Abstract
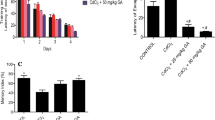
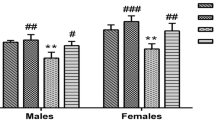
Cadmium (Cd) is a highly toxic heavy metal. It accumulates in biological tissues, especially in fish which constitutes a first rank food for humans, particularly in the coastal areas. This study investigates the effect of long-term exposure to low Cd concentration (17 μg/kg/day) in rat striatum and hippocampus. In this study, the neurobehavioral ability changes were assessed by applying cognitive standard testing at the end of the rats’ exposure period. In addition, the examination of mitochondrial swelling was performed at the same time of evaluation of its redox status in the brain regions studied through stress parameters (GSH, MDA, GST, and CAT). This study examined also whether this long-term exposure can modify the apoptotic signaling pathway via assessment of apoptotic markers (caspase-8 and 9, Bax, Bcl-2, and Cyt-c) in cell lysates. The results of this study showed changes in neurobehavioral abilities of animals and a stronger mitochondrial swelling associated with a significant decrease in antioxidant systems (GSH, GST, and CAT) and conversely an increase in the lipoperoxidation end product (MDA) in both the striatal and hippocampal mitochondria. In addition, the results revealed a significant increase in pro-apoptotic intracellular components such as caspase-9, Cyt-c, and Bax, and showed also an evident decrease in Bcl-2 levels. In conclusion, our results reported that chronic exposure to Cd produces behavioral and cognitive perturbations, enhances oxidative stress associated with mitochondrial edema and Cyt-c leakage, and, ultimately, potentiates apoptosis signaling pathway in both brain regions in rats.








Similar content being viewed by others
References
Abdalla FH, Schmatz R, Cardoso AM, Carvalho FB, Baldissarelli J, de Oliveira JS, Rosa MM, Gonçalves Nunes MA, Rubin MA, da Cruz IBM, Barbisan F, Dressler VL, Pereira LB, Schetinger MRC, Morsch VM, Gonçalves JF, Mazzanti CM (2014) Quercetin protects the impairment of memory and anxiogenic-like behavior in rats exposed to cadmium: possible involvement of the acetylcholinesterase and Na+,K+-ATPase activities. Physiol Behav 135:152–167
Abdeen A, El-Shawarby GA, Abdel-Aleem R, El-Shewy N, Abdo E et al (2017) Protective effect of cinnamon against cadmium-induced hepatorenal oxidative damage in rats. Int J Pharmacol Toxicol 5(1):17–22
Abdeen A, Abou-zaid OA, Abdel-Maksoud HA, Aboubakr M, Abdelkader A et al (2019) Cadmium overload modulates piroxicam-regulated oxidative damage and apoptotic pathways. Environ Sci Pollut Res 26(24):25167–25177. https://doi.org/10.1007/s11356-019-05783-x
Agnihotri SK, Agrawal U, Ghosh I (2015) Brain most susceptible to cadmium induced oxidative stress in mice. J Trace Elem Med Biol 30:184–193
Ahmed MB, Ahmed ML, Meki A, Abdraboh N (2013) Neurotoxic effect of lead on rats: relationship to apoptosis. Int J Health Sci 7:192–199
Alnahdi HS, Sharaf IA (2019) Possible prophylactic effect of omega-3 fatty acids on cadmium-induced neurotoxicity in rats’ brains. Environ Sci Pollut Res 26:31254–31262. https://doi.org/10.1007/s11356-019-06259-8
Andjelkovic M, BuhaDjordjevic A, Antonijevic E et al (2019) Toxic effect of acute cadmium and lead exposure in rat blood, liver, and kidney. Int J Environ Res Public Health 16(2):274
Andra SS, Austin S, Kumar D et al (2017) Trends in the application of high-resolution mass spectrometry for human biomonitoring: an analytical primer to studying the environmental chemical space of the human exposome. Env Inter 100:32–61
Antunes M, Biala G (2011) The novel object recognition memory: neurobiology, test procedure, and its modifications. Cogn Process 13(2):93–110
Assefa Z, Van Laethem A, Garmyn M, Agostinis P (2005) Ultraviolet radiation induced apoptosis in keratinocytes: on the role of cytosolic factors. Biochim Biophys Acta 1755:90–106
ATSDR (2004). Guidance Manual for the Assessment of Joint Toxic Action of Chemical Mixtures. Atlanta GA:Available: Agency for Toxic Substances and Disease Registry. http://www.atsdr.cdc.gov/interactionprofiles/ipga.html. Accessed 1 Mar 2005
Baldo B, and Petersén Å (2015) Analysis of nonmotor features in murine models of huntington disease. Movement Disorders, 583–602. https://doi.org/10.1016/B978-0-12-405195-9.00035-4 In book: Movement Disorders, pp.583-602
Batool Z, Agha F, Tabassum S, Batool TS, Siddiqui RA, Haider S (2019) Prevention of cadmium-induced neurotoxicity in rats by essential nutrients present in nuts. Acta Neurobiol Exp (Wars) 79(2):169–183
Beghoul A, Kebieche M, Gasmi S, Chouit Z, Amiour C, Lahouel A, Lakroun Z, Rouabhi R, Fetoui H, Soulimani R (2017) Impairment of mitochondrial integrity and redox status in brain regions during a low-dose long-term exposition of rats to pyrethrinoïds: the preventive effect of quercetin. Environ Sci Pollut Res Int 24:19714–19722
Bonora M, Morganti C, Morciano G, Giorgi C, Wieckowski MR, Pinton P (2016) Comprehensive analysis of mitochondrial permeability transition pore activity in living cells using fluorescence-imaging-based techniques. Nat Protoc 11:1067–1080. https://doi.org/10.1038/nprot.2016.064
Bradford MA (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein – dye binding. Anal Biochem 72:248–254
Calabrese V, Bates TE, Stella AMG (2000) NO synthase and NO-dependent signal pathways in brain aging and neurodegenerative disorders: the role of oxidant/antioxidant balance. Neurochem Res 25:1315–1341
Chen L, Liu L, Huang S (2008) Cadmium activates the mitogen-activated protein kinase (MAPK) pathway via induction of reactive oxygen species and inhibition of protein phosphatases 2A and 5. Free Radical Bio Med 45(7):1035–1044
Chen H, Yoshioka H, Seok Kim G et al (2011) Oxidative stress in ischemic brain damage: mechanisms of cell death and potential molecular targets for neuroprotection. Antioxid Redox Signal 14(8):1505–1517. https://doi.org/10.1089/ars.2010.3576
Chen X, Wang Z, Zhu G, Nordberg GF, Jin T, Ding X (2019) The association between cumulative cadmium intake and osteoporosis and risk of fracture in a Chinese population. J Expo Sci Environ Epidemiol 29:435–443. https://doi.org/10.1038/s41370-018-0057-6
Chin-Chan M, Navarro-Yepes J, Quintanilla-Vega B (2015) For neurodegenerative disorders: Alzheimer and Parkinson diseases. Front Cell Neurosci. https://doi.org/10.3389/fncel.2015.00124
Claiborne A (1985) Catalase activity. In: Greenwald RA (ed) Handbook of methods in oxygen radical research. CRC Press, Boca Raton, pp 283–284
Clayton R, Clark JB, Sharpe M (2005) Cytochrome c release from rat brain mitochondria is proportional to the mitochondrial functional deficit: implications for apoptosis and neurodegenerative disease. J Neurochem 92:840–849. https://doi.org/10.1111/j.1471-4159.2004.02918.x
Cobbina SJ, Chen Y, Zhou Z, Wu X, Zhao T, Zhang Z, Yang L (2015) Toxicity assessment due to sub-chronic exposure to individual and mixtures of four toxic heavy metals. J Hazard Mater 294:109–120
Crawley JN, Collins JK, Crabbe JC, Henderson WF et al (1997) Behavioral phenotypes of inbred mouse strains: implications and recommendations for molecular studies. Psychopharmacol 132:107–124
Crépeaux G, Bouillaud-Kremarik P, Sikhayeva N, Rychen G, Soulimani R, Schroeder H (2012) Late effects of a perinatal exposure to a 16 PAH mixture: increase of anxiety-related behaviours and decrease of regional brain metabolism in adult male rats. Toxicol Lett 211(2):105–113
Deacon BJ (2013) The biomedical model of mental disorder: a critical analysis of its validity, utility, and effects on psychotherapy research. Clin Psychol Rev 33(7):846–861. https://doi.org/10.1016/j.cpr.2012.09.007
Del Pino J, Zeballos G, Anadon MJ, Capo MA, Díaz MJ, García J, Frejo MT (2014) Higher sensitivity to cadmium induced cell death of basal forebrain cholinergic neurons: a cholinesterase dependent mechanism. Toxicol 325:151–159
EFSA (European Food Safety Authority) (2011).Guidance on default assumptions used by the EFSA scientific panels and committee, and EFSA units in the absence of actual measured data: 1-30
Ekshyyan O, Aw TY (2004) Apoptosis: a key in neurodegenerative disorders. Curr Neurovasc Res 1(4):355–371
El-kott AF, Bin-Meferij MM, Eleawa SM et al (2020) Kaempferol protects against cadmium chloride-induced memory loss and hippocampal apoptosis by increased intracellular glutathione stores and activation of PTEN/AMPK induced inhibition of Akt/mTOR signaling. Neurochem Res 45:295–309. https://doi.org/10.1007/s11064-019-02911-4
Ellman GL (1959) Tissue sulfhydryl groups. Arch Biochem Biophys 82:70–77
Engwa GA, Ferdinand PU, Nwalo FN, & Unachukwu MN (2019) Mechanism and health effects of heavy metal toxicity in humans. In Poisoning in the Modern World New Tricks for an Old Dog? IntechOpen
Ennaceur A, Chazot PL (2016) Preclinical animal anxiety research–flaws and prejudices. Pharmacol Res Perspect 4(2):e00223
Gasmi S, Chafaa S, Lakroun Z, Rouabhi R, Touahria C, Kebieche M, Soulimani R (2019) Neuronal apoptosis and imbalance of neurotransmitters induced by acetamiprid in rats. Toxicol Environ Heal Sci 11:305–311. https://doi.org/10.1007/s13530-019-0417-1
Gould TD (2009) Mood and anxiety related phenotypes in mice: Characterization Using Behavioral Tests, neuromethode 42 .springer protocols. Humanapress, p: 55
Habig WH, Pabst MJ and Jakoby WB (1974) Glutathione S-transferases. The first enzymatic step in mercapturic acid formation. J Biol Chem 249(22):7130-7139
Habila S (2018) Evaluation de risque écologique et sanitaire de la contamination des eaux et des sédiments du barrage Beni Haroun (wilaya de Mila, Algerie). thèse de doctorat en science, SNV, Université de Jijel, Algérie, p: 44. https://www.researchgate.net/publication/326677936_Evaluation_du_risque_ecologique_et_sanitaire_de_la_contamination_des_eaux_et_des_sediments_d_u_barrage_Beni_Haroun_Wilaya_de_Mila
Haider S, Anis L, Batool Z, Sajid I, Naqvi F, Khaliq S, Ahmed S (2014) Short term cadmium administration dose dependently elicits immediate biochemical, neurochemical and neurobehavioral dysfunction in male rats. Metab Brain Dis 30:83–92
Järup L (2003) Hazards of heavy metal contamination. Br Med Bull 68(1):167–182
Jomova K, Valko M (2011) Advances in metal-induced oxidative stress and humandisease. Toxicol 283:65–87
Kalueff AV, Keisala T, Minasyan A, Kumar SR, LaPorte JL, Murphy DL, Tuohimaa P (2008) The regular and light-dark Suok tests of anxiety and sensorimotor integration: utility for behavioral characterization in laboratory rodents. Nat Protoc 3(1):129–136
Kaoud H, Kamel MM, Abeer H, Abdel-Razek AH et al (2010) Neurobehavioural, neurochemical and neuromorphological effects of cadmium in male rats. J Am Sci 6(5):189–202
Kim W, Sun Yim H, Dae Young Yoo D, Hwang I (2016) Dendropanax morbifera Léveille extract ameliorates cadmium-induced impairment in memory and hippocampal neurogenesis in rats. BMC Complement Altern Med 16(1). https://doi.org/10.1186/s12906-016-1435-z
Lahouel A, Kebieche M, Lakroun Z, Rouabhi R, Fetoui H, Chtourou Y, Zama D, Soulimani R (2016) Nurobehavioral deficits and brain oxidative stress induced by chronic low dose exposure of persistent organic pollutants mixture in adult female rat. Environ Sci Pollut Res 23:19030–19040. https://doi.org/10.1007/s11356-016-6913-9
Lakroun Z, Kebieche M, Lahouel A, Zama D, Soulimani R (2015) Oxidative stress and brain mitochondria swelling induced by endosulfan and protective role of quercetin in rat. Environ Sci Pollut Res 22:7776–7781. https://doi.org/10.1007/s11356-014-3885-5
Lakroun Z, Kebieche M, Lahouel A, Beghoul A, Gesmi S, Rouabhi R, Fetoui H, Soulimani R (2017) Potentiation of apoptosis in rat striatum exposed to endosulfan and the role of quercetin. Toxicol Environ Heal Sci 9:229–236. https://doi.org/10.1007/s13530-017-0325-1
Lamtai M, Chaibat J, Ouakki S, Berkiks I et al (2018) Effect of chronic administration of cadmium on anxiety-like, depression-like and memory deficits in male and female rats: possible involvement of oxidative stress mechanism. J Behavioral Brain Sci 8(5)
Lecorps B, Rödel HG, Féron C (2016) Assessment of anxiety in open field and elevated plus maze using infrared thermography. Physiol Behav 1(157):209–216. https://doi.org/10.1016/j.physbeh.2016.02.014
Li J, Yu W, Li X, Li B (2014) The effects of propofol on mitochondrial dysfunction following focal cerebral ischemia–reperfusion in rats. Neuropharmacol. 77:358–368
Lister RG (1987) The use of a plus-maze to measure anxiety in the mouse. Psychopharmacol 92:180–185
Mahdavi S, Khodarahmi P, Roodbari NH (2018) Effects of cadmium on Bcl-2/Bax expression ratio in rat cortex brain and hippocampus. Hum Exp Toxicol 37(3):321–328. https://doi.org/10.1177/0960327117703687
Mason LH, Harp JP, Han DY (2014) Pb neurotoxicity: neuropsychological effects of lead toxicity. Biomed Res Int:1–9
McStay GP, Salvesen GS, Green DR (2008) Overlapping cleavage motif selectivity of caspases: implications for analysis of apoptotic pathways. Cell Death Differ 15(2):322–331
Ohkawa H, Ohishi N, Yagi K (1979) Assay of lipid peroxides in animal tissue by thiobarbituric reaction. Anal Biochem 95:351–358
Pathak N, Khandelwal S (2006) Influence of cadmium on murine thymocytes: potentiation of apoptosis and oxidative stress. Toxicol Lett 165(2):121–132
Pi H, Li M, Tian L, Yang Z, Yu Z, Zhou Z (2017) Enhancing lysosomal biogenesis and autophagic flux by activating the transcription factor EB protects against cadmium-induced neurotoxicity. Sci Rep 7:43466. https://doi.org/10.1038/srep43466
Richetti SK, Rosemberg DB, Ventura-Lima J, Monserrat JM, Bogo MR, Bonan CD (2011) Acetylcholinesterase activity and antioxidant capacity of zebrafish brain is altered by heavy metal exposure. Neurotoxicol. 32(1):116–122
Rios C, Méndez-Armenta M (2019) Cadmium neurotoxicity. Book chapter, Elsevize Encyclopedia of Environmental Health, pp 474–481. https://doi.org/10.1016/B978-0-12-409548-9.11571-4
Sahu BD, Tatireddy S, Koneru M, Borkar RM, MaheshKumar J et al (2014) Naringin ameliorates gentamicin-induced nephrotoxicity and associated mitochondrial dysfunction, apoptosis and inflammation in rats: possible mechanism of nephroprotection. Toxicol App Pharmacol 277(1):8–20
Shi H, Hudson LG, Liu KJ (2004) Oxidative stress and apoptosis in metal ion induced carcinogenesis. Free Radic Biol Med 37:582–593
Silvane V, Cambraia J, Ribeiro C, Oliveira JA, Oliva MA (2011) Cadmiuminduced oxidative stress and antioxidative enzyme response in water hyacinth and salvinia. Braz J Plant Physiol 23(2):131–139
Tatem KS, Quinn JL, Phadke A et al (2014) Behavioral and locomotor measurements using an open field activity monitoring system for skeletal muscle diseases. J Vis Exp 91
Tobwala S, Wang HJ, Carey JW, Banks WA, Ercal N (2014) Effects of lead and cadmium on brain endothelial cell survival, monolayer permeability, and crucial oxidative stress markers in an in vitro model of the blood-brain barrier. Toxics 2(2):258–275
Valerio Branca JJ, Morucci G, Becatti M et al (2019) Cannabidiol protects dopaminergic neuronal cells from cadmium. Int J Environ Res Public Health 16(22):4420. https://doi.org/10.3390/ijerph16224420
Wang B, & Du Y (2013) Cadmium and its neurotoxic effects. Oxidative med cell longev Volume 2013, Article ID 898034 | 12 pages. https://doi.org/10.1155/2013/898034
Wang T, Wang Q, Song R, Zhang Y, Zhang K, Yuan Y, Bian J, Liu X, Gu J, Liu Z (2015) Autophagy plays a cytoprotective role during cadmium-induced oxidative damage in primary neuronal cultures. Biol Trace Elem Res 168:481–489. https://doi.org/10.1007/s12011-015-0390-8
Whiteside JR, Box CL, McMillan TJ, Allinson SL (2010) Cadmium and copper inhibit both DNA repair activities of polynucleotide kinase. DNA repair 9(1):83–89
Wylly GNR, Pedraza-Chaverrí J (2014) Protective effect of curcumin against heavy metals-induced liver damage. Food Chem Toxicol 69:182–201
Xu Z, Jin X, Pan T, Liu T, Wan N, Li S (2017) Antagonistic effects of selenium on cadmium-induced apoptosis by restoring the mitochondrial dynamic equilibrium and energy metabolism in chicken spleens. Oncotarget 8(32):52629–52641
Yuan Y, Zhang Y, Zhao S, Chen J, Yang J, Wang T, Zou H, Wang Y, Gu J, Liu X, Bian J, Liu Z (2018) Cadmium-induced apoptosis in neuronal cells is mediated by Fas/FasL-mediated mitochondrial apoptotic signaling pathway. Sci Rep 8:8837. https://doi.org/10.1038/s41598-018-27106-9
Acknowledgments
This study was extracted from the PhD thesis of Ms. Zeyneb Chouit (first author); it was supported by the Ministry of High Education and Scientific Research of Algeria, via the University of Jijel and University of Batna2. This work was done without a specific grant from any funding agency (public or private).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All animal experiments in the current study are approved by PIA under the ethical code: N° Batna-Univ2.2020.231.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Responsible Editor: Mohamed M. Abdel-Daim
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chouit, Z., Djellal, D., Haddad, S. et al. Potentiation of the apoptotic signaling pathway in both the striatum and hippocampus and neurobehavioral impairment in rats exposed chronically to a low−dose of cadmium. Environ Sci Pollut Res 28, 3307–3317 (2021). https://doi.org/10.1007/s11356-020-10755-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-020-10755-7