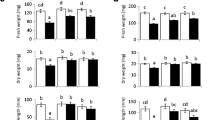
Abstract
Drought is an abiotic stress that limits plant growth and productivity, and the development of trees with improved drought tolerance is expected to expand potential plantation areas and to promote sustainable development. Previously we reported that transgenic poplars (Populus tremula × P. tremuloides, T89) harboring the stress-responsive galactinol synthase gene, AtGolS2, derived from Arabidopsis thaliana were developed and showed improved drought stress tolerance in laboratory conditions. Herein we report a field trial evaluation of the AtGolS2-transgenic poplars. The rainfall-restricted treatments on the poplars started in late May 2020, 18 months after transplanting to the field, and were performed for 100 days. During these treatments, the leaf injury levels were observed by measuring photosynthetic quantum yields twice a week. Observed leaf injury levels varied in response to soil moisture fluctuation and showed a large difference between transgenic and non-transgenic poplars during the last month. Comparison of the leaf injury levels against three stress classes clustered by the machine learning approach revealed that the transgenic poplars exhibited significant alleviation of leaf injuries in the most severe stress class. The transgenes and transcript levels were stable in the transgenic poplars cultivated in the field conditions. These results indicated that the overexpression of AtGolS2 significantly improved the drought stress tolerance of transgenic poplars not only in the laboratory but also in the field. In future studies, molecular breeding using AtGolS2 will be an effective method for developing practical drought-tolerant forest trees.





Similar content being viewed by others
Abbreviations
- GolS2:
-
Galactinol synthase 2
- RFO:
-
Raffinose family oligosaccharide
- QY:
-
Quantum yield
- ANOVA:
-
Analysis of variant
- HSD:
-
Honestly significant difference
- GMM:
-
Gaussian mixture model
References
Allen CD, Macalady AK, Chenchouni H, Bachelet D, McDowell N, Vennetier M, Kitzberger T, Rigling A, Breshears DD, Hogg EH, Gonzalez P, Fensham R, Zhang Z, Castro J, Demidova N, Lim JH, Allard G, Running SW, Semerci A, Cobb N (2010) A global overview of drought and heat-induced tree mortality reveals emerging climate change risks for forests. Forest Ecol Manag 259:660–684. https://doi.org/10.1016/j.foreco.2009.09.001
Balfagón D, Zandalinas SI, Baliño P, Muriach M, Gómez-Cadenas A (2018) Involvement of ascorbate peroxidase and heat shock proteins on citrus tolerance to combined conditions of drought and high temperatures. Plant Physiol Biochem 127:194–199. https://doi.org/10.1016/j.plaphy.2018.03.029
Cessna S, Demmig-Adams B, Adams WW (2010) Exploring photosynthesis and plant stress using inexpensive chlorophyll fluorometers. J Nat Resour Life Sci Educ 39:22–30. https://doi.org/10.4195/jnrlse.2009.0024u
Cui LH, Byun MY, Oh HG, Kim SJ, Lee J, Lee H, Kim WT, Park H (2020) Poaceae type II galactinol synthase 2 from Antarctic flowering plant Deschampsia antarctica and rice improves cold and drought tolerance by accumulation of raffinose family oligosaccharides in transgenic rice plants. Plant Cell Physiol 61:88–104. https://doi.org/10.1093/pcp/pcz180
dos Santos TB, Vieira LGE (2020) Involvement of the galactinol synthase gene in abiotic and biotic stress responses: A review on current knowledge. Plant Gene 24:100258. https://doi.org/10.1016/j.plgene.2020.100258
Egert A, Eicher B, Keller F, Peters S (2015) Evidence for water deficit-induced mass increases of raffinose family oligosaccharides (RFOs) in the leaves of three Craterostigma resurrection plant species. Front Physiol 6:1–6. https://doi.org/10.3389/fphys.2015.00206
Fang S, Xue J, Tang L (2007) Biomass production and carbon sequestration potential in poplar plantations with different management patterns. J Environ Manag 85:672–679. https://doi.org/10.1016/j.jenvman.2006.09.014
FAO (2020) Global Forest Resources Assessment 2020—Key findings, https://www.fao.org/documents/card/en/c/ca8753en
FCCC (2021) Glasgow Climate Pact. Conference of the Parties serving as the meeting of the Parties to the Paris Agreement. United Nations Framework Convention on Climate Change (FCCC), FCCC/PA/CMA/2021/L.16. https://unfccc.int/sites/default/files/resource/cma2021_L16_adv.pdf
Franco-Navarro JD, Díaz-Rueda P, Rivero-Núñez CM, Brumós J, Rubio-Casal AE, De Cires A, Colmenero-Flores JM, Rosales MA (2021) Chloride nutrition improves drought resistance by enhancing water deficit avoidance and tolerance mechanisms. J Exp Botany 72:5246–5261. https://doi.org/10.1093/jxb/erab143
Gangola MP, Jaiswal S, Kannan U, Gaur PM, Båga M, Chibbar RN (2016) Galactinol synthase enzyme activity influences raffinose family oligosaccharides (RFO) accumulation in developing chickpea (Cicer arietinum L.) seeds. Phytochemistry 125:88–98. https://doi.org/10.1016/j.phytochem.2016.02.009
Häggman H, Raybould A, Borem A, Fox T, Handley L, Hertzberg M, Lu MZ, Macdonald P, Oguchi T, Pasquali G, Pearson L, Peter G, Quemada H, Séguin A, Tattersall K, Ulian E, Walter C, McLean M (2013) Genetically engineered trees for plantation forests: Key considerations for environmental risk assessment. Plant Biotechnol J 11:785–798. https://doi.org/10.1111/pbi.12100
Handayani T, Watanabe K (2020) The combination of drought and heat stress has a greater effect on potato plants than single stresses. Plant Soil Environ 66:175–182. Doi: https://doi.org/10.17221/126/2020-PSE
Honna PT, Fuganti-Pagliarini R, Ferreira LC, Molinari MDC, Marin SRR, de Oliveira MCN, Farias JRB, Neumaier N, Mertz-Henning LM, Kanamori N, Nakashima K, Takasaki H, Urano K, Shinozaki K, Yamaguchi-Shinozaki K, Desidério JA, Nepomuceno AL (2016) Molecular, physiological, and agronomical characterization, in greenhouse and in field conditions, of soybean plants genetically modified with AtGolS2 gene for drought tolerance. Mol Breed 36:157. https://doi.org/10.1007/s11032-016-0570-z
Iseki K, Takahashi Y, Muto C, Naito K, Tomooka N (2018) Diversity of drought tolerance in the genus Vigna. Front Plant Sci 9:729. https://doi.org/10.3389/fpls.2018.00729
Jansson S, Douglas CJ (2007) Populus: A model system for plant biology. Annu Rev Plant Biol 58:435–458. https://doi.org/10.1146/annurev.arplant.58.032806.103956
Khan MS, Ahmad D, Khan MA (2015) Utilization of genes encoding osmoprotectants in transgenic plants for enhanced abiotic stress tolerance. Electronic J Biotechnol 18:257–266. https://doi.org/10.1016/j.ejbt.2015.04.002
Ko SS, Liu YC, Chung MC, Shih MC, Mohammadmehdi H, Oguchi T, Watanabe KN, Yeh KW (2019) Environmental biosafety assessment on transgenic Oncidium orchid modified by RNA interference of Phytoene Synthase genes. Plant Biotechnol 36:181–185. https://doi.org/10.5511/plantbiotechnology.19.0814a
MAFF (2013) Concerning the Application for Approval of Type 1 Use Regulations with regard to the genetically modified plants, the production or circulation of which falls within the jurisdiction of the Minister of Agriculture, Forestry and Fisheries. Notification No.8999, Food Safety and Consumer Affairs Bureau, Ministry of Agriculture, Forestry, and Fishery, Japan
Nishizawa A, Yabuta Y, Shigeoka S (2008) Galactinol and raffinose constitute a novel function to protect plants from oxidative damage. Plant Physiol 147:1251–1263. https://doi.org/10.1104/pp.108.122465
Oguchi T, Kashimura Y, Mimura M, Yu X, Matsunaga E, Nanto K, Shimada T, Kikuchi A, Watahabe KN (2014) A multi-year assessment of the environmental impact of transgenic Eucalyptus trees harboring a bacterial choline oxidase gene on biomass, precinct vegetation and the microbial community. Transgenic Res 23:767–777. https://doi.org/10.1007/s11248-014-9809-9
Peterbauer T, Richter A (2001) Biochemistry and physiology of raffinose family oligosaccharides and galactosyl cyclitols in seeds. Seed Sci Res 11:185–197. Doi: https://doi.org/10.1079/SSR200175
Peters S, Mundree SG, Thomson JA, Farrant JM, Keller F (2007) Protection mechanisms in the resurrection plant Xerophyta viscosa (Baker): both sucrose and raffinose family oligosaccharides (RFOs) accumulate in leaves in response to water deficit. J Exp Bot 58:1947–1956. https://doi.org/10.1093/jxb/erm056
Polle A, Douglas C (2010) The molecular physiology of poplars: paving the way for knowledge-based biomass production. Plant Biol 12:239–241. https://doi.org/10.1111/j.1438-8677.2009.00318.x
Polle A, Chen SL, Eckert C, Harfouche A (2019) Engineering drought resistance in forest trees. Front Plant Sci 9:1875. https://doi.org/10.3389/fpls.2018.01875
Rahman SM, Kakuda K, Sasaki Y, Makabe-Sasaki S, Ando H (2013) Early growth stage water management effects on the fate of inorganic N, growth and yield in rice. Soil Sci Plant Nutrition 59:921–932. https://doi.org/10.1080/00380768.2013.865509
Saravitz DM, Pharr DM, Carter TE (1987) Galactinol synthase activity and soluble sugars in developing seeds of four soybean genotypes. Plant Physiol 83:185–189. https://doi.org/10.1104/pp.83.1.185
Selvaraj MG, Ishizaki T, Valencia M, Ogawa S, Dedicova B, Ogata T, Yoshiwara K, Maruyama K, Kusano M, Saito K, Takahashi F, Shinozaki K, Nakashima K, Ishitani M (2017) Overexpression of an Arabidopsis thaliana galactinol synthase gene improves drought tolerance in transgenic rice and increased grain yield in the field. Plant Biotechnol J 15:1465–1477. https://doi.org/10.1111/pbi.12731
Sengupta S, Mukherjee S, Basak P, Majumder AL (2015) Significance of galactinol and raffinose family oligosaccharide synthesis in plants. Front Plant Sci 6:656. https://doi.org/10.3389/fpls.2015.00656
Shimosaka E, Ozawa K (2015) Overexpression of cold-inducible wheat galactinol synthase confers tolerance to chilling stress in transgenic rice. Breed Sci 65:363–371. https://doi.org/10.1270/jsbbs.65.363
Shinozaki K, Yamaguchi-Shinozaki K (2007) Gene networks involved in drought stress response and tolerance. J Exp Botany 58:221–227. https://doi.org/10.1093/jxb/erl164
Singh M, Kumar J, Singh S, Singh VP, Prasad SM (2015) Roles of osmoprotectants in improving salinity and drought tolerance in plants: a review. Rev Environ Sci Biotechnol 14:407–426. https://doi.org/10.1007/s11157-015-9372-8
Scrucca L, Fop M, Murphy TB, Raftery AE (2016) mclust5: clustering, classification and density estimation using Gaussian finite mixture models. The R Journal 8(1):289–317. Doi: https://doi.org/10.32614/RJ-2016-021
Taji T, Ohsumi C, Iuchi S, Seki M, Kasuga M, Kobayashi M, Yamaguchi-Shinozaki K, Shinozaki K (2002) Important roles of drought- and cold-inducible genes for galactinol synthase in stress tolerance in Arabidopsis thaliana. Plant J 29:417–426. https://doi.org/10.1046/j.0960-7412.2001.01227.x
Tran NHT, Oguchi T, Matsunaga E, Kawaoka A, Watanabe KN, Kikuchi A (2018a) Transcriptional enhancement of a bacterial choline oxidase A gene by an HSP terminator improves the glycine betaine production and salinity stress tolerance of Eucalyptus camaldulensis trees. Plant Biotechnol 35:215–224. https://doi.org/10.5511/plantbiotechnology.18.0510b
Tran NHT, Oguchi T, Matsunaga E, Kawaoka A, Watanabe KN, Kikuchi A (2018b) Environmental risk assessment of impacts of transgenic Eucalyptus camaldulensis events highly expressing bacterial Choline Oxidase A gene. Plant Biotechnol 35:393–397. https://doi.org/10.5511/plantbiotechnology.18.0831a
Tran NHT, Oguchi T, Akatsuka N, Matsunaga E, Kawaoka A, Yamada A, Ozeki Y, Watanabe KN, Kikuchi A (2019) Development and evaluation of novel salt-tolerant Eucalyptus trees by molecular breeding using an RNA-binding-protein gene derived from common ice plant (Mesembryanthemum crystallinum L.). Plant Biotechnol J 17:801–811. https://doi.org/10.1111/pbi.13016
Tran NHT, Oguchi T, Matsunaga E, Kawaoka A, Watanabe KN, Kikuchi A (2021) Evaluation of potential impacts on biodiversity of the salt-tolerant transgenic Eucalyptus camaldulensis harboring an RNA chaperonic RNA-Binding-Protein gene derived from common ice plant. Transgenic Res 30:23–34. https://doi.org/10.1007/s11248-020-00227-6
Watanabe KN, Taeb M, Okusu H (2004) Japanese controversies over transgenic crop regulation. Science 305:1572. https://doi.org/10.1126/science.1100734
Williams AP, Allen CD, Macalady AK, Griffin D, Woodhouse CA, Meko DM, Swetnam TW, Rauscher SA, Seager R, Grissino-Mayer HD, Dean JS, Cook ER, Gangodagamage C, Cai M, Mcdowell NG (2013) Temperature as a potent driver of regional forest drought stress and tree mortality. Nat Clim Chang 3:292–297. https://doi.org/10.1038/nclimate1693
WMO (2021) State of Global Climate 2021 WMO Provisional report. https://public.wmo.int/en/our-mandate/climate/wmo-statement-state-of-globalclimate
Yu X, Kikuchi A, Matsunaga E, Kawaoka A, Ebinuma H, Watanabe KN (2013) A field trial to assess the environmental biosafety of codA-transgenic Eucalyptus camaldulensis cultivation. Plant Biotechnol 30:357–363. https://doi.org/10.5511/plantbiotechnology.13.0430a
Yu X, Ohtani M, Kusano M, Nishikubo N, Uenoyama M, Umezawa T, Saito K, Shinozaki K, Demura T (2017) Enhancement of abiotic stress tolerance in poplar by overexpression of key Arabidopsis stress response genes, AtSRK2C and AtGolS2. Mol Breed 37:57. https://doi.org/10.1007/s11032-016-0618-0
Zhou J, Yang Y, Yu J, Wang L, Yu X, Ohtani M, Kusano M, Saito K, Demura T, Zhuge Q (2014) Responses of Populus trichocarpa galactinol synthase genes to abiotic stresses. J Plant Res 127:347–358. https://doi.org/10.1007/s10265-013-0597-8
Acknowledgements
We thank Ms. Kazue Mase (Univ. Tsukuba), Ms. Akiko Sato and Ms. Kayo Kitaura (RIKEN, Japan), and Ms. Ayumi Ihara and Ms. Yuki Mitsubayashi (NAIST, Japan) for their excellent technical supports. This work was supported in part by a grant from the Center for Sustainable Resource Science, RIKEN, Japan (TO, MO, TD, and KNW), and by Cooperative Research Grants of the Plant Transgenic Design Initiative Program, Gene Research Center, University of Tsukuba (Grant nos. 1524, 1828, 1928, 2028, and 2140) (TO, MO, and TD).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shikakura, Y., Oguchi, T., Yu, X. et al. Transgenic poplar trees overexpressing AtGolS2, a stress-responsive galactinol synthase gene derived from Arabidopsis thaliana, improved drought tolerance in a confined field. Transgenic Res 31, 579–591 (2022). https://doi.org/10.1007/s11248-022-00321-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11248-022-00321-x