Abstract
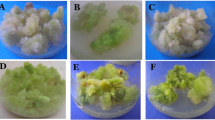
With the current trends in high density plantations of fruit trees, numerous clonal rootstocks of apple have been developed through various breeding programs. Among them, Merton 793 is the most popular in India because of the desirable traits of vigorous growth and resistance to woolly apple aphid and collar rot. The planting material of this rootstock cannot be multiplied at a desirable rate by means of conventional vegetative propagation methods, so micropropagation techniques are being explored to augment scarce planting material. Large number of plants can be produced in vitro under aseptic conditions, but there is always a danger of producing somaclonal variants by tissue culture technology. Thus, it is advisable to check the clonal fidelity of in vitro raised plants, especially of perennials prior to their field transplantation. The genetic stability of in vitro raised plants of apple rootstock Merton 793, multiplied through enhanced axillary bud proliferation up to 22 subculture passages, was tested by intersimple sequence repeat (ISSR) assay. Of 24 ISSR primers screened, 15 primers produced clear reproducible bands, resulting in a total of 134 distinct bands with an average of 8.9 bands per primer. Apple rootstock MM 111 and scion Jonathan, taken as outliers with tissue culture-raised progenies of Merton 793, ruled out the possibility that the invariant banding pattern occurred because of inefficiency of ISSR primers in detecting variations. The homogenous amplification profile observed for all the micropropagated plants compared to the donor plant confirmed the clonal fidelity of the tissue culture-raised Merton 793 plants. This suggests that axillary bud multiplication is the safest mode for multiplication of true-to-type plants. This is the first study that evaluates the applicability of ISSR markers in establishing clonal fidelity of tissue culture-raised apple plants.


Similar content being viewed by others
References
Agarwal M.; Shrivastava N.; Padh H. Advances in molecular marker techniques and their applications in plant sciences. Plant Cell Rep. 27: 617–631; 2008.
Alizadeh M.; Singh S. K. Molecular assessment of clonal fidelity in micropropagated grape (Vitis spp.) rootstock genotypes using RAPD and ISSR markers. Iranian J. Biotech. 7: 37–44; 2009.
Bhatia R.; Singh K. P.; Jhang T.; Sharma T. R. Assessment of clonal fidelity of micropropagated gerbera plants by ISSR markers. Sci. Hortic. 119: 208–211; 2009.
Bindiya K.; Kanwar K. Random amplified polymorphic DNA (RAPDs) markers for genetic analysis in micropropagated plants of Robinia pseudoacacia L. Euphytica 132: 41–47; 2003.
Bopana N.; Saxena S. In vitro regeneration of clonally uniform plants of Crataeva magna: a high value medicinal tree by axillary branching method. New. For. 38: 53–65; 2009.
Caboni E.; Lauri P.; Damiano C.; D’Angeli S. Somaclonal variation induced by adventitious shoot regeneration in pear and apple. Acta Hortic. 530: 195–202; 2000.
Chowdari K. V.; Ramakrishna W.; Tamhankar S. A.; Hendre R. R.; Gupta V. S.; Sahasrabudhe N. A.; Ranjekar P. K. Identification of minor DNA variations in rice somaclonal variants. Plant Cell Rep. 18: 55–58; 1998.
Chuang S. J.; Chen C. L.; Chen J. J.; Chou W. Y.; Sung J. M. Detection of somaclonal variation in micro-propagated Echinacea purpurea using AFLP marker. Sci. Hortic. 120: 121–126; 2009.
Devarumath R. M.; Nandy S.; Rani V.; Marimuthu S.; Muraleedharan N.; Raina S. N. RAPD, ISSR and RFLP fingerprints as useful markers to evaluate genetic integrity of micropropagated plants of three diploid and triploid elite tea clones representing Camellia sinensis (China type) and C. asamica ssp. Assamica (Assam-India type). Plant Cell Rep. 21: 166–173; 2002.
Doyle J. J.; Doyle J. L. Isolation of plant DNA from fresh tissue. Focus 12: 13–15; 1990.
Fang D. Q.; Roose M. L. Identification of closely related citrus cultivars with inter-simple sequence repeat markers. Theor. Appl. Genet. 95: 408–417; 1997.
Grandbastien M. A.; Spielman A.; Caboche M. Tnt1, a mobile retroviral-like transposable element of tobacco isolated by plant cell genetics. Nature 337: 376–380; 1989.
Harris S. A.; Robinson J. P.; Juniper B. E. Genetic clues to the origin of the apple. Trends Genet. 18: 426–430; 2002.
Hirochika H.; Sugimoto K.; Otsuki Y.; Tsugawa H.; Kanda M. Retrotransposons of rice involved in mutations induced by tissue culture. Proc. Natl. Acad. Sci. USA. 93: 7783–7788; 1996.
Huang W. J.; Ning G. G.; Liu G. F.; Bao M. Z. Determination of genetic stability of long-term micropropagated plantlets of Platanus acerifolia using ISSR markers. Biol. Plant. 53: 159–163; 2009.
Jaligot E.; Beulé T.; Rival A. Methylation-sensitive RFLPs: characterisation of two oil palm markers showing somaclonal variation-associated polymorphism. Theor. Appl. Genet. 104: 1263–1269; 2002.
Jones O. P. Propagation of apple in vitro. In: Ahuja M. R. (ed) Micropropagation of woody plants. Kluwer Academic, Dordrecht, pp 169–186; 1993.
Joshi P.; Dhawan V. Assessment of genetic fidelity of micropropagated Swertia chirayita Plantlets by ISSR marker assay. Biol. Plant. 51: 22–26; 2007.
Karp A.; Seberg O. L. E.; Buiatti M. Molecular techniques in the assessment of botanical diversity. Ann. Bot. 78: 143–149; 1996.
Kojima T.; Nagaoka T.; Noda N.; Ogihara Y. Genetic linkage map of ISSR and RAPD markers in Einkorn wheat in relation to that of RFLP markers. Theor. Appl. Genet. 96: 37–45; 1998.
Korbin M.; Kuras A.; Zurawicz E. Fruit plant germplasm characterization using molecular markers generated in RAPD and ISSR-PCR. Cell. Mol. Biol. Lett. 7: 785–794; 2002.
Larkin P. J.; Scrowcroft W. Somaclonal variation—a novel source of variability from cell cultures for plant improvement. Theor. Appl. Genet. 60: 197–214; 1981.
Lattoo S. K.; Bamotra S.; Sapru D. R.; Khan S.; Dha A. K. Rapid plant regeneration and analysis of genetic fidelity of Chlorophytum arundinaceum Baker—an endangered medicinal herb. Plant Cell Rep. 25: 499–506; 2006.
Leroy X. J.; Leon K.; Charles G.; Branchard M. Cauliflower somatic embryogenesis and analysis of regenerant stability by ISSRs. Plant Cell Rep. 19: 1102–1107; 2000.
Martin K. P.; Pachathudikandi S.; Zhang C. L.; Slater A.; Madassery J. RAPD analysis of a variant of banana (Musa sp.) cv. Grande naine and its propagation via shoot tip culture. In Vitro Cell. Dev. Biol. Plant. 42: 188–192; 2006.
Martins M.; Sarmento D.; Oliveira M. M. Genetic stability of micropropagated almond plantlets as assessed by RAPD and ISSR markers. Plant Cell Rep. 23: 492–496; 2004.
Modgil M.; Mahajan K.; Chakrabarti S. K.; Sharma D. R.; Sobti R. C. Molecular analysis of genetic stability in micropropagated apple rootstock MM 106. Sci. Hortic. 104: 151–210; 2005.
Mohan J. S. Tissue culture-derived variation in crop improvement. Euphytica 118: 153–166; 2001.
Montecelli S.; Gentile A.; Damino C. In vitro shoot regeneration of apple cultivar Gala. Acta Hortic. 530: 219–234; 2000.
Murashige T.; Skoog F. A revised medium for rapid growth and bioassay with tobacco tissue cultures. Physiol. Plant. 15: 473–497; 1962.
Nagaoka T.; Ogihara Y. Applicability of inter-simple sequence repeat polymorphisms in wheat for use as DNA markers in comparison to RFLP and RAPD markers. Theor. Appl. Genet. 94: 597–602; 1997.
Negi D.; Saxena S. Ascertaining clonal fidelity of tissue culture raised plants of Bambusa balcoa Roxb. using inter simple sequence repeat markers. New. For. 40: 1–8; 2010.
Palombi M. A.; Damiano C. Comparison between RAPD and SSR molecular markers in detecting genetic variation in kiwifruit (Actinidia deliciosa A. Chev). Plant Cell Rep. 20: 1061–1066; 2002.
Pathak H.; Dhawan V. Molecular analysis of micropropagated apple rootstock MM 111 using ISSR markers for ascertaining clonal fidelity. Acta. Hortic. 865: 73–80; 2010.
Phillips R. L.; Kaeppler S. M.; Olhoft P. Genetic instability of plant tissue cultures: breakdown of normal controls. Proc. Natl. Acad. Sci. USA 91: 5222–5226; 1994.
Piola F.; Rohr R.; Heizmann P. Rapid detection of genetic variation within and among in vitro propagated cedar (Cedrus ibani Loudon) clones. Plant. Sci. 141: 159–163; 1999.
Rani V.; Raina S. N. Genetic analysis of enhanced axillary branching derived Eucalyptus treticornis and E. camaldulensis plants. Plant Cell Rep. 17: 236–242; 1998.
Rani V.; Raina S. N. Genetic fidelity of organized meristem derived micropropagated plants: a critical reappraisal. In Vitro Cell. Dev. Biol. Plant. 36: 319–330; 2000.
Rani V.; Raina S. N. Molecular DNA marker analysis to assess the genetic fidelity of micropropagated woody plants. In: Jain S. M.; Ishii K. (eds) Micropropagation of woody trees and fruits. Kluwer Academic, Dordrecht, pp 75–102; 2003.
Ray T.; Dutta I.; Saha P.; Das S.; Roy S. C. Genetic stability of three economically important micropropagated banana (Musa spp.) cultivars of lower Indo-Gangetic plains, as assessed by RAPD and ISSR markers. Plant Cell Tiss. Org. Cult. 85: 11–21; 2006.
Reddy P. M.; Saral N.; Siddiq E. A. Inter simple sequence repeat (ISSR) polymorphism and its application in plant breeding. Euphytica 128: 9–17; 2002.
Sreedhar R. V.; Lakshmanan V.; Bhagyalakshmi N. Genetic fidelity of long-term micropropagated shoot cultures of vanilla (Vanilla planifolia Andrews) as assessed by molecular markers. Biotech. J. 2: 1007–1013; 2007.
Venkatachalam L.; Sreedhar R. V.; Bhagyalakshmi. Micropropagation in banana using high levels of cytokinins does not involve any genetic changes as revealed by RAPD and ISSR markers. Plant Growth Regul. 51: 193–205; 2007.
Virscek-Marn M.; Javornik B.; Stampar F.; Bohanec B. Assessment of genetic variation among regenerants from in vitro apple leaves using molecular markers. Acta. Hortic. 484: 299–303; 1998.
Wallner E.; Weising K.; Rompf R.; Kahl G.; Kopp B. Oligonucleotide and RAPD analysis of Achillea species: characterization and long-term monitoring of micropropagated clones. Plant Cell Rep. 15: 647–652; 1996.
Webster A. D. Temperate fruit tree rootstock propagation. New Zealand J. Crop. Hortic. Sci. 23: 355–372; 1995.
Weising K.; Nybom H.; Wolff K.; Kahl G. DNA fingerprinting in plants: principles, methods, and applications. CRC, New York; 2005.
Williams J. G. K.; Kubelik A.; Livak K. J.; Rafalski J. A.; Tingey S. V. DNA polymorphisms amplified by arbitrary primers are useful as genetic markers. Nucl. Acids. Res. 18: 6531–6535; 1990.
Yang W.; de Oliveira A. C.; Godwin I.; Schertz K.; Bennetzen J. L. Comparison of DNA marker technologies in characterizing plant genome diversity: variability in Chinese sorghums. Crop. Sci. 36: 1669–1676; 1996.
Zietkiewicz E.; Rafalski A.; Labuda D. Genome fingerprinting by simple sequence repeat (SSR)-anchored polymerase chain reaction amplification. Genomics 20: 176–183; 1994.
Zimmerman R. H. Micropropagation of temperate zone fruit and nut crops. In: Debergh P. C.; Zimmerman R. H. (eds) Micropropagation. Kluwer Academic, Dordrecht, pp 231–246; 1991.
Acknowledgments
The authors are grateful to Dr. RK Pachauri, Director General, TERI, for infrastructure support. Thanks are due to Dr. Anandita Singh, Dr. SB Tripathi, and Mr. MS Negi for their assistance. The Senior Research Fellowship from the Council of Scientific and Industrial Research to Ms. Harshita Pathak is duly acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Editor: J. Finer
Rights and permissions
About this article
Cite this article
Pathak, H., Dhawan, V. ISSR assay for ascertaining genetic fidelity of micropropagated plants of apple rootstock Merton 793. In Vitro Cell.Dev.Biol.-Plant 48, 137–143 (2012). https://doi.org/10.1007/s11627-011-9385-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-011-9385-0