Abstract
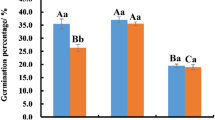
Researches have reported that reactive oxygen species (ROS)-induced oxidative stress plays an important role in cell cryodamage during cryopreservation. In the current study, pollen from Magnolia denudata and Paeonia lactiflora ‘Zi Feng Chao Yang’ was cryopreserved and incubated with exogenous catalase (CAT) and malate dehydrogenase (MDH) immediately after thawing. The effect of CAT and MDH on the germination of cryopreserved pollen was measured. Based on that, the ROS level, lipid peroxidation and antioxidants activities in fresh pollen, cryopreserved pollen added with or without CAT or MDH were determined to investigate their relationship with oxidative stress. Pollen from Magnolia and Paeonia showed a significant loss of germination, but marked increase of ROS and malondialdehyde (MDA) production after cryostorage. Antioxidant profiles in them were also enhanced. CAT and MDH addition increased the post-LN pollen germination of Magnolia and Paeonia significantly. Their germination rate achieved the highest with 100 IU ml−1 MDH and 400 IU ml−1 CAT application, respectively. Compared to their untreated controls, ROS and MDA accumulation reduced significantly in cryopreserved Magnolia pollen treated with 100 IU ml−1 MDH, while superoxide dismutase (SOD) activity improved markedly. In the case of Paeonia, significantly lower level of ROS and MDA, but higher activity of CAT and SOD were observed in cryopreserved pollen treated with 400 IU ml−1 CAT. In conclusion, pollen deterioration after cryopreservation is associated with ROS-induced oxidative stress. Exogenous CAT and MDH can reduce the oxidative damage through the activity stimulation of antioxidant enzymes, and play a protective role in the pollen during cryopreservation.




Similar content being viewed by others
References
Alba V, Bisignano V, Alba E, De Stradis A, Polignano GB (2011) Effects of cryopreservation on germinability of olive (Olea europaea L.) pollen. Genet Resour Crop Evol 58:977–982
Amini MR, Kohram H, Zare-Shahaneh A, Zhandi M, Sharideh H, Nabi MM (2015) The effects of different levels of catalase and superoxide dismutase in modified Beltsville extender on rooster post-thawed sperm quality. Cryobiology 70:226–232
Apel K, Hirt H (2004) Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol 55:373–399
Benson EE, Bremner D (2004) Oxidative stress in the frozen plant: a free radical point of view. In: Fuller BJ, Lane N, Benson EE (eds) Life in the frozen state. CRC Press, Boca Raton, pp 206–241
Benson EE, Withers LA (1987) Gas chromatographic analysis of volatile hydrocarbon production by cryopreserved plant tissue cultures: a nondestructive method for assessing stability. Cryoletters 8:35–46
Chen GQ, Ren L, Zhang J, Reed BM, Zhang D, Shen XH (2015) Cryopreservation affects ROS-induced oxidative stress and antioxidant response in Arabidopsis seedlings. Cryobiology 70:38–47
Connor KF, Towill LE (1993) Pollen-handling protocol and hydration/dehydration characteristics of pollen for application to long-term storage. Euphytica 68:77–84
Craddock JH, Reed SM, Schlarbaum SE, Sauve RJ (2000) Storage of flowering dogwood (Cornus florida L.) pollen. Hortscience 35:108–109
Day JG, Fleck RA, Benson EE (2000) Cryopreservation-recalcitrance in microalgae: novel approaches to identify and avoid cryo-injury. J Appl Phycol 12:369–377
Di W, Jia MX, Liu Y, Xu J, Li BL (2017) Exogenous CAT and PDH improve viability and affect oxidative stress of nobile-type Dendrobium protocorm-like bodies following cryopreservation. Cryoletters 38:228–238
Engelmann F (2004) Cryopreservation: progress and prospects. In Vitro Cell Dev Plant 40:427–433
Fang J, Wetten A, Johnston J (2008) Headspace volatile markers for sensitivity of cocoa (Theobroma cacao L.) somatic embryos to cryopreservation. Plant Cell Rep 27:453–461
Heber U (1974) Metabolic exchange between chloroplasts and cytoplasm. Ann Rev Plant Biol 25(25):393–421
Houdan A, Véron B, Claquin P, Lefebvre S, Poncet JM (2005) Cryopreservation of the coccolithophore, Emiliania huxleyi (Haptophyta, Prymnesiophyceae). J Appl Phycol 17:413–422
Jia MX, Di W, Liu Y, Shi Y, Xie YR (2016) ROS-induced oxidative stress in nobile-type Dendrobium protocorm-like bodies (PLBs) during vitrification. Cryoletters 37:253–263
Jia MX, Shi Y, Di W, Jiang XR, Xu J, Liu Y (2017) ROS-induced oxidative stress is closely related to pollen deterioration following cryopreservation. In Vitro Cell Dev Biol Plant 53:433–439
Johnston JW, Harding K, Benson E (2007) Antioxidant status and genotypic tolerance of Ribes in vitro cultures to cryopreservation. Plant Sci 172:524–534
Kaczmarczyk A, Funnekotter B, Menon A, Phang PY, Al-Hanbali A, Bunn E, Mancera RL (2012) Current issues in plant cryopreservation. In: Katkov II (ed) Current frontiers in cryobiology, under CC BY 3.0 license, In Tech, pp 417–438
Kampfenkel K, Montagu MV, Inzé D (1995) Extraction and determination of ascorbate and dehydroascorbate from plant tissue. Anal Biochem 225:165–167
Karun A, Sajini KK, Nair M, Kumaran PM, Samsudheen K (2006) Cryopreservation of coconut (Cocos nucifera. L) pollen. J Plant Crops 34:568–571
Li GQ (2005) Studies on cryopreservation of Camellia pollen. Dissertation, Beijing Forestry University (in Chinese)
Li BL (2010) Studies on differentially expressed protein of pollen cryopreservation and cryobank construction of Paeonia spp. Dissertation, Beijing Forestry University (in Chinese)
Li HS, Sun Q, Zhao SJ, Zhang WH (2000) Assay of malondialdehyde in plants. Experiment principle and technology of plant physiology and biochemistry. Higher Education Press, Beijing, pp 260–261 (in Chinese)
Li ZL, Lin QL, Liu RJ, Xiao WF, Liu WM (2010) Protective effects of ascorbate and catalase on human spermatozoa during cryopreservation. J Androl 31:437–444
Li BL, Wang H, Liu Y (2011) Pollen cryopreservation of Camellia. Acta Hortic 908:265–268
Luza JG, Polito VS (1985) In vitro germination and storage of English walnut pollen. Sci Hortic (Amsterdam) 27:303–316
Moubasher AE, El Din AME, Ali ME, El-sherif WT, Gaber HD (2013) Catalase improves motility, vitality and DNA integrity of cryopreserved human spermatozoa. Andrologia 45:135–139
Pandhair V, Sekhon BS (2006) Reactive oxygen species and antioxidants in plants: an overview. J Plant Biochem Biotechnol 15:71–78
Prochazkova D, Sairam RK, Srivastava GC, Singh DV (2001) Oxidative stress and antioxidant activity as the basis of senescence in maize leaves. Plant Sci 161:765–771
Rajasekharan PE, Rao TM, Janakiram T, Ganeshan S (1994) Freeze preservation of gladiolus pollen. Euphytica 80:105–109
Reed BM (2012) Are antioxidants a magic bullet for reducing oxidative stress during cryopreservation? Cryobiology 65(3):340
Roca J, Rodríguez MJ, Gil MA, Carvajal G, Garcia EM, Cuello C, Vazquez JM, Martinez EA (2005) Survival and in vitro fertility of boar spermatozoa frozen in the presence of superoxide dismutase and/or catalase. J Androl 26:15–24
Shafiei M, Forouzanfar M, Hosseini SM, Nasr Esfahani MH (2015) The effect of superoxide dismutase mimetic and catalase on the quality of postthawed goat semen. Theriogenology 83:1321–1327
Tomaz T, Bagard M, Pracharoenwattana I, Lindén P, Lee CP, Carroll AJ, Ströher E, Smith SM, Gardeström P, Millar AH (2010) Mitochondrial malate dehydrogenase lowers leaf respiration and alters photorespiration and plant growth in Arabidopsis. Plant Physiol 154:1143–1157
Uchendu EE, Leonard SW, Traber MG, Reed BM (2010) Vitamins C and E improve regrowth and reduce lipid peroxidation of blackberry shoot tips following cryopreservation. Plant Cell Rep 29:25–35
Varghese B, Naithani SC (2008) Oxidative metabolism-related changes in cryogenically stored neem (Azadirachta indica A. Juss) seeds. J Plant Physiol 165:755–765
Wang YR, Li P, Liu HX, Zeng SX, Chen DF, Guo JY (1985) The effect of low temperature on malate dehydrogenase in various organelles of cucumber seedling cotyledons with different cold tolerance. Acta Phytophysiologia Sinica 11:147–154
Wen B, Wang RL, Cheng HY, Song SQ (2010) Cytological and physiological changes in orthodox maize embryos during cryopreservation. Protoplasma 239:57–67
Whitaker C, Beckett RP, Minibayeva FV, Kranner I (2010) Production of reactive oxygen species in excised, desiccated and cryopreserved explants of Trichilia dregeana Sond. S Afr J Bot 76:112–118
Xi YZ (1984) The pollen morphology and exine ultrastructure of Paeonia L. China. Acta Botanica Sinica 26(3):241–246 (in Chinese)
Xu J (2014) A study on the mechanism of Magnolia denudata pollen cryopreservation. Dissertation, Beijing Forestry University (in Chinese)
Xu J, Li BL, Liu Q, Shi Y, Peng JG, Jia MX, Liu Y (2014a) Wide-scale pollen banking of ornamental plants through cryopreservation. Cryoletters 35:312–319
Xu J, Liu Q, Jia MX, Liu Y, Li BL, Shi Y (2014b) Generation of reactive oxygen species during cryopreservation may improve Lilium × siberia pollen viability. In Vitro Cell Dev Biol Plant 50:369–375
Xu J, Liu Y, Li B, Wang Z, Liu Q, Shi Y (2017) Effects of catalase and malate dehydrogenase on cryopreservation of Euonymus fortunei. Propag Ornam Plants 17:20–28
Zhang YL, Chen RD, Huang CJ, Liu Y (2009) Cryo-banking of Prunus mume pollen and its application in cross-breeding. Cryoletters 30:165–170
Zhang D, Ren L, Chen GQ, Zhang J, Reed BM, Shen XH (2015) ROS-induced oxidative stress and apoptosis-like event directly affect the cell viability of cryopreserved embryogenic callus in Agapanthus praecox. Plant Cell Rep 34:1499–1513
Zhao Y, Qi LW, Wang WM, Saxena PK, Liu CZ (2011) Melatonin improves the survival of cryopreserved callus of Rhodiola crenulata. J Pineal Res 50:83–88
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No. 31370693).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by M. Lambardi.
Rights and permissions
About this article
Cite this article
Jia, M.X., Jiang, X.R., Xu, J. et al. CAT and MDH improve the germination and alleviate the oxidative stress of cryopreserved Paeonia and Magnolia pollen. Acta Physiol Plant 40, 37 (2018). https://doi.org/10.1007/s11738-018-2612-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11738-018-2612-0