Abstract
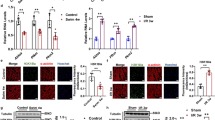
During aging, organs such as skeletal muscle and heart require sufficient NAD+ both as a coenzyme for oxidative-reductive electron transfer and as a substrate for multiple signaling pathways. Sirtuins (SIRTs), a family of NAD+-dependent deacetylase, play an important role in regulating mitochondrial homeostasis and antioxidant defense by deacetylating transcription factors and enzymes such as PGC-1α, p65, GCN5, and SOD2. However, age-related DNA damage and increased SASP activate PARP-1 and CD38, the enzymes competing with SIRTs for NAD+. Thus, it is important to know how aging alters intracellular NAD+ status and NAD+-depending enzyme expression in muscles. In this study, we report that the acetylation level of muscle protein pool, as well as major SIRTs target proteins (PGC-1α, GCN5, p65, and SOD2), was significantly increased in hindlimb and cardiac muscles of 24-month old mice compared with their 6-month old counterparts, despite the fact that most members of the SIRT family were upregulated with aging. Aging increased the protein content of PARP-1 and CD38, whereas decreased NAD+ levels in both skeletal and heart muscles. Aged muscles demonstrated clear signs of mitochondrial dysfunction, oxidative stress, and inflammation. Taken together, our data suggest that despite the upregulation of SIRTs, aged muscles suffered from NAD+ deficit partly due to the competition of elevated CD38 and PARP-1. The enhanced acetylation of several key proteins involved in broad cellular functions may contribute to the age-related muscle deterioration.







Similar content being viewed by others
References
Badreh F, Joukar S, Badavi M, Rashno M, Dehesh T. The effects of age and fasting models on blood pressure, insulin/glucose profile, and expression of longevity proteins in male rats. Rejuvenation Res. 2019. https://doi.org/10.1089/rej.2019.2205.
Bai P, Cantó C, Oudart H, et al. PARP-1 inhibition increases mitochondrial metabolism through SIRT1 activation. Cell Metab. 2011. https://doi.org/10.1016/j.cmet.2011.03.004.
Camacho-Pereira J, Tarragó MG, Chini CS, et al. CD38 dictates age-related NAD decline and mitochondrial dysfunction through and SIRT3-dependent mechanism. Cell Metab. 2016. https://doi.org/10.1016/j.cmet.2016.05.006.
Cantó C, Menzies KJ, Auwerx J. NAD+ metabolism and the control of energy homeostasis: a balancing act between mitochondria and the nucleus. Cell Metab. 2015. https://doi.org/10.1016/j.cmet.2015.05.023.
Chaitanya GV, Alexander JS, Babu PP. PARP-1 cleavage fragments: signatures of cell-death proteases in neurodegeneration. Cell Commun Signal. 2010. https://doi.org/10.1186/1478-811X-8-31.
Chini EN. CD38 as a regulator of cellular NAD: a novel potential pharmacological target for metabolic conditions. Curr Pharm Des. 2009. https://doi.org/10.2174/138161209787185788.
Chini EN, Chini CS, Netto JME, et al. The pharmacology of CD38/NADase: an emerging target for cancer and aging diseases. Trends Pharmacol Sci. 2018. https://doi.org/10.1016/j.tips.2018.02.001.
Chini CS, Hogan KA, Warner GM, et al. The NADase CD38 is induced by factors secreted from senescent cells providing a potential link between senescence and age-related cellular NAD (+) decline. Biochem Biophys Res Commun. 2019. https://doi.org/10.1016/j.bbrc.2019.03.199.
Chini CS, Tarragó MG, Chini EN. NAD and the aging process: Role in life, death and everything in between. Mol Cell Endocrinol. 2017. https://doi.org/10.1016/j.mce.2016.11.003.
Dominy JE, Lee Y, Jedrychowski MP, et al. The deacetylase Sirt6 activates the acetyltransferase GCN5 and suppresses hepatic gluconeogenesis. Mol Cell. 2012. https://doi.org/10.1016/j.molcel.2012.09.030.
Fang EF, Scheibye-Knudsen M, Brace LE, et al. Defective mitophagy in XPA via PARP-1 hyperactivation and NAD+/SIRT1 reduction. Cell. 2014. https://doi.org/10.1016/j.cell.2014.03.026.
Fang EF, Kassahun H, Croteau DL, et al. NAD+ replenishment improves lifespan and healthspan in ataxia telangiectasia models via mitophagy and DNA repair. Cell Metab. 2016. https://doi.org/10.1016/j.cmet.2016.09.004.
Fernandez-Marcos PJ, Auwerx J. Regulation of PGC-1α, a nodal regulator of mitochondrial biogenesis. Am J Clin Nutr. 2011. https://doi.org/10.3945/ajcn.110.001917.
Frederick DW, Loro E, Liu L, et al. Loss of NAD homeostasis leads to progressive and reversible degeneration of skeletal muscle. Cell Metab. 2016. https://doi.org/10.1016/j.cmet.2016.07.005.
Gómez LA, Hagen TM. Age-related decline in mitochondrial bioenergetics: does supercomplex destabilization determine lower oxidative capacity and higher superoxide production? Semin Cell Dev Biol doi. 2012. https://doi.org/10.1016/j.semcdb.2012.04.002.
Guan KL, Xiong Y. Regulation of intermediary metabolism by protein acetylation. Trends Biochem Sci. 2011. https://doi.org/10.1016/j.tibs.2010.09.003.
Haigis MC, Guarente LP. Mammalian sirtuins - emerging roles in physiology, aging, and calorie restriction. Genes Dev. 2006. https://doi.org/10.1101/gad.1467506.
Handschin C. The biology of PGC-1α and its therapeutic potential. Trends Pharmacol Sci. 2009. https://doi.org/10.1016/j.tips.2009.03.006.
Hepple RT. Mitochondrial involvement and impact in aging skeletal muscle. Front Aging Neurosci. 2014. https://doi.org/10.3389/fnagi.2014.00211.
Imai SI, Guarente L. NAD+ and sirtuins in aging and disease. Trends Cell Biol. 2014. https://doi.org/10.1016/j.tcb.2014.04.002.
Johnson S, Imai S. NAD+ biosynthesis, aging, and disease. F1000Research. 2018. https://doi.org/10.12688/f1000research.12120.1.
Kabiljo J, Murko C, Pusch O, Zupkovitz G. Spatio-temporal expression profile of sirtuins during aging of the annual fish Nothobranchius furzeri. Gene Expr Patterns. 2019. https://doi.org/10.1016/j.gep.2019.05.001.
Kang C, Chung E, Diffee G, Ji LL. Exercise training attenuates aging-associated mitochondrial dysfunction in rat skeletal muscle: role of PGC-1α. Exp Gerontol. 2013. https://doi.org/10.1016/j.exger.2013.08.004.
Kang C, Goodman CA, Hornberger TA, Ji LL. PGC‐1α overexpression by transfection attenuates mitochondrial deterioration of skeletal muscle caused by immobilization. FASEB J. 2015. https://doi.org/10.1096/fj.14-266619.
Kawahara TLA, Michishita E, Adler AS, et al. SIRT6 links histone H3 lysine 9 deacetylation to NF-κB-dependent gene expression and organismal life span. Cell. 2009. https://doi.org/10.1016/j.cell.2008.10.052.
Khanh VC, Zulkifli AF, Tokunaga C, et al. Aging impairs beige adipocyte differentiation of mesenchymal stem cells via the reduced expression of Sirtuin 1. Biochem Biophys Res Commun. 2018. https://doi.org/10.1016/j.bbrc.2018.04.136.
Kilic U, Gok O, Erenberk U, et al. A remarkable age-related increase in SIRT1 protein expression against oxidative stress in elderly: SIRT1 gene variants and longevity in human. PLoS One. 2015. https://doi.org/10.1371/journal.pone.0117954.
Kim SC, Sprung R, Chen Y, et al. Substrate and functional diversity of lysine acetylation revealed by a proteomics survey. Mol Cell. 2006. https://doi.org/10.1016/j.molcel.2006.06.026.
Koltai E, Szabo Z, Atalay M, et al. Exercise alters SIRT1, SIRT6 NAD and NAMPT levels in skeletal muscle of aged rats. Mech Ageing Dev. 2010. https://doi.org/10.1016/j.mad.2009.11.002.
Kolthur-Seetharam U, Dantzer F, McBurney MW, et al. Control of AIF-mediated cell death by the functional interplay of SIRT1 and PARP-1 in response to DNA damage. Cell Cycle. 2006. https://doi.org/10.4161/cc.5.8.2690.
Konopka AR, Suer MK, Wolff CA, Harber MP. Markers of human skeletal muscle mitochondrial biogenesis and quality control: effects of age and aerobic exercise training. J Gerontol A Biol Sci Med Sci. 2014. https://doi.org/10.1093/gerona/glt107.
Kugel S, Mostoslavsky R. Chromatin and beyond: the multitasking roles for SIRT6. Trends Biochem Sci. 2014. https://doi.org/10.1016/j.tibs.2013.12.002.
Lombard DB, Alt FW, Cheng H-L, et al. Mammalian Sir2 homolog SIRT3 regulates global mitochondrial lysine acetylation. Mol Cell Biol. 2007. https://doi.org/10.1128/mcb.01636-07.
Michan S, Sinclair D. Sirtuins in mammals: insights into their biological function. Biochem J. 2007. https://doi.org/10.1042/BJ20070140.
Michishita E. Evolutionarily conserved and nonconserved cellular localizations and functions of human SIRT proteins. Mol Biol Cell. 2005. https://doi.org/10.1091/mbc.e05-01-0033.
Mills KF, Yoshida S, Stein LR, et al. Long-term administration of nicotinamide mononucleotide mitigates age-associated physiological decline in mice. Cell Metab. 2016. https://doi.org/10.1016/j.cmet.2016.09.013.
Mitchell SJ, Bernier M, Aon MA, et al. Nicotinamide improves aspects of healthspan, but not lifespan, in mice. Cell Metab. 2018. https://doi.org/10.1016/j.cmet.2018.02.001.
Mouchiroud L, Houtkooper RH, Moullan N, et al. The NAD+/sirtuin pathway modulates longevity through activation of mitochondrial UPR and FOXO signaling. Cell. 2013. https://doi.org/10.1016/j.cell.2013.06.016.
Nemoto S, Fergusson MM, Finkel T. SIRT1 functionally interacts with the metabolic regulator and transcriptional coactivator PGC-1α. J Biol Chem. 2005. https://doi.org/10.1074/jbc.M501485200.
Palikaras K, Tavernarakis N. Mitochondrial homeostasis: the interplay between mitophagy and mitochondrial biogenesis. Exp Gerontol doi. 2014. https://doi.org/10.1016/j.exger.2014.01.021.
Picca A, Calvani R, Bossola M, et al. Update on mitochondria and muscle aging: all wrong roads lead to sarcopenia. Biol Chem. 2018. https://doi.org/10.1515/hsz-2017-0331.
Sack MN, Finkel T. Mitochondrial metabolism, sirtuins, and aging. Cold Spring Harb Perspect Biol. 2012. https://doi.org/10.1101/cshperspect.a013102.
Sakellariou GK, Pearson T, Lightfoot AP, Nye GA, Wells N, Giakoumaki II, et al. Mitochondrial ROS regulate oxidative damage and mitophagy but not age-related muscle fiber atrophy. Sci Rep. 2016;6:1–15. https://doi.org/10.1038/srep33944.
Salminen A, Kaarniranta K. NF-κB signaling in the aging process. J Clin Immunol. 2009. https://doi.org/10.1007/s10875-009-9296-6.
Sanchez AMJ, Candau RB, Bernardi H. FoxO transcription factors: their roles in the maintenance of skeletal muscle homeostasis. Cell Mol Life Sci. 2014. https://doi.org/10.1007/s00018-013-1513-z.
Sasaki T, Maier B, Bartke A, Scrable H. Progressive loss of SIRT1 with cell cycle withdrawal. Aging Cell. 2006. https://doi.org/10.1111/j.1474-9726.2006.00235.x.
Schöndorf DC, Ivanyuk D, Baden P, et al. The NAD+ precursor nicotinamide riboside rescues mitochondrial defects and neuronal loss in iPSC and fly models of Parkinson’s disease. Cell Rep. 2018. https://doi.org/10.1016/j.celrep.2018.05.009.
Schultz MB, Sinclair DA. Why NAD+ declines during aging: it’s destroyed. Cell Metab. 2017. https://doi.org/10.1016/j.cmet.2016.05.022.
Tao R, Coleman MC, Pennington JD, et al. Sirt3-mediated deacetylation of evolutionarily conserved lysine 122 regulates MnSOD activity in response to stress. Mol Cell. 2010. https://doi.org/10.1016/j.molcel.2010.12.013.
Tarragó MG, Chini CS, Kanamori KS, et al. A potent and specific CD38 inhibitor ameliorates age-related metabolic dysfunction by reversing tissue NAD+ decline. Cell Metab. 2018. https://doi.org/10.1016/j.cmet.2018.03.016.
Trammell SAJ, Weidemann BJ, Chadda A, et al. Nicotinamide riboside opposes type 2 diabetes and neuropathy in mice. Sci Rep. 2016. https://doi.org/10.1038/srep26933.
Verdin E. NAD + in aging, metabolism, and neurodegeneration. Science. 2015. https://doi.org/10.1126/science.aac4854.
Wagner GR, Payne RM. Mitochondrial acetylation and diseases of aging. J Aging Res. 2011. https://doi.org/10.4061/2011/234875.
Webb AE, Brunet A. FOXO transcription factors: key regulators of cellular quality control. Trends Biochem Sci. 2014. https://doi.org/10.1016/j.tibs.2014.02.003.
Yeo D, Kang C, Gomez-Cabrera MC, Vina J, Ji LL. Intensified mitophagy in skeletal muscle with aging is downregulated by PGC-1alpha overexpression in vivo. Free Radic Biol Med. 2019. https://doi.org/10.1016/j.freeradbiomed.2018.10.456.
Yeung F, Hoberg JE, Ramsey CS, et al. Modulation of NF-κB-dependent transcription and cell survival by the SIRT1 deacetylase. EMBO J. 2004. https://doi.org/10.1038/sj.emboj.7600244.
Zhu X, Liu Q, Wang M, et al. Activation of Sirt1 by resveratrol inhibits TNF-α induced inflammation in fibroblasts. PLoS One. 2011. https://doi.org/10.1371/journal.pone.0027081.
Funding
This research was supported by a grant from the Office of Vice President for Research at the University of Minnesota, Grant-in-Aid, 146825.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 203 kb)
About this article
Cite this article
Yeo, D., Kang, C. & Ji, L.L. Aging alters acetylation status in skeletal and cardiac muscles. GeroScience 42, 963–976 (2020). https://doi.org/10.1007/s11357-020-00171-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11357-020-00171-7