Abstract
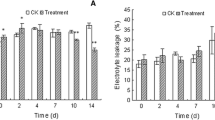
Hyperhydricity can cause significant economic loss for the micro-propagation industry that produces blueberry. In order to predict and control the occurrence of hyperhydricity, better understanding of the anatomical and physiological features of hyperhydric plantlets is required. In this study, we investigated the ultrastructural and physiological changes associated with hyperhydric blueberry plantlets. Compared to normal plantlets, hyperhydric plantlets exhibited reduced cell wall thickness, damaged membrane and guard cell structure, decreased number of mitochondria and starch granule, higher cell vacuolation, more intercellular spaces, and collapse of vascular tissues. In addition, excessive accumulation of reactive oxygen species (ROS) and ethylene, decreased stomatal aperture and water loss, as well as abnormity of stomatal movement were also evident in the hyperhydric plantlets. The results suggested that excessive ethylene and ROS produced in response to the stress arising from in vitro culture could lead to abnormal stomatal closure, causing the accumulation of water in the tissues. This would lead to subsequent induction of oxidative stress (due to hypoxia) and cell damage, especially guard cell structure, eventually giving rise to the symptoms of hyperhydricity. Reducing the content of ethylene and ROS, and protecting the structure and function of the stomata could be considered as potential strategies for inhibiting hyperhydricity or restoring the hyperhydric plants to their normal state.









Similar content being viewed by others
References
Acharya BR, Assmann SM (2009) Hormone interactions in stomatal function. Plant Mol Biol 69(4):451–462
Apóstolo NM, Llorente BE (2000) Anatomy of normal and hyperhydric leaves and shoots of in vitro grown Simmondsia chinesis (Link) Schn. In Vitro Cell Dev 36(4):243–249
Bais HP, Sudha GS, Ravishankar GA (2000) Putrescine and silver nitrate influences shoot multiplication, in vitro flowering and endogenous titers of polyamines in Cichorium intybus L. cv. Lucknow local. J Plant Growth Regul 19:238–248
Basu A, Rhone M, Lyons TJ (2010) Berries: emerging impact on cardiovascular health. Nutr Rev 68(3):168–177
Cao X, Fordham I, Douglass L, Hammerschlag F (2003) Sucrose level influences micropropagation and gene delivery into leaves from in vitro propagated highbush blueberry shoots. Plant Cell Tissue Organ Cult 75(3):255–259
Cassells AC, Curry RF (2001) Oxidative stress and physiological, epigenetic and genetic variability in plant tissue culture: implications for micropropagators and genetic engineers. Plant Cell Tissue Organ Cult 64(2–3):145–157
Casson SA, Hetherington AM (2010) Environmental regulation of stomatal development. Curr Opin Plant Biol 13(1):90–95
Chakrabarty D, Park S, Ali M, Shin K, Paek K (2006) Hyperhydricity in apple: ultrastuctural and physiological aspects. Tree Physiol 26(3):377–388
Dimasi-Theriou K, Economou AS, Sfakiotakis EM (1993) Promotion of petunia (Petunia hybrida L.) regeneration in vitro by ethylene. Plant Cell Tissue Organ Cult 32:219–225
El-Shiekh A, Wildung DK, Luby JJ, Sargent KL, Read PE (1996) Long-term effects of propagation by tissue culture or softwood single-node cuttings on growth habit, yield, and berry weight of ‘Northblue’ blueberry. J Am Soc Hortic Sci 121(2):339–342
Fal M, Majada J, Gonzalez A, Tamés RS (1999) Differences between Dianthus caryophyllus L. cultivar in in vitro growth and morphogenesis are related to their ethylene production. Plant Growth Regul 27:131–136
Fan S, Jian D, Wei X, Chen J, Beeson RC, Zhou Z, Wang X (2017) Micropropagation of blueberry ‘Bluejay’ and ‘Pink Lemonade’ through in vitro shoot culture. Sci Hortic 226:277–284
Feng JC, Yu X, Shang X, Li J, Wu Y (2010) Factors influencing efficiency of shoot regeneration in Ziziphus jujuba Mill. ‘Huizao’. Plant Cell Tissue Organ Cult 101:111–117
Fontes MA, Otoni WC, Carolino SMB, Brommonschenkel SH, Fontes EPB, Fári M, Louro RP (1999) Hyperhydricity in pepper plants regenerated in vitro: involvement of BiP (binding protein) and ultrastructural aspects. Plant Cell Rep 19(1):81–87
Franck T, Kevers C, Gaspar T, Dommes J, Deby C, Greimers R, Serteyn D, Deby Dupont G (2004) Hyperhydricity of Prunus avium shoots cultured on gelrite: acontrolled stress response. Plant Physiol Biochem 42:519–527
Gao H, Xia X, An L, Xin X, Liang Y (2017) Reversion of hyperhydricity in pink (Dianthus chinensis L.) plantlets by AgNO3 and its associated mechanism during in vitro culture. Plant Sci 254:1–11
Ge XM, Cai HL, Lei X, Zhou X, Yue M, He JM (2015) Heterotrimeric G protein mediates ethylene-induced stomatal closure via hydrogen peroxide synthesis in Arabidopsis. Plant J 82(1):138–150
Hassannejad S, Bernard F, Mirzajani F, Gholami M (2012) SA improvement of hyperhydricity reversion in Thymus daenensis shoots culture may be associated with polyamines changes. Plant Physiol Biochem 51:40–46
Inskeep WP, Bloom PR (1985) Extinction coefficients of chlorophyll a and b in N,N-dimethylformamide and 80% acetone. Plant Physiol 77(2):483–485
Ivanova M, Staden JV (2010) Natural ventilation effectively reduces hyperhydricity in shoot cultures of Aloe polyphylla Schönland ex Pillans. Plant Growth Regul 60(2):143–150
Ivanova M, Staden JV (2011) Influence of gelling agent and cytokinins on the control of hyperhydricity in Aloe polyphylla. Plant Cell Tissue Organ Cult 104(1):13–21
Jaspers P, Kangasjärvi J (2010) Reactive oxygen species in abiotic stress signaling. Physiol Plant 138(4):405–413
Jausoro V, Llorente BE, Apóstolo NM (2010) Structural differences between hyperhydric and normal in vitro shoots of Handroanthus impetiginosus (Mart. ex DC) Mattos (Bignoniaceae). Plant Cell Tissue Organ Cult 101(2):183–191
Jia W, Zhang J (2008) Stomatal movements and long-distance signaling in plants. Plant Signal Behav 3(10):772–777
Kevers C, Gaspar T (1985) Vitrification of carnation in vitro: changes in ethylene production, ACC level and capacity to convert ACC to ethylene. Plant Cell Tissue Organ Cult 4(3):215–223
Kevers C, Prat R, Gaspar T (1987) Vitrification of carnation in vitro: changes in cell wall mechanical properties, cellulose and lignin content. Plant Growth Regul 5(1):59–66
Kevers C, Franck T, Strasser RJ, Dommes J, Gaspar T (2004) Hyperhydricity of micropropagated shoots: a typically stress-induced change of physiological state. Plant Cell Tissue Organ Cult 77(2):181–191
Lai CC, Lin HM, Nalawade SM, Fang W, Tsay HS (2005) Hyperhydricity in shoot cultures of Scrophularia yoshimurae can be effectively reduced by ventilation of culture vessels. J Plant Physiol 162(3):355–361
Lakshmanan P, Lee C, Goh C (1997) An efficient in vitro method for mass propagation of a woody ornamental Ixora coccinea L. Plant Cell Rep 16:572–577
Li T, Yun Z, Zhang D, Yang C, Hong Z, Jiang Y, Duan X (2015) Proteomic analysis of differentially expressed proteins involved in ethylene-induced chilling tolerance in harvested banana fruit. Front Plant Sci 6(845):845
Louro RP, Dos Santos AV, Machado RD (1999) Ultrastructure of Eucalyptus grandis × Eucalyptus urophylla. I. shoots cultivated in vitro in multiplication and elongation-rooting media. Int J Plant Sci 160(160):217–227
Mayor M, Nestares G, Zorzoli R, Picardi L (2003) Reduction of hyperhydricity in sunflower tissue culture. Plant Cell Tissue Organ Cult 72(1):99–103
Mensuali-Sodi A, Panizza M, Serra G, Tognoni F (1993) Involvement of activated charcoal in the modulation of abiotic and biotic ethylene levels in tissue cultures. Sci Hortic 54:49–57
Neill S, Desikan R, Hancock J (2002) Hydrogen peroxide signaling. Curr Opin Plant Biol 5(5):388–395
Neto CC (2007) Cranberry and blueberry: evidence for protective effects against cancer and vascular diseases. Mol Nutr Food Res 51(6):652–664
Olmos E, Hellín E (1998) Ultrastructural differences of hyperhydric and normal leaves from regenerated carnation plants. Sci Hortic 75(1):91–101
Picoli EA, Otoni WC, Figueira ML, Carolino S, Almeida RS, Silva EA, Carvalho CR, Fontes EP (2001) Hyperhydricity in in vitro eggplant regenerated plants: structural characteristics and involvement of BiP (binding protein). Plant Sci 160(5):857–868
Picoli E, Paiva EAS, Xavier A, Aguiar RM, Carolino SM, Otoni WC (2008) Ultrastructural and biochemical aspects of normal and hyperhydric eucalypt. J Hortic Sci 14(3):61–69
Rogiers SY, Hardie WJ, Smith JP (2011) Stomatal density of grapevine leaves (Vitis vinifera L.) responds to soil temperature and atmospheric carbon dioxide. Aust J Grape Wine Res 17(2):147–152
Rojas-Martinez L, Visser RG, de Klerk GJ (2010) The hyperhydricity syndrome: waterlogging of plant tissues as a major cause. Propag Ornam Plants 10(4):169–175
Rowland LJ, Alkharouf N, Darwish O, Ogden EL, Polashock JJ, Bassil NV, Main D (2012) Generation and analysis of blueberry transcriptome sequences from leaves, developing fruit, and flower buds from cold acclimation through deacclimation. BMC Plant Biol 12(1):46
Saher S, Piqueras A, Hellin E, Olmos E (2004) Hyperhydricity in micropropagated carnation shoots: the role of oxidative stress. Physiol Plant 120(1):152–161
Saher S, Piqueras A, Hellin E, Olmos E (2005) Prevention of hyperhydricity in micropropagated carnation shoots by bottom cooling: implications of oxidative stress. Plant Cell Tissue Organ Cult 81(2):149–158
Sarkar D, Sud KC, Chakrabarti SK, Naik PS (2002) Growing of potato microplants in the presence of alginate-silverthiosulfate capsules reduces ethylene-induced culture abnormalities during minimal growth conservation in vitro. Plant Cell Tissue Organ Cult 68:79–89
Song Y, Miao Y, Song CP (2014) Behind the scenes: the roles of reactive oxygen species in guard cells. New Phytol 201(4):1121–1140
Soundararajan P, Manivannan A, Cho YS, Jeong BR (2017) Exogenous supplementation of silicon improved the recovery of hyperhydric shoots in Dianthus caryophyllus L. by stabilizing the physiology and protein expression. Front Plant Sci 8:738
Stepanova AN, Alonso JM (2009) Ethylene signaling and response: where different regulatory modules meet. Curr Opin Plant Biol 12(5):548–555
Sun X, Zhao T, Gan S, Ren X, Fang L, Karungo SK, Wang Y, Chen L, Li S, Xin H (2016) Ethylene positively regulates cold tolerance in grapevine by modulating the expression of ETHYLENE RESPONSE FACTOR 057. Sci Rep 6:24066
Tabart J, Franck T, Kevers C, Dommes J (2015) Effect of polyamines and polyamine precursors on hyperhydricity in micropropagated apple shoots. Plant Cell Tissue Organ Cult 120:11–18
Tian J, Jiang F, Wu Z (2015) The apoplastic oxidative burst as a key factor of hyperhydricity in garlic plantlet in vitro. Plant Cell Tissue Organ Cult 120(2):571–584
Tian J, Cheng Y, Kong X, Liu M, Jiang F, Wu Z (2017) Induction of reactive oxygen species and the potential role of NADPH oxidase in hyperhydricity of garlic plantlets in vitro. Protoplasma 254:379–388
van den Dries N, Giannì S, Czerednik A, Krens FA, de Klerk GJ (2013) Flooding of the apoplast is a key factor in the development of hyperhydricity. J Exp Bot 64(16):5221–5230
Vinoth A, Ravindhran R (2015) Reduced hyperhydricity in watermelon shoot cultures using silver ions. In Vitro Cell Dev 51(3):258–264
Wi SJ, Su JJ, Park KY (2010) Inhibition of biphasic ethylene production enhances tolerance to abiotic stress by reducing the accumulation of reactive oxygen species in Nicotiana tabacum. Mol Cells 30(1):37–49
Xie SS, Wu HJ, Zang HY, Wu LM, Zhu QQ, Gao XW (2014) Plant growth promotion by spermidine-producing Bacillus subtilis OKB105. Mol Plant Microbe Interact 27(7):655–663
Yin CC, Ma B, Collinge DP, Pogson BJ, He SJ, Xiong Q, Duan KX, Chen H, Yang C, Lu X (2015) Ethylene responses in rice roots and coleoptiles are differentially regulated by a carotenoid isomerase-mediated abscisic acid pathway. Plant Cell 27(4):1061–1081
Zifkin M, Jin A, Ozga JA, Zaharia LI, Schernthaner JP, Gesell A, Abrams SR, Kennedy JA, Constabel CP (2012) Gene expression and metabolite profiling of developing highbush blueberry fruit indicates transcriptional regulation of flavonoid metabolism and activation of abscisic acid metabolism. Plant Physiol 158(1):200–224
Zobayed S, Armstrong J, Armstrong W (2001) Micropropagation of potato: evaluation of closed, diffusive and forced ventilation on growth and tuberization. Ann Bot 87(1):53–59
Acknowledgements
This project is sponsored by the Scientific Research Foundation for the Returned Overseas Chinese Scholars, State Education Ministry.
Author information
Authors and Affiliations
Contributions
HG and XX conceived the experiments. HG performed the experiments. HG, XX and LA analyzed the data and wrote the manuscript. JL and HJ helped with some experiments. All authors have reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Henryk Flachowsky.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gao, H., Li, J., Ji, H. et al. Hyperhydricity-induced ultrastructural and physiological changes in blueberry (vaccinium spp.). Plant Cell Tiss Organ Cult 133, 65–76 (2018). https://doi.org/10.1007/s11240-017-1361-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-017-1361-x