiya 57:533–537
Makri A, Fakas S, Aggelis G (2010) Metabolic activities of biotechnological interest in Yarrowia lipolytica grown on glycerol in repeated batch cultures. Bioresour Technol 101(7):2351–2358
Lozinov AB, Glazunova LM, Ermakova IT (1976) Activity of enzymes of the citrate glyoxylate and pentose phosphate cycles during growth of yeasts on hexadecane and glucose. Microbiology (Moscow) 45:33–40
Whitworth DA, Ratledge C (1975) An analysis of intermediary metabolism and its control in a fat-synthesizing yeast (Candida 107) growing on glucose or alkanes. J Gen Microbiol 88:275–288
Rohr M, Kubicek CP (1981) Regulatory aspects of citric acid fermentation by Aspergillus niger. Process Biochem 16:34–37
Evans CT, Ratledge C (1985) The physiological significance of citric acid in the control of metabolism in lipid-accumulating yeasts. Biotechnol Genet Eng Rev 3:85–111
Holdsworth JE, Veenhuis H, Ratledge C (1988) Enzyme activities in oleaginous yeasts accumulating and utilizing exogenous or endogenous lipids. J Gen Microbiol 134:2907–2915
Bati N, Hammond EG, Glatz BA (1984) Biomodification of fats and oils: trials with Candida lipolytica. J Am Oil Chem Soc 61:1743–1746
Glatz BA, Hammond EG, Hsu KH, Baehman L, Bati N, Bednarski W, Brown D, Floetenmeyer M (1984) Production and modification of fats and oils by yeast fermentation. In: Ratledge C, Rattray JBM, Dawson PSS (eds) Biotechnology for the oils and fats industry. AOCS Press, Champaign, pp 163–176
Koritala S, Hesseltine CW, Pryde EH, Mounts TL (1987) Biochemical modification of fats by microorganisms: a preliminary study. J Am Oil Chem Soc 64:509–513
Papanikolaou S, Chevalot I, Komaitis M, Aggelis G, Marc I (2001) Kinetic profile of the cellular lipid composition in an oleaginous Yarrowia lipolytica capable of producing a cocoa-butter substitute from industrial fats. Antonie van Leeuwenhoek 80:215–224
Del Rio JL, Serra P, Valero F, Poch M, Sola C (1990) Reaction scheme of lipase production by Candida rugosa growing on olive oil. Biotechnol Lett 12:835–838
Kamzolova SV, Morgunov IG, Finogenova TV (2008) Microbiological production of citric and isocitric acid from sunflower oil. Food Technol Biotechnol 46:51–59
Entian KD, Shuller HJ (1997) Glucose repression (catabolite repression) in yeast. In: Zimmerman FK, Entian KD (eds) Yeast sugar metabolism, biochemistry, genetics, biotechnology and applications. Technomoc Publishing, Basel, pp 409–434
Gancedo JM (1998) Yeast carbon catabolite repression. Microbiol Mol Biol Rev 62:334–361
Cho IH, Lu ZR, Yu JR, Park YD, Yang JM, Hahn MJ, Zou F (2009) Towards profiling the gene expression of tyrosinase-induced melanogenesis in HEK293 cells: a functional DNA chip microarray and interactomics studies. J Biomol Struct Dyn 27:331–345
Papanikolaou S, Muniglia L, Chevalot I, Aggelis G, Marc I (2003) Accumulation of a cocoa-butter-like lipid by Yarrowia lipolytica cultivated on agro-industrial residues. Curr Microbiol 46:124–130
Iske U, Gwenner Ch, Bullmann M, Uhlenhut G-J, Schindler G (1983) Zur Kinetik der Mischsubstratutilisation bei der Citronensäure-akkumulation durch Saccharomycopsis lipolytica EH 59. Acta Biotechnol 3:143–153
Behrens U, Weissbrodt E, Lehmann W (1978) Kinetics of citric acid production in Candida lipolytica. Z Allg Mikrobiol 18:549–558 (in German)
Franke-Rinker D, Behrens U, Nöckel E, Forner C, Portnowa A (1983) Joint utilization of glucose and n-alkanes in citric acid synthesis by Saccharomycopsis lipolytica. Z Allg Mikrobiol 23:9–16 (in German)
Stottmeister U, Weissbrodt E (1991) Product formation by Yarrowia lipolytica—some generalizing aspects. In: Metabolism alkanow i cwerchsintes produktow mikroorganismami. Sbornik naushnich trudow, Pushino, pp 147–158
Babel W (1990) The mixed substrate concept, applied for microbial syntheses of metabolites. Biotech Adv 8:261–275
Babel W (2009) The auxiliary substrate concept: from simple considerations to heuristically valuable knowledge. Eng Life Sci 9:285–290
Heretsch P, Thomas F, Aurich A, Krautscheid H, Sicker D, Giannis A (2008) Syntheses with a chiral building block from the citric acid cycle: (2R,3S)-isocitric acid by fermentation of sunflower oil. Angew Chem Int Ed Engl 47:1958–1960
Good DW, Droniuk R, Lawford RG, Fein JE (1985) Isolation and characterization of a Saccharomycopsis lipolytica mutant showing increased production of citric acid from canola oil. Can J Microbiol 31:436–440
Aurich A, Förster A, Mauesberger S, Barth G, Stottmeister U (2003) Citric acid production from renewable resources by Yarrowia lipolytica. Biotechn Adv 21:454–455
Wojtatowicz M, Rymowicz W, Kautola H (1991) Comparison of different strains of the yeast Yarrowia lipolytica for citric acid production from glucose hydrol. Appl Biochem Biotechnol 31:165–174
Crolla A, Kennedy KJ (2004) Fed-batch production of citric acid by Candida lipolytica grown on n-paraffin. J Biotechnol 110:73–84
Rywińska A, Rymowicz W (2010) High-yield production of citric acid by Yarrowia lipolytica on glycerol in repeated-batch bioreactors. J Ind Microbiol Biotechnol 37:431–435
Morgunov IG, Kamzolova SV, Sokolov AP, Finogenova TV (2004) The isolation, purification, and some properties of NAD-dependent isocitrate dehydrogenase from the organic acid-producing yeast Yarrowia lipolytica. Microbiology (Moscow) 73:249–254
Meers JL, Milsom PE (1987) Organic acids and amino acids. In: Bu’Lock J, Kristiansen B (eds) Basic biotechnology. Academic Press, Orlando, pp 359–383
Acknowledgments
The authors are grateful to Dr Irina F. Puntus (Institute of Biochemistry and Physiology of Microorganisms, Russian Academy of Sciences) for her help in obtaining of mutant Y. lipolytica NG40/UV7.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Kamzolova, S.V., Lunina, J.N. & Morgunov, I.G. Biochemistry of Citric Acid Production from Rapeseed Oil by Yarrowia lipolytica Yeast. J Am Oil Chem Soc 88, 1965–1976 (2011). https://doi.org/10.1007/s11746-011-1954-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11746-011-1954-1