Abstract
Galactinol synthase (GolS; EC 2.4.1.123) is a member of the glycosyltransferase eight family that catalyzes the first step in the biosynthesis pathway of the raffinose family of oligosaccharides (RFOs). The accumulation of RFOs in response to abiotic stress indicates a role for RFOs in stress adaptation. To obtain information on the roles of RFOs in abiotic stress adaptation in trees, we investigated the expression patterns of nine Populus trichocarpa GolS (PtrGolS) genes with special reference to stress responses. PtrGolS genes were differentially expressed in different organs, and the expressions of PtrGolS4 and PtrGolS6 were relatively high in all tested organs. The expression levels of all PtrGolS genes, except PtrGolS9, changed in response to abiotic stress in gene- and stress-type-specific manners. Moreover, short- and long-term stress treatments revealed that induction of PtrGolS by salt stress is obvious only in the early period of treatment (within 24 h), whereas water-deficit stress treatments continued to upregulate PtrGolS gene expression after two days of treatment, in addition to induction within 24 h of treatment. Consistent with these expression patterns, the galactinol content in leaves increased after four days of drought stress, but not under salt stress. Our findings suggest divergent roles for PtrGolS genes in abiotic stress responses in poplars.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Abiotic stresses trigger a series of responses in plants starting with stress perception, leading to activation of signaling pathways and alteration of gene expression levels, resulting in altered plant physiology, growth and development (Loescher 2010). One of the adaptation mechanisms of plant cells is the production of regulatory compounds to protect cells against the effects of osmotic, cold and abiotic stresses. These compounds are known as compatible solutes; mannitol, proline and a large number of soluble oligosaccharides, such as trehalose, raffinose and stachyose (Cuin and Shabala 2008), are examples of compatible solutes responding to abiotic stresses.
RFOs are composed of alpha-galactosyl derivatives of sucrose. The potential role of RFOs in stress tolerance has been intensively studied in seeds, particularly with respect to desiccation tolerance and longevity in the dehydrated state (Bentsink et al. 2000; Bernal-Lugo and Leopold 1992; Downie et al. 2003; Garcia et al. 2006; Peterbauer et al. 2001; Peterbauer et al. 2002; Saravitz et al. 1987). Raffinose is the most common RFO, composed of galactose, fructose and glucose and is synthesized by the donation of galactose from galactinol, a conjugate of myo-inositol and galactose, to sucrose by raffinose synthase (RAFS; EC 2.4.1.82). Subsequent additions of galactose units to raffinose result in generation of stachyose, verbascose and other RFOs. Thus, galactinol biosynthesis is likely an important step in the biosynthesis of RFOs. The amount of galactinol was reported to exhibit seasonal changes and to increase under cold conditions (Bachmann et al. 1994, Miao et al. 2007).
Galactinol is synthesized from UDP-D-galactose and myo-inositol by galactinol synthase (GolS; EC 2.4.1.123) (Liu et al. 1995; Saravitz et al. 1987). Extensive biochemical studies have characterized GolS in many plant species, including the common bugle (Ajuga reptans) (Bachmann et al. 1994), zucchini squash (Cucurbita pepo) (Liu et al. 1995; Smith et al. 1991), kidney bean (Phaseolus vulgaris) (Liu et al. 1995), soybean (Glycine max) (Riberio et al. 2000) and cucumber (Cucumis sativus) (Wakiuchi et al. 2003). In addition, molecular biological approaches have accelerated the studies of stress responses of GolS genes. Takahashi et al. (1994) reported that OsGolS mRNA accumulated in response to cold at 4 °C and to osmotic stress in rice seedlings (Oryza sativa). Seven genes belonging to the GolS family are present in the genome of Arabidopsis thaliana. Among them, AtGolS1–AtGolS13 were investigated for their response to abiotic stresses: AtGolS1 and AtGolS2 were induced by drought, salt and heat stress and AtGolS3 was upregulated by cold stress (Taji et al. 2002). GolS genes have been identified in other plant species, such as tomato (Lycopersicon esculentum) (Downie et al. 2003), Boea hygrometrica (Wang et al. 2009), coffee (Coffea arabica) (dos Santos et al. 2011), Salvia miltiorrhiza (Wang et al. 2012), maize (Zea mays) (Zhou et al. 2012), grape (Vitis vinifera) (Pillet et al. 2012) and Medicago falcate (Zhuo et al. 2012), and most GolS genes were reported to be upregulated by abiotic stress treatment. Overexpression of GolS genes increases the amounts of galactinol and raffinose with improved abiotic stress tolerance in GolS-overexpressing plants (Taji et al. 2002), suggesting that GolS genes are good targets for molecular breeding and/or engineering to improve the abiotic stress tolerance of commercial plants.
In this study, we characterized nine poplar GolS genes from Populus trichocarpa (PtrGolS1–PtrGolS9). P. tremuloides exhibits seasonal alteration in the amount of RFOs: endogenous RFO levels increase in early winter with decreasing temperatures and diminish in spring with increasing temperatures (Cox and Stushnoff 2001). Recently, two GolS isoforms have been isolated from hybrid poplar (Populus alba × grandidentata), one of which shows seasonal changes in gene expression level (Unda et al. 2012). Thus, RFO levels appear to increase via the regulation of GolS expression in cold acclimation of poplars. To further elucidate the roles of RFOs in woody plants, we performed expression analysis of PtrGolS1–PtrGolS9 with special reference to stress response. Our results reveal gene-specific responses of PtrGolS genes under different stress conditions, demonstrating diverse roles of PtrGolS genes in abiotic stress responses.
Materials and methods
Plant materials
Populus trichocarpa (Nisqually-1 strain, Tuskan et al. 2006) was used in this study. The young poplar plants were propagated and maintained aseptically on medium containing McCown’s Woody Plant Basal Salt Mixture (pH 5.6; Sigma-Aldrich) under 16-h light/8-h dark conditions at 25 °C. The poplars used for expression analysis and RFO quantification were planted in soil pots (8.5-cm diameter, 14-cm height) and grown in a greenhouse (16-h light/8-h dark, 25 °C) for two months.
Molecular cloning of PtrGolS genes
Sequence information for PtrGolS was obtained by performing a BLAST search of the P. trichocarpa genome at the Phytozome website (http://www.phytozome.net/) using the AtGolS sequences available in NCBI (http://www.ncbi.nlm.nih.gov/pubmed/). The primers were designed to amplify the coding region of PtrGolS genes (Supplementary Table 1) and used for reverse transcription-polymerase chain reaction (RT-PCR) with first strand cDNA template synthesized from total RNA derived from the leaves of poplars. The PCR products were cloned into the pMD 18-T vector (TaKaRa Japan) for sequencing. The experimentally-determined sequences of the PtrGolS genes were not identical to the PtGolS genes (Supplementary Table 2) described in Unda et al. (2012). The nine genes were submitted to GenBank and the accession numbers are KF496084, KF49608, KF496086, KF496087, KF496088, KF496089, KF496090, KF496091 and KF496092.
Phylogenetic analysis
The putative amino acid sequences were obtained using the GENESCAN program (http://genes.mit.edu/GENSCAN.html) by submitting the PtrGolS cDNA sequences obtained from the molecular cloning analysis. The phylogenetic tree was constructed by the neighbor-joining (NJ) method using the MEGA 5.0 software (Tamura et al. 2011). Bootstrap analysis was performed with 1,000 replicates to evaluate the reliability of different phylogenetic groupings. The obtained tree was drawn using the TreeView software.
In silico prediction of cis-acting elements
The 2-kb region upstream of the translation start site of each PtrGolS gene was used as the putative promoter region. Prediction of cis-acting elements was performed using the PlantCARE (http://bioinformatics.psb.ugent.be/webtools/plantcare/html/) and PLACE (http://www.dna.affrc.go.jp/PLACE/index.html) software to detect well-known abiotic stress-associated elements (ABRE, DRE/CRT and LTRE) (Maruyama et al. 2012; Tuteja 2007).
Stress and ABA plant treatments
For short-term treatments, young poplars grown on MS agar medium for one month (approximately 9 cm-height) were used. Three plants were transferred to liquid MS medium containing 200 mM NaCl (salt stress treatment), 0.2 % PEG6000 (osmotic stress treatment) or 100 μM ABA. For cold treatment, poplars grown at 25 °C were transferred to a growth chamber adjusted to 4 °C. Whole plants were collected as samples after 0, 2, 10 and 24 h of treatment for salt and osmotic stresses or 24 h of treatment for cold stress and ABA treatment, and immediately frozen in liquid nitrogen. The experiments were repeated three times.
For long-term stress treatment, plants grown in soil pots for two months (approximately 100-cm height) were used. For salt stress treatment, three plants were irrigated with 1 l of 200 mM NaCl solution every day. For drought stress treatment, the water supply was cut off. Leaves were sampled from the treated plants after 0, 2, 4 and 6 days of treatment and immediately frozen in liquid nitrogen after separation into two pieces. One piece was used for measurement of raffinose and galactinol content and the other was used for expression analysis. The experiments were repeated three times.
Quantitative RT-PCR analysis of PtrGolS genes
Total RNAs were isolated from the collected samples using the RNeasy® plant mini kit (Qiagen). To remove contaminating genomic DNA, total RNA was treated with DNase I and then mixed with an equal volume of phenol: chloroform: isoamylalcohol solution, centrifuged at 10,000 rpm, left for 10 min at RT and then the aqueous phase was transferred to a new tube. The RNA was recovered by Dr. GenTLE™ Precipitation Carrier (Takara). For complementary DNA synthesis, 2 μg of total RNA were reverse transcribed using the Transcriptor First Strand cDNA Synthesis Kit (Roche) with oligo (dT) 12–16 primer. Quantitative real-time PCR reactions consisted of 5-μl FS Universal SYBR® Green Master Mix (Roche), 5 pmol forward and reverse primers for PtrGolS1–PtrGolS9 or EFL4A genes, 0.5-μl cDNA and water to a total volume of 10 μl. The gene-specific primers were designed using the Primer 5.0 software (Supplementary Table 3). The quantitative PCR analysis was performed with a Lightcycle 480 II instrument (Roche) and the FS Universal SYBR® Green Master Mix (Roche). The reaction conditions were 50 °C for 2 min, 95 °C for 10 min, 45 cycles at 95 °C for 2 min, 62 °C for 30 s and 72 °C for 30 s. The EFL4A gene was used as an internal control. The expression ratio was calculated as 2−ΔΔCq (Bustin et al. 2009). The experiment was repeated three times.
Statistical analysis
The data were analyzed using one-way ANOVA and subsequent post hoc multiple comparison Duncan’s test or Mann–Whitney U-test using the SPSS 13.0 software.
Quantification of raffinose and galactinol content
After stress treatment, leaves were harvested and cut into pieces 5 mm in width, then immediately frozen in liquid nitrogen. Samples were subjected to gas chromatography–mass spectrophotometry (GC–MS) as described by Kusano et al. (2007), with small modifications. Samples were lyophilized using a freeze dryer (Tokyo Rikakikai). After crushing each sample in a Shake Master neo grinder (Biomedical Science) at 1,000 rpm for 5 min, approximately 5 mg (DW) of each sample were weighed and then extracted at a concentration of 2.5 mg (DW) of tissue per milliliter extraction medium (methanol/chloroform/water [3:1:1 v/v/v]) containing 10 stable isotope reference compounds. Five hundred micrograms (DW) were derived, of which 6 μg (DW) were used for GC–MS analysis.
Results
GolS genes in Populus trichocarpa
Nine GolS-related genes were identified from the poplar (P. trichocarpa) genome database (http://www.phytozome.net/search.php) by a BLAST search using A. thaliana GolS. As shown in Table 1, we named the obtained GolS sequences PtrGolS1 to -9. The open reading frames of the obtained sequences encode polypeptides of 325–338 amino acid residues. Based on the sequence information, we designed the primer sets to amplify the coding region of each gene and carried out molecular cloning and sequence analysis of the PtrGolS genes to experimentally determine the sequences of the genes. All of the predicted GolS protein sequences have the conserved domains of the glycosyl transferase 8 family; only PtrGolS5 shows a single substitution of alanine to leucine in the characteristic hydrophobic pentapeptide (APSAA) at the carboxyl terminal end (Fig. 1).
Alignment of deduced amino acid sequences of GolS proteins from P. trichocarpa (PtrGolS1–PtrGolS9) generated using the ClustalW software. Conserved residues are indicated by asterisks and similar residues by a colon or dot. The characteristic hydrophobic pentapeptide (APSAA) is located at the end of the sequence
To elucidate the phylogenic relationship between PtrGolS and known GolS proteins of other plant species, phylogenic analysis was performed using the MEGA 5.0 software. Fifteen full-length amino acid sequences from A. thaliana (AtGolS1–AtGolS7), maize (ZmGolS1–ZmGolS3), A. reptans (ArGolS1, ArGolS2), Brassica napus (BnGolS1) and Triticum aestivum (TaGolS1, TaGolS2) were obtained from GenBank. Five clades (I–V) were recognized in the phylogenetic tree (Fig. 2), although the bootstrap values for several clades were low. Branch distribution showed that PtrGolS1–PtrGolS3 (similarity of 89–91 %) grouped with AtGolS2 and AtGolS3 in clade I. PtrGolS6–PtrGolS8 (similarity of 93–98 %) clustered with AtGolS1 and BnGolS1 in clade II. ArGolS2, TaGolS1, TaGolS2, and ZmGolS1–ZmGolS3 grouped together in clade III, which is considered to be a monocot GolS family. ArGolS2 (A. reptans) had a low bootstrap value and was not considered part of this clade. PtrGolS5 was in clade IV and PtrGolS4 and PtrGolS9 (similarity of 92 %) were in clade V. PtrGolS proteins were distributed in all clades, except clade III. Thus, the sequences of PtrGolS genes were as divergent as those of other plant GolS-family genes (Philippe et al. 2010).
Phylogenetic tree of predicted amino acid sequences of PtrGolS. Unrooted phylogenetic tree of GolS was established by the neighbor-joining method. GenBank accession numbers are provided for GolS proteins other than PtrGolS. Numbers are bootstrap values (1,000 replicates). Bars 0.05 amino acid substitutions per site
Putative cis-elements in promoter regions of PtrGolS genes
To obtain information on stress-related transcriptional regulation mechanisms of PtrGolS genes, an in silico search of cis-elements was performed of their putative promoter regions. The PLACE database search showed that abiotic stress responsive elements, such as the ABA responsive element (ABRE) (Zhang et al. 2005), the dehydration and cold responsive elements (DRE/CRT) (Qin et al. 2004) and the low-temperature responsive element (LTRE) (Gao et al. 2002), are present in the promoter region of several PtrGolS genes (Table 2). PtrGolS4 does not possess any abiotic stress responsive cis-element, whereas the other PtrGolS genes all contain the ABRE element. The DRE/CRT element was found in PtrGolS1, PtrGolS3, PtrGolS8 and PtrGolS9 and LTRE was present in PtrGolS1 and PtrGolS7–PtrGolS9. These results suggest that the expression of PtrGolS genes is regulated through the corresponding cis-elements in response to stress.
Expression patterns of PtrGolS genes in organs
Total RNAs were isolated from shoot apices, young and mature leaves, stems (separated into upper and bottom regions) and roots of the soil-grown poplar plants and subjected to quantitative RT-PCR. PtrGolS4 and PtrGolS6 were most abundant in these organs, and PtrGolS3 and PtrGolS7 were relatively highly expressed in stems and roots (Fig. 3). PtrGolS expression was not abundant in shoot apices. Since the expression levels differed by gene and organ, PtrGolS genes might play different roles in the development of poplars.
Expression patterns of PtrGolS genes in organs by quantitative RT-PCR. Parts of the shoot apices, young and mature leaves, and the upper and lower regions of stems and roots were separately sampled and subjected to total RNA extraction. Results are means ± SE of three replicates. Different letters indicate statistically significant differences (post hoc multiple comparison Duncan’s test following ANOVA; p < 0.05)
Short-term responses of PtrGolS expression to abiotic stresses
To examine the stress responses of PtrGolS genes, we treated poplar plants with salt, osmotic, and cold stresses, as well as applied ABA for 24 h. As shown in Fig. 4, the responses of PtrGolS genes differed according to the stress to which they were exposed. Salt and osmotic stress treatments significantly induced the expression of PtrGolS1–PtrGolS3, PtrGolS5 and PtrGolS6, whereas cold treatment induced only PtrGolS2 and PtrGolS8 (Fig. 4). PtrGolS2 and PtrGolS5–PtrGolS7 were upregulated by ABA treatment; however, the expression levels were lower than those induced by stress treatment. Notably, PtrGolS4 and PtrGolS9 were not induced by any stresses and PtrGolS4 expression was decreased by the treatments (Fig. 4).
Expression analysis of PtrGolS genes in response to salt, osmotic or cold stress and ABA treatment. Poplar plants were cultured in medium containing 200 mM NaCl (salt stress), 0.2 % PEG6000 (osmotic stress) or 100 μM ABA for 24 h. Cold stress treatment was performed by transferring plants to a growth chamber adjusted to 4 °C. Quantitative RT-PCR was performed using total RNA extracted from whole parts of treated plants. Results are means ± SD of three replicates. Different letters indicate statistically significant differences (post hoc multiple comparison Duncan’s test following ANOVA; p < 0.05)
Next we examined the early response of PtrGolS expression to salt and osmotic stress (Fig. 5). In the salt stress treatment, PtrGolS genes other than PtrGolS4 and PtrGolS9 were transiently expressed and peaked at 2 h (PtrGolS5–PtrGolS7) or at 10 h (PtrGolS1–PtrGolS3 and PtrGolS8) (Fig. 5a). The expression levels decreased at 24 h of salt treatment (Fig. 5a). Thus, the early responses of PtrGolS genes to salt stress are completed within 24 h. In contrast, the expression patterns of PtrGolS in response to osmotic stress differed from those to salt stress (Fig. 5b). PtrGolS7 and PtrGolS9 showed no changes in expression level by the osmotic stress treatment. Other PtrGolS genes exhibited two types of response: a rapid expression response that resulted in increases in mRNA accumulation at 2 h (PtrGolS3, PtrGolS4, PtrGolS6 and PtrGolS8), and subsequent upregulation of gene expression at 24 h (PtrGolS1–PtrGolS3, PtrGolS5 and PtrGolS6). PtrGolS3 and PtrGolS6 showed both types of responses (Fig. 5b). Interestingly, PtrGolS4 mRNA was transiently induced by osmotic stress with a peak at 10 h (Fig. 5b), although PtrGolS4 expression decreased gradually during salt stress treatment (Fig. 5a). Our data demonstrated that all PtrGolS genes, except PtrGolS9, change their expression level in response to abiotic stresses, and the expression patterns differ according to stress type.
Expression patterns of PtrGolS genes under salt or osmotic stress. Poplar plants were cultured in liquid medium containing 200 mM NaCl (a) or 0.2 % PEG6000 (b) for 2, 10 and 24 h. Quantitative RT-PCR was performed using total RNA extracted from whole parts of treated plants. Results are means ± SE of three replicates. Different letters indicate statistically significant differences (post hoc multiple comparison Duncan’s test following ANOVA; p < 0.05)
Changes in galactinol and raffinose contents and PtrGolS expression patterns under long-term stress treatments
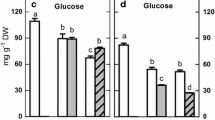
Finally, we monitored changes in the amounts of galactinol and raffinose and the expression patterns of PtrGolS under long-term stress treatment. Plants grown in soil pots were subjected to salt stress by feeding a NaCl solution or drought stress by cutting the water supply, and leaves were sampled after 0, 2, 4 and 6 days. In the salt stress treatment, the leaves turned yellow on the fourth day of treatment while in the drought stress treatment, plants wilted after the fourth day of treatment. Galactinol accumulation clearly increased under drought stress, but not under salt stress, whereas raffinose increased under both stresses (Fig. 6a). The increases in galactinol and raffinose were observed by 4 days of treatment; the raffinose content subsequently decreased (Fig. 6a). mRNA accumulation of PtrGolS genes other than PtrGolS8 and PtrGolS9 was detected under salt stress, and PtrGolS2, PtrGolS3 and PtrGolS6 reached a peak on the fourth day (Fig. 6b). PtrGolS4 was highly expressed but downregulated by salt stress (Fig. 6b). In contrast, PtrGolS genes other than PtrGolS4 were upregulated by drought stress treatment (Fig. 6c). It is suggested that all PtrGolS genes, except PtrGolS4, may contribute to the accumulation of galactinol and raffinose under drought stress.
Changes in galactinol and raffinose content and PtrGolS gene expression in leaves under long-term salt or drought stress. Plants grown in soil pots for three weeks were subjected to salt or drought stress; leaves were sampled at 0, 2, 4 and 6 days after t he start of treatment. a Galactinol and raffinose content determined by GC–MS. Asterisks indicate statistically significant increases compared with the content on day 0 (Mann–Whitney U-test; p < 0.05). (b and c) Results of quantitative RT-PCR analysis of PtrGolS genes under salt (b) or drought (c) stress. Results are means ± SE of three replicates. Different letters in b and c indicate statistically significant differences (post hoc multiple comparison Duncan’s test following ANOVA; p < 0.05)
Discussion
Many GolS genes have been reported to be induced by abiotic stresses, such as salt, drought and cold (OsGolS, Takahashi et al. 1994; ArGolS1 and ArGolS2, Sprenger and Keller 2000; AtGolS1–AtGolS3, Taji et al. 2002; MsGolS, Cunningham et al. 2003; AmGolS, Cao et al. 2009), or by biotic stress (CsGolS1, Kim et al. 2008; PtdGolS1, PtdGolS2, PtdGolS6 and PtGolS3, Philippe et al. 2010). GolS proteins expressed during seed development are considered to be related to the acquisition of desiccation stress tolerance (Downie et al. 2003). Moreover, GolS is proposed to play distinct physiological roles, synthesizing RFOs for storage and translocating carbon (Sprenger and Keller 2000).
In this paper, we assessed nine putative GolS genes from P. trichocarpa, PtrGolS1–PtrGolS9 (Table 1). The phylogenetic analysis showed that PtrGolS proteins are distributed in four clades, suggesting that PtrGolS genes evolved from four ancestors (Fig. 2; Philippe et al. 2010). Several PtrGolS genes were arrayed in pairs (PtrGolS1 and PtrGolS2, PtrGolS7 and PtrGolS8, PtrGolS4 and PtrGolS9) (Fig. 2), following the idea of multiple gene duplications in the Populus lineage (Tuskan et al. 2006). The results of our expression analysis revealed that the PtrGolS genes were differentially expressed in an organ-specific manner. The expressions of PtrGolS4 and PtrGolS6 were relatively high in all tested organs, while PtrGolS9 was transcribed only in mature leaves at a low level. Other PtrGolS genes were preferentially expressed in stems (PtrGolS1–PtrGolS3, PtrGolS5, PtrGolS7 and PtrGolS8) and/or roots (PtrGolS3 and PtrGolS7) (Fig. 3). Because GolS is involved in RFO synthesis for storage or transport (Sprenger and Keller 2000), PtrGolS expression in mature leaves (PtrGolS4 and PtrGolS6) might function in the synthesis of storage RFOs. The PtrGolS expression profiles under normal conditions appear to be independent of their distribution in the phylogenetic tree (Figs. 2, 3).
Supporting the possibility that PtrGolS is stress-related, multiple stress-related cis-elements are detected in the putative promoter regions of PtrGolS genes, except for PtrGolS4 (Table 2). The expression levels of PtrGolS1–PtrGolS8 are significantly influenced by abiotic stress, and each PtrGolS responded differently according to different types of stresses (Figs. 4, 5, 6). Almost all PtrGolS genes responded to salt, osmotic and drought stresses, whereas cold stress treatment induced only two PtrGolS genes, PtrGolS2 and PtrGolS8 (Fig. 4). Thus, PtrGolS2 and PtrGolS8 might be associated with cold acclimation and seasonal mobilization of carbohydrates, as suggested for GolS from hybrid poplar (Unda et al. 2012). Interestingly, the expression patterns of PtrGolS4 under stress conditions were distinct from those of the others: PtrGolS4 mRNA was decreased by 24-h stress treatment and ABA, although the other PtrGolS genes were induced (Fig. 4). Further analysis revealed transient upregulation of PtrGolS4 by osmotic stress during the early treatment period (Fig. 4). These unique patterns for PtrGolS4 might be related to the fact that no cis-elements were found in its promoter region (Table 2). Furthermore, for all PtrGolS, the impact of ABA on expression was smaller than that of the stress treatments (Fig. 4), suggesting that the PtrGolS genes are also controlled through an ABA-independent pathway during stress responses in poplars.
Regarding long-term stress treatments, galactinol levels were increased only under drought conditions, while raffinose levels were increased under both salt and drought stress conditions (Fig. 6a). The increases in galactinol and raffinose were observed after 4 days of treatment. Consistent with the changes in galactinol content, the expression level of PtrGolS was relatively low under salt stress (Fig. 6b), although the induced expression of PtrGolS was higher during the early period of salt stress treatment (Fig. 5a). These findings suggest that the expression of PtrGolS under salt stress is sufficient to supply galactinol for raffinose biosynthesis, but would not produce excess amounts of galactinol (Fig. 6). Since the expression levels of PtrGolS3, PtrGolS4 and PtrGolS6 appeared to be relatively high among PtrGolS genes, even though PtrGolS4 was downregulated, they may be major regulators of galactinol synthesis under salt stress conditions. In the case of drought stress, all PtrGolS genes other than PtrGolS4 were relatively highly upregulated, as compared to salt stress (Fig. 6b, c). Thus, under drought stress, the upregulation of PtrGolS would result in a significant increase in the amount of galactinol.
In conclusion, we propose different roles for PtrGolS genes in stress responses in poplars: PtrGolS2 and PtrGolS8 are involved in cold acclimation (Fig. 4) and PtrGolS3, PtrGolS4 and PtrGolS6 mainly contribute to galactinol production under long-term salt-deficit conditions (Fig. 6), whereas all PtrGolS genes would function in galactinol production under long-term water-deficit conditions (Fig. 6). Rapid responses to salt and osmotic stresses were also detected for other PtrGolS genes (Fig. 5), which suggests the involvement of other PtrGolS genes in salt- and water-deficit responses, particularly early responses. Many studies have reported that GolS isozymes differ in their enzymatic properties as well as physiological conditions for their activities (Bachmann et al. 1994; Liu et al. 1995; Riberio et al. 2000; Unda et al. 2012). Further characterization of PtrGolS proteins is thus required to elucidate the roles of GolS in RFO biosynthesis in response to abiotic stresses.
Abbreviations
- ABA:
-
Abscisic acid
- ABRE:
-
ABA responsive element
- DRE/CRT:
-
Dehydration and cold responsive element
- GolS:
-
Galactinol synthase
- LTRE:
-
Low-temperature responsive element
- MS:
-
Murashige and Skoog
- NJ:
-
Neighbor-joining
- RT-PCR:
-
Reverse transcription-polymerase chain reaction
- RFOs:
-
Raffinose family of oligosaccharides
References
Bachmann M, Matile P, Keller F (1994) Metabolism of the raffinose family oligosaccharides in leaves of Ajuga reptans L. (cold acclimation, translocation, and sink to source transition: discovery of chain elongation enzyme). Plant Physiol 105:1335–1345
Bentsink L, Alonso-Blanco C, Vreugdenhil D, Tesnier K, Groot SP, Koornneef M (2000) Genetic analysis of seed-soluble oligosaccharides in relation to seed storability of Arabidopsis. Plant Physiol 124:1595–1604
Bernal-Lugo I, Leopold AC (1992) Changes in soluble carbohydrates during seed storage. Plant Physiol 98:1207–1210
Bustin SA, Benes V, Garson JA, Hellemans J, Huggett J, Kubista M, Mueller R, Nolan T, Pfaffl MW, Shipley GL, Jo V, Carl TW (2009) The MIQE guidelines: minimum information for publication of quantitative real-time PCR experiments. Clin Chem 55:611–622
Cao P, Song J, Zhou C, Weng M, Liu J, Wang F, Zhao F, Feng D, Wang B (2009) Characterization of multiple cold induced genes from Ammopiptanthus mongolicus and functional analyses of gene AmEBP1. Plant Mol Biol 69:529–539
Cox SE, Stushnoff C (2001) Temperature-related shifts in soluble carbohydrate content during dormancy and cold acclimation in Populus tremuloides. Can J Forest Res 31:730–737
Cuin TA, Shabala S (2008) Compatible solutes mitigate damaging effects of salt stress by reducing the impact of stress-induced reactive oxygen species. Plant Signal Behav 3:207–208
Cunningham SM, Nadeau P, Castonguay Y, Laberge S, Volenec JJ (2003) Raffinose and stachyose accumulation, galactinol synthase expression, and winter injury of contrasting alfalfa germplasms. Crop Sci 43:562–570
dos Santos TB, Budzinski IG, Marur CJ, Petkowicz CL, Pereira LF, Vieira LG (2011) Expression of three galactinol synthase isoforms in Coffea arabica L. and accumulation of raffinose and stachyose in response to abiotic stresses. Plant Physiol Biochem 49:441–448
Downie B, Gurusinghe S, Dahal P, Thacker RR, Snyder JC, Nonogaki H, Yim K, Fukanaga K, Alvarado V, Bradford KJ (2003) Expression of a GALACTINOL SYNTHASE gene in tomato seeds is up-regulated before maturation desiccation and again after imbibition whenever radicle protrusion is prevented. Plant Physiol 131:1347–1359
Gao MJ, Allard G, Byass L, Flanagan AM, Singh J (2002) Regulation and characterization of four CBF transcription factors from Brassica napus. Plant Mol Biol 49:459–471
Garcia IS, Souza A, Barbedo CJ, Dietrich SM, Figueiredo-Ribeiro RC (2006) Changes in soluble carbohydrates during storage of Caesalpinia echinata LAM. (Brazilwood) seeds, an endangered leguminous tree from the Brazilian Atlantic Forest. Braz J Biol 66:739–745
Kim MS, Cho SM, Kang EY, Im YJ, Hwangbo H, Kim YC, Ryu CM, Yang KY, Chung GC, Cho BH (2008) Galactinol is a signaling component of the induced systemic resistance caused by Pseudomonas chlororaphis O6 root colonization. Mol Plant Microbe Interact 21:1643–1653
Kusano M, Fukushima A, Kobayashi M, Hayashi N, Jonsson P, Moritz T, Ebana K, Saito K (2007) Application of a metabolomic method combining one-dimensional and two-dimensional gas chromatography-time-of-flight/mass spectrometry to metabolic phenotyping of natural variants in rice. J Chromatogr B Analyt Technol Biomed Life Sci 855:71–79
Liu JJ, Odegard W, de Lumen BO (1995) Galactinol synthase from kidney bean cotyledon and zucchini leaf. Purification and N-terminal sequences. Plant Physiol 109:505–511
Loescher W (2010) Abiotic stress adaptation in plants: physiological, molecular and genomic foundation. Choice Cur Rev Acad Lib 48:713
Maruyama K, Todaka D, Mizoi J, Yoshida T, Kidokoro S, Matsukura S, Takasaki H, Sakurai T, Yamamoto YY, Yoshiwara K, Kojima M, Sakakibara H, Shinozaki K, Yamaguchi-Shinozaki K (2012) Identification of cis-acting promoter elements in cold- and dehydration-induced transcriptional pathways in Arabidopsis, rice, and soybean. DNA Res 19:37–49
Miao M, Xu X, Chen X, Xue L, Cao B (2007) Cucumber carbohydrate metabolism and translocation under chilling night temperature. J Plant Physiol 164:621–628
Peterbauer T, Lahuta LB, Blochl A, Mucha J, Jones DA, Hedley CL, Gorecki RJ, Richter A (2001) Analysis of the raffinose family oligosaccharide pathway in pea seeds with contrasting carbohydrate composition. Plant Physiol 127:1764–1772
Peterbauer T, Mach L, Mucha J, Richter A (2002) Functional expression of a cDNA encoding pea (Pisum sativum L.) raffinose synthase, partial purification of the enzyme from maturing seeds, and steady-state kinetic analysis of raffinose synthesis. Planta 215:839–846
Philippe RN, Ralph SG, Mansfield SD, Bohlmann J (2010) Transcriptome profiles of hybrid poplar (Populus trichocarpa × deltoides) reveal rapid changes in undamaged, systemic sink leaves after simulated feeding by forest tent caterpillar (Malacosoma disstria). New Phytol 188:787–802
Pillet J, Egert A, Pieri P, Lecourieux F, Kappel C, Charon J, Gomes E, Keller F, Delrot S, Lecourieux D (2012) VvGOLS1 and VvHsfA2 are involved in the heat stress responses in grapevine berries. Plant Cell Physiol 53:1776–1792
Qin F, Sakuma Y, Li J, Liu Q, Li YQ, Shinozaki K, Yamaguchi-Shinozaki K (2004) Cloning and functional analysis of a novel DREB1/CBF transcription factor involved in cold-responsive gene expression in Zea mays L. Plant Cell Physiol 45:1042–1052
Riberio M, Carlos RF, Silenede PL (2000) Soybean seed galactinol synthase activity as determined by a novel colorimetric assay. R Bras Fisiol Veg 12:203–212
Saravitz DM, Pharr DM, Carter TE (1987) Galactinol synthase activity and soluble sugars in developing seeds of four soybean genotypes. Plant Physiol 83:185–189
Smith PT, Kuo TM, Crawford CG (1991) Purification and characterization of galactinol synthase from mature zucchini squash leaves. Plant Physiol 96:693–698
Sprenger N, Keller F (2000) Allocation of raffinose family oligosaccharides to transport and storage pools in Ajuga reptans: the roles of two distinct galactinol synthases. Plant J 21:249–258
Taji T, Ohsumi C, Iuchi S, Seki M, Kasuga M, Kobayashi M, Yamaguchi-Shinozaki K, Shinozaki K (2002) Important roles of drought- and cold-inducible genes for galactinol synthase in stress tolerance in Arabidopsis thaliana. Plant J 29:417–426
Takahashi R, Joshee N, Kitagawa Y (1994) Induction of chilling resistance by water stress, and cDNA sequence analysis and expression of water stress regulated genes in rice. Plant Mol Biol 26:339–352
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum-likelihood, evolutionary distance, and maximum-parsimony methods. Mol Biol Evol 28:2731–2739
Tuskan GA, Difazio S, Jansson S, Bohlmann J, Grigoriev I, Hellsten U, Putnam N, Ralph S, Rombauts S, Salamov A, Schein J, Sterck L, Aerts A, Bhalerao RR, Bhalerao RP, Blaudez D, Boerjan W, Brun A, Brunner A, Busov V, Campbell M, Carlson J, Chalot M, Chapman J, Chen GL, Cooper D, Coutinho PM, Couturier J, Covert S, Cronk Q, Cunningham R, Davis J, Degroeve S, Dejardin A, Depamphilis C, Detter J, Dirks B, Dubchak I, Duplessis S, Ehlting J, Ellis B, Gendler K, Goodstein D, Gribskov M, Grimwood J, Groover A, Gunter L, Hamberger B, Heinze B, Helariutta Y, Henrissat B, Holligan D, Holt R, Huang W, Islam-Faridi N, Jones S, Jones-Rhoades M, Jorgensen R, Joshi C, Kangasjarvi J, Karlsson J, Kelleher C, Kirkpatrick R, Kirst M, Kohler A, Kalluri U, Larimer F, Leebens-Mack J, Leple JC, Locascio P, Lou Y, Lucas S, Martin F, Montanini B, Napoli C, Nelson DR, Nelson C, Nieminen K, Nilsson O, Pereda V, Peter G, Philippe R, Pilate G, Poliakov A, Razumovskaya J, Richardson P, Rinaldi C, Ritland K, Rouze P, Ryaboy D, Schmutz J, Schrader J, Segerman B, Shin H, Siddiqui A, Sterky F, Terry A, Tsai CJ, Uberbacher E, Unneberg P, Vahala J, Wall K, Wessler S, Yang G, Yin T, Douglas C, Marra M, Sandberg G, Van de Peer Y, Rokhsar D (2006) The genome of black cottonwood, Populus trichocarpa (Torr. and Gray). Science 313:1596–1604
Tuteja N (2007) Abscisic Acid and abiotic stress signaling. Plant Signal Behav 2:135–138
Unda F, Canam T, Preston L, Mansfield SD (2012) Isolation and characterization of galactinol synthases from hybrid poplar. J Exp Bot 63:2059–2069
Wakiuchi N, Shiomi R, Tamaki H (2003) Production of galactinol from sucrose by plant enzymes. Biosci Biotechnol Biochem 67:1465–1471
Wang Z, Zhu Y, Wang L, Liu X, Liu Y, Phillips J, Deng X (2009) A WRKY transcription factor participates in dehydration tolerance in Boea hygrometrica by binding to the W-box elements of the galactinol synthase (BhGolS1) promoter. Planta 230:1155–1166
Wang D, Yao W, Song Y, Liu W, Wang Z (2012) Molecular characterization and expression of three galactinol synthase genes that confer stress tolerance in Salvia miltiorrhiza. J Plant Physiol 169:1838–1848
Zhang W, Ruan J, Ho TH, You Y, Yu T, Quatrano RS (2005) Cis-regulatory element based targeted gene finding: genome-wide identification of abscisic acid- and abiotic stress-responsive genes in Arabidopsis thaliana. Bioinformatics 21:3074–3081
Zhou ML, Zhang Q, Zhou M, Sun ZM, Zhu XM, Shao JR, Tang YX, Wu YM (2012) Genome-wide identification of genes involved in raffinose metabolism in Maize. Glycobiology 22:1775–1785
Zhuo C, Wang T, Lu S, Zhao Y, Li X, Guo Z (2012) A cold responsive galactinol synthase gene from Medicago falcata (MfGolS1) is induced by myo-inositol and confers multiple tolerances to abiotic stresses. Physiol Plant. doi:10.1111/ppl.12019
Acknowledgments
We thank Ms. Ayumi Ihara, Ms. Arika Takebayashi and Mr. Makoto Kobayashi (RIKEN) for their technical assistance and Ms. Akiko Sato and Ms. Kayo Kitaura (RIKEN) for propagating the poplar trees. This work was supported by the Forestry Public Benefic Research Program (No. 201304102), the National 863 Program of China (No. 2013AA102703), the National Science Foundation of China (No. 30571518), the Graduate Innovative Project of Jiangsu Province (CXZZ11_0506), the Doctoral Degree Thesis Innovation Foundation of Nanjing Forestry University (No. 2011YB010), the Priority Academic Program Development of Jiangsu Higher Education Institutions, the Program for Innovative Research Team in University of Educational Department and Jiangsu Province, China, the RIKEN Biomass Engineering Program and the RIKEN International Program Associate program.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Zhou, J., Yang, Y., Yu, J. et al. Responses of Populus trichocarpa galactinol synthase genes to abiotic stresses. J Plant Res 127, 347–358 (2014). https://doi.org/10.1007/s10265-013-0597-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10265-013-0597-8