Abstract
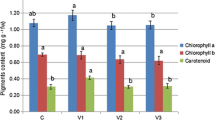
Lead (Pb) environmental contamination remains prevalent. Pisum sativum L. plants have been used in ecotoxicological studies, but some cultivars showed to tolerate and accumulate some levels of Pb, opening new perspectives to their use in phytoremediation approaches. However, the putative use of pea plants in phytoremediation requires reliable toxicity endpoints. Here, we evaluated the sensitivity of a large number of photosynthesis-related biomarkers in Pb-exposed pea plants. Plants (cv. “Corne de Bélier”) were exposed to Pb concentrations up to 1,000 mg kg−1 soil during 28 days. The photosynthetic potential biomarkers that were analyzed included pigments, chlorophyll (Chl) a fluorescence, gas exchange, ribulose-1,5-bisphosphate carboxylase/oxygenase (RuBisCO) activity, and carbohydrates. Flow cytometry (FCM) was also used to assess the morpho-functional status of chloroplasts. Finally, Pb-induced nutrient disorders were also evaluated. Net CO2 assimilation rate (A) and RuBisCO activity decreased strongly in Pb-exposed plants. Plant dry mass (DM) accumulation, however, was only reduced in the higher Pb concentrations tested (500 and 1,000 mg kg−1 soil). Pigment contents increased solely in plants exposed to the largest Pb concentration, and in addition, the parameters related to the light-dependent reactions of photosynthesis, F v/F m and ΦPSII, were not affected by Pb exposure. In contrast to this, carbohydrates showed an overall tendency to increase in Pb-exposed plants. The morphological status of chloroplasts was affected by Pb exposure, with a general trend of volume decrease and granularity increase. These results point the endpoints related to the light-independent reactions of photosynthesis as more sensitive predictors of Pb-toxicity than the light-dependent reactions ones. Among the endpoints related to the light-independent photosynthesis reactions, RuBisCO activity and A were found to be the most sensitive. We discuss here the advantages of using these parameters as biomarkers for Pb toxicity in plants. Finally, we report that, despite showing physiological disorders, these cultivar plants survived and accumulated high doses of Pb, and their use in environmental/decontamination studies is open to debate.




Similar content being viewed by others
References
Alzandi AR (2012) The use of Pisum sativum L as bioindicator for lead ions detoxification. Int J Sci Technol Res 1(2):42–45
An YJ (2004) Soil ecotoxicity assessment using cadmium sensitive plants. Environ Pollut 127:21–6
Aravind P, Prasad MN, Malec P, Waloszek A, Strzalka K (2009) Zinc protects Ceratophyllum demersum L. (free-floating hydrophyte) against reactive oxygen species induced by cadmium. J Trace Elem Med Biol 23:50–60
Bah A, Sun H, Chen F, Zhou J, Dai H, Zhang G, Wu F (2010) Comparative proteomic analysis of Typha angustifolia leaf under chromium, cadmium and lead stress. J Hazard Mater 184:191–203
Berti W, Cunningham S (1997) In-place inactivation of Pb in Pb-contaminated soils. Environ Sci Technol 31:1359–1364
Bertrand M, Poirier I (2005) Photosynthetic organisms and excess of metals. Photosynthetica 43:345–353
Bibi M, Hussain M (2005) Effect of copper and lead on photosynthesis and plant pigments in black gram [Vigna mungo (L.) Hepper]. Bull Environ Contam Toxicol 74:1126–33
Breckle SW, Kahle H (1992) Effects of toxic heavy-metals (Cd, Pb) on growth and mineral-nutrition of beech (Fagus sylvatica L). Vegetatio 101:43–53
Burzynski M, Klobus G (2004) Changes of photosynthetic parameters in cucumber leaves under Cu, Cd, and Pb stress. Photosynthetica 42:505–510
Capelo A, Santos C, Loureiro S, Pedrosa M (2012) Phytotoxicity of lead on Lactuca sativa: effects on growth, mineral nutrition, photosynthetic activity and oxidant metabolism. Fresenius Envir Bull 21:450–459
Cenkci S, Ciğerci IH, Yıldız M, Özay C, Bozdağ A, Terzi H (2010) Lead contamination reduces chlorophyll biosynthesis and genomic template stability in Brassica rapa L. Environ Exp Bot 67:467–473
Choudhury NK, Behera RK (2001) Photoinhibition of photosynthesis: role of carotenoids in photoprotection of chloroplast constituents. Photosynthetica 39:481–488
Clijsters H, Van Assche F (1985) Inhibition of photosynthesis by heavy metals. Photosynth Res 7:31–40
Devi R, Mundiral N, Gupta A, Kaur N (2013) Effect of exogenous lead on growth and carbon metabolism of pea (Pisum sativum L) seedlings. Physiol Mol Biol Plants 19(1):81–89
Dias MC, Brüggemann W (2010) Limitations of photosynthesis in Phaseolus vulgaris under drought stress: gas exchange, chlorophyll fluorescence and Calvin cycle enzymes. Photosynthetica 48:96–102
Dias MC, Monteiro C, Moutinho-Pereira J, Correia C, Gonçalves B, Santos C (2013) Cadmium toxicity affects photosynthesis and plant growth at different levels. Acta Physiol Plant 35:1281–1289
Gupta K, Huang H, Corpas F (2013) Lead tolerance in plants: strategies for phytoremediation. Environ Sci Pollut Res 20:2150–2161
Hall JL (2002) Cellular mechanism of heavy metal detoxification and tolerance. J Exp Bot 53:1–11
Hardison DW Jr, Ma LQ, Luongo T, Harris WG (2004) Lead contamination in shooting range soils from abrasion of lead bullets and subsequent weathering. Sci Total Envir 328:175–183
ISO (1993) Soil quality—determination of the effects of pollutants on soil flora—Part 1: method for the measurement of inhibition of root growth. ISO 11269-1. Genève, Switzerland
ISO (1995) Soil quality—determination of the effects of pollutants on soil flora—Part 2: effects of pollutants on the emergence and growth of higher plants. ISO 11269-2:1995. Genève, Switzerland
John R, Ahmad P, Gadgil K, Sharma S (2009) Heavy metal toxicity: effect on plant growth, biochemical parameters and metal accumulation by Brassica juncea L. Int J Plant Prod 3:65–76
Jones MGK, Outlaw WH, Lowry OH (1977) Enzymic assay of 10-7 to 10-14 moles of sucrose in plant-tissues. Plant Physiol 60:379–383
Kaznina NM, Laidinen GF, Titov AF, Talanov AV (2005) Effect of lead on the photosynthetic apparatus of annual grasses. Biol Bull 32:147–150
Kosobrukhov A, Knyazeva I, Mudrik V (2004) Plantago major plants responses to increase content of lead in soil: growth and photosynthesis. Plant Growth Regul 42:145–151
Krishna R, Dan T, Saxena P (2000) A fragrant solution to soil remediation. Int J Phytorem 2:117–132
Krupa Z, Baszynski T (1985) Requirement of galactolipids for photosystem I activity in lyophilized spinach chloroplasts. Biochim Biophys Acta 408:26–34
Kučera T, Horáková H, Šonská A (2008) Toxic metal ions in photoautotrophic organisms. Photosynthetica 46:481–489
Lee KR, Roh KS (2003) Influence of cadmium on RuBisCO activation in Canavalia ensiformis L. leaves. Biotechnol Bioproc E 8:94–100
Li J, Xie ZM, Zhu YG, Naidu R (2005) Risk assessment of heavy metal contaminated soil in the vicinity of a lead/zinc mine. J Environ Sci (China) 17:881–5
Lichtenhaler HK (1987) Chlorophylls and carotenoids: pigments of photosynthetic biomembranes. Meth Enzymol 148:350–382
Lilley RM, Walker DA (1974) Improved spectrophotometric assay for ribulose-bis-phosphate carboxylase. Biochim Biophys Acta 358:226–229
Loureiro S, Santos C, Pinto G, Costa A, Monteiro A, Soares A, Nogueira A (2006). Toxicity Assessment of Two Soils from Jales Mine (Portugal) Using Plants: Growth and Biochemical Parameters. Arch. Environ Contam Toxicol 50:182–190
Malecka A, Piechalak A, Mensinger A, Hannc A, Balalkiewicz D, Tomaszewska B (2012) Antioxidant defense system in Pisum sativum roots exposed to heavy metals (Pb, Cu, Cd, Zn). Pol J Environ Stud 2:1721–1730
Monteiro M, Santos C, Mann M, Soares A, Lopes T (2007) Evaluation of cadmium genotoxicity in Lactuca sativa L. using nuclear microsatellites. Environ Exp Bot 60(3):421–427
Monteiro MS, Santos C, Soares AMVM, Mann RM (2009) Assessment of biomarkers of cadmium stress in lettuce. Ecotox Environ Safe 72:811–818
Monteiro C, Santos C, Pinho S, Dias C (2012) Cadmium-induced cyto- and genotoxicity are organ-dependent in lettuce. Chem Res Tox 25:1423–1434
Moustakas M, Lanaras T, Symeonidis L, Karataglis S (1994) Growth and photosynthetic characteristics of field-grown Avena sativa under copper and lead stress. Photosynthetica 30:389–396
OECD (2006) Terrestrial plant test: seedling emergence and seedling growth test. OECD-208. France
OEHHA (2009) Integrated Risk Assessment Branch, office of environmental health hazard assessment. California Environmental Protection Agency, USA, 153pp
Päivöke AEA, Simola LK (2001) Arsenate toxicity to Pisum sativum: mineral nutrients, chlorophyll content, and phytase activity. Ecotoxicol Environ Saf 49:111–121
Parys E, Romanowska E, Siedlecka M, Poskuta JW (1998) The effect of lead on photosynthesis and respiration in detached leaves and in mesophyll protoplasts of Pisum sativum. Acta Physiol Plant 20:313–322
Peralta-Videa JR, Lopez ML, Narayan M, Saupe G, Gardea-Torresdey J (2009) The biochemistry of environmental heavy metal uptake by plants: implications for the food chain. Int J Biochem Cell Biol 41:1665–1677
Piechalak A, Tomaszewskaa B, Barakiewicz D (2003) Enhancing phytoremediative ability of Pisum sativum by EDTA application. Phytochemistry 64:1239–1251
Podazza G, Rosa M, Gonzalez JA, Hilal M, Prado FE (2006) Cadmium induces changes in sucrose partitioning, invertase activities, and membrane functionality in roots of Rangpur lime (Citrus limonia L. Osbeck). Plant Biol 8:706–714
Pourrut B, Shahid M, Dumat C, Winterton P, Pinelli E (2011) Lead uptake, toxicity, and detoxification in plants. Rev Environ Contam T 213:113–136
Prasad M, Strzalka S (2002) Physiology and biochemistry of metal toxicity and tolerance in plants. Kluwer Academic, Dordrecht
Prüss-Üstün A, Campbell-Lendrum D, Corvalán C, Woodward A (2003) Assessing the environmental burden of disease at national and local levels. In: Fewtrell L, Kaufmann R, Prüss-Üstün A (eds), World Health Organization, Protection of the Human Environment, Geneva
Rodriguez E, Azevedo R, Fernandes P, Santos C (2011) Cr(VI) induces DNA damage, cell cycle arrest and polyploidization. Chemical Res Toxicol 24:1040–1047
Rodriguez E, Santos C, Azevedo R, Moutinho-Pereira J, Correia C, Dias MC (2012) Chromium (VI) induces toxicity at different photosynthetic levels in pea. Plant Physiol Biochem 53:94–100
Rodriguez E, Azevedo R, Moreira H, Souto L, Santos C (2013a) Pb2+ exposure induced microsatellite instability in Pisum sativum in a locus related with glutamine metabolism. Plant Physiol Biochem 62:19–22
Rodriguez E, Azevedo R, Moreira H, Souto L, Santos C (2013b) Pb2+ exposure induced microsatellite instability in Pisum sativum in a locus related with glutamine metabolism. Plant Physiol Biochem 62:19–22
Romanowska E, Wasilewska W, Fristedt R, Vener A, Zienkiewicz M (2012) Phosphorylation of PSII proteins in maize thylakoids in the presence of Pb ions. J Plant Physiol 169(4):345–352
Ruley A, Sharma N, Sahi S, Sajwan K (2006) Effects of lead and chelators on growth, photosynthetic activity and Pb uptake in Sesbania drummondii grown in soil. Environ Pollut 144:11–18
Silva S, Pinto-Carnide O, Martins-Lopes P, Guedes-Pinto H, Santos C (2010) Differential aluminium changes on nutrient accumulation and root differentiation in an Al sensitive vs. tolerant wheat. Environ Exp Bot 68:91–98
Sims DA, Gamon JA (2002) Relationships between leaf pigment content and spectral reflectance across a wide range of species, leaf structures and developmental stages. Remote Sens Environ 81:337–354
Stitt M, Bulpin PV, Rees TA (1978) Pathway of starch breakdown in photosynthetic tissues of Pisum sativum. Biochim Biophys Acta 544:200–214
Tawinteung N, Parkpian P, DeLaune RD, Jugsujinda A (2005) Evaluation of extraction procedures for removing lead from contaminated soil. J Environ Sci Health 40(2):385–407
Tripathy BC, Bhatia B, Mohanty P (1981) Inactivation of chloroplast photosynthetic electron-transport activity by Ni2+. Biochim Biophys Acta 638:217–224
Visioli G, Marmiroli N (2013) The proteomics of heavy metal hyperaccumulation by plants. J Proteomics 79:133–45
Vojtĕchová M, Leblová S (1991) Uptake of lead and cadmium by maize seedlings and the effect of heavy-metals on the activity of phosphoenolpyruvate carboxylase isolated from maize. Biol Plantarum 33:386–394
Von Caemmerer S, Farquhar GD (1981) Some relationships between the biochemistry of photosynthesis and the gas-exchange of leaves. Planta 153:376–387
Wang C, Wang X, Tian Y, Xue Y, Xu X, Sui Y, Yu H (2008) Oxidative stress and potential biomarkers in tomato seedlings subjected to soil lead contamination. Ecotoxicol Environ Saf 71:685–691
WHO (2010) Childhood lead poisoning 2010. ISBN 978 92 4 150033 3
Yanagiya T, Imura N, Enomoto S, Kondo Y, Himeno S (2000) Suppression of a high affinity transport system for manganese in cadmium-resistant metallothionein-null cells. J Pharmacol Exp Therap 292:1080–1086
Zeng LS, Liao M, Chen CL, Huang CY (2006) Effects of lead contamination on soil microbial activity and rice physiological indices in soil-Pb-rice (Oryza sativa L.) system. Chemosphere 65(4):567–574
Acknowledgments
This work was supported by the Portuguese Foundation for Science and Technology (FCT) FCT/PTDC/AAC-AMB/112804/2009, BioRem: integration of multiple BIOmarkers of toxicity in an assay of phytoremediation in contaminated sites. FCT also supported a post-doctoral fellowship of M. C. Dias (SFRH/BPD/41700/2007), E. Rodriguez (SFRH/BPD/75696/2011), and J.M.P. Ferreira de Oliveira (SFRH/BPD/74868/2010).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Elena Maestri
Rights and permissions
About this article
Cite this article
Rodriguez, E., da Conceição Santos, M., Azevedo, R. et al. Photosynthesis light-independent reactions are sensitive biomarkers to monitor lead phytotoxicity in a Pb-tolerant Pisum sativum cultivar. Environ Sci Pollut Res 22, 574–585 (2015). https://doi.org/10.1007/s11356-014-3375-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-014-3375-9