Abstract
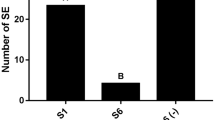
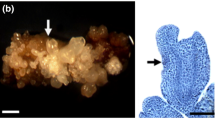
The compound 2,4-Dicholorophenoxyacetic acid (2,4-D) is an important growth regulator which is used in the majority of embryogenic cell and tissue culture systems. However, 2,4-D also appears to have a negative effect on growth and development of plant tissues and organs cultured in vitro. For example, 2,4-D exerts inhibition on in vitro somatic embryo initiation and/or development of most citrus species. To understand the molecular mechanism by which 2,4-D inhibits somatic embryogenesis (SE), proteomic changes of Valencia sweet orange (Citrus sinensis) embryogenic callus induced by treatments with a high concentration of 2,4-D (6 mg l−1) was investigated. Nine 2,4-D-responsive proteins were identified, of which eight were up-regulated and one was down-regulated. Interestingly, three of the eight up-regulated proteins were osmotic stress-associated, suggesting that 2,4-D induced osmotic stress in Valencia embryogenic callus. This speculation was supported by results from our physiological studies: 2,4-D treated callus cells exhibited increased cytoplasm concentration with a significant reduction in relative water content (RWC) and an obvious increase in levels of two osmolytes (proline and soluble sugar). Taken together, our results suggested that 2,4-D could inhibit somatic embryo initiation by, at least in part, inducing osmotic stress to citrus callus cells.






Similar content being viewed by others
Abbreviations
- 2,4-D:
-
2,4-Dichlorophenoxy acetic acid
- 2-DE:
-
Two-dimensional gel electrophoresis
- ACN:
-
Acetonitril
- CBB:
-
Coomassie brilliant blue
- CHAPS:
-
3-[(3-Cholamidopropyl)dimethylammonio]propanesulfonic acid
- DTT:
-
DL-Dithiothreitol
- EST:
-
Expressed sequence tag
- FW:
-
Fresh weight
- GSH:
-
Glutathione
- MALDI-TOF:
-
Matrix-assisted laser desorption ionization time-of-flight
- MS:
-
Mass spectrum
- RWC:
-
Relative water content
- SE:
-
Somatic embryogenesis
- TFA:
-
Trifluoroacetic acid
References
Anil VS, Rao KS (2000) Calcium-mediated signaling during sandalwood somatic embryogenesis. Role for exogenous calcium as second messenger. Plant Physiol 123:1301–1311
Balibrea ME, Rus-Alvarez AM, Bolarin MC, Perez-Alfocea F (1997) Fast changes in soluble carbohydrates and proline contents in tomato seedlings in response to ionic and non-ionic iso-osmotic stresses. J Plant Physiol 151:221–226
Bouché N, Yellin A, Snedden WA, Fromm H (2005) Plant-specific calmodulin-binding proteins. Annu Rev Plant Biol 56:435–466
Chugh A, Khurana P (2002) Gene expression during somatic embryogenesis-recent advances. Curr Sci 86:715–730
Dai SJ, Chen TT, Chong K, Xue YB, Liu SQ, Wang T (2007) Proteomic identification of differentially expressed proteins associated with pollen germination and tube growth reveals characteristics of germinated Oryza sativa pollen. Mol Cell Proteomics 6:207–230
Davletova S, Mészáros T, Miskolczi P, Oberschall A, Török K, Magyar Z, Dudits D, Deák M (2001) Auxin and heat shock activation of a novel member of the calmodulin like domain protein kinase gene family in cultured alfalfa cells. J Exp Bot 52:215–221
Edward JV, Helen LM, Ramachandran K, Ishita C, Bernard AK (1998) DNA repair in higher plants. Mutat Res 400:187–200
Fanous A, Weiland F, Lűck C, Görg A, Friess A, Parlar H (2007) A proteome analysis of Corynebacterium glutamicum after exposure to the herbicide 2,4-Dichlorophenoxy acetic acid (2,4-D). Chemosphere 69:25–31
Fehér A, Pasternak T, Miskolczi P, Ayaydin F, Dudits D (2001) Induction of the embryogenic pathway in somatic plant cells. Acta Hort 560:293–298
Fehér A, Pasternak T, Ötvös K, Miskolczi P, Dudits D (2002) Induction of embryogenic competence in somatic plant cells: a review. Biologia 57:5–12
Fehér A, Pasternak TP, Dudits D (2003) Transition of somatic plant cells to an embryogenic state. Plant Cell Tissue Organ Cult 74:201–228
Foyer CH, Noctor G (2005) Redox homeostasis and antioxidant signaling: a metabolic interface between stress perception and physiological responses. Plant Cell 17:1866–1875
Gillm IS, Zora S, Dhillon BS, Gosal SS (1994) Somatic embryogenesis and plantlet regeneration on calluses derived from seedlings explants of ‘Kinnow’ mandarin (Citrus nobilis Lour.×Citrus deliciosa Tenora). J Hortic Sci 69:231–236
Giovanni C, Maurizio B, Luca M, Laura S, Gian Marco G, Barbara C, Paola O, Luciano Z, Pier GR (2004) Blue silver: a very sensitive colloidal coomassie G-250 staining for proteome analysis. Electrophoresis 25(:1327–1333
Goldsworthy A, Mina MG (1991) Electrical patterns of tobacco cells in media containing indole-3-acetic acid or 2,4-Dichloro-phenoxyacetic acid. Planta 183:368–373
Hadrami I, D’auzac J (1992) Effects of growth regulators on polyamine content and peroxidase activity in Hevea brasiliensis callus. Ann Bot 69:323–325
Hao JU, Deng XX (2002) Occurrence of chromosomal variations and plant regeneration from long-term-cultured citrus callus. In Vitro Cell Dev Biol Plant 38:472–476
Huo HQ, Deng XX (2000) Induction, storage and utilization of embryogenic calli of citrus. Plant Physiol Rep 36:181–187 (in Chinese)
Ikeda-Iwai M, Umehara M, Satoh S, Kamada H (2003) Stress-induced somatic embryogenesis in vegetative tissues of Arabidopsis thaliana. Plant J 34:107–114
Jansen MAK, Booij H, Schel JHN, de Vries SC (1990) Calcium increases the yield of somatic embryos in carrot embryogenic suspension cultures. Plant Cell Rep 9:221–223
Kamada H, Ishikawa K, Saga H, Harada H (1993) Induction of somatic embryogenesis in carrot by osmotic stress. Plant Tissue Cult Lett 10:38–44
Kearus EV, Assmann SM (1993) The guard cell-environment connection. Plant Physiol 102:711–715
Kennedy CD, Stewart RA (1980) The effect of 2,4-Dichlorophenoxyacetic acid on ion uptake by maize roots. J Exp Bot 31:135–140
Lloyd CW, Lowe SB, Peace GW (1980) The mode of action of 2,4-D in counter-acting the elongation of carrot cells grown in culture. J Cell Sci 195:309–312
LoSchiavo F, Filippini F, Cozzani F, Vallone D, Terzi M (1991) Modulation of auxin-binding proteins in cell suspensions I Differential responses of carrot embryo cultures. Plant Physiol 97:60–64
Michalczuk L, Ribnicky DM, Cooke TJ, Cohen JD (1992) Regulation of indole-3-acetic acid biosynthetic pathways in carrot cell cultures. Plant Physiol 100:1346–1353
Mittler R, Vanderauwera S, Gollery M, Breusegem FV (2004) Reactive oxygen gene network of plants. Trends Plant Sci 9:490–498
Moore GA (1985) Factors affecting in vitro embryogenesis from undeveloped ovules of mature citrus fruit. J Am Soc Hort Sci 110:66–70
Moyen C, Bonmort J, Roblin G (2007) Membrane effects of 2,4-Dichlorophenoxyacetic acid in motor cells of Mimosa pudica L. Plant Physiol Biochem 45:420–426
Murashige T, Tucker DPH (1969) Growth factors requirement of citrus tissue cultures. In: Chapman HD (ed) Proceedings of the international citrus symposium, vol 3. Riverside, California, pp 1155–1161
Noctor G, Foyer CH (1998) Ascorbate and glutathione: keeping active oxygen under control. Annu Rev Plant Physiol Plant Mol Biol 49:249–279
Pan ZY, Guan R, Zhu SP, Deng XX (2009) Proteomic analysis of somatic embryogenesis in Valencia sweet orange (Citrus sinensis Osbeck). Plant Cell Rep 28:281–289
Pasternak T, Prinsen E, Ayaydin F, Miskolczi P, Potters G, Asard H, Van Onckelen H, Dudits D, Fehér A (2002) The role of auxin, pH and stress in the activation of embryogenic cell division in leaf protoplast-derived cells of alfalfa (Medicago sativa L.). Plant Physiol 129:1807–1819
Roblin G (1979) Mimosa Pudica: a model for the study of the excitability in plants. Biol Rev 54:135–153
Saladin G, Clément C, Magné C (2003) Stress effects of flumioxazin herbicide on grapevine (Vitis vinifera L.) grown in vitro. Plant Cell Rep 21:1221–1227
Sasaki K, Shimomura K, Kamada H, Harada H (1994) IAA metabolism in embryogenic and non-embryogenic carrot cells. Plant Cell Physiol 35:1159–1164
Schauf CL, Bringle B, Stillwell W (1987) Membrane-directed effects of the plant hormones abscisic acid, indole-3-acetic acid and 2,4-Dichlorophenoxyacetic acid. Biochem Biophys Res Commun 143:1085–1091
Shimabukuro RH, Walsh WC, Wright JP (1989) Effect of diclofop-methyl and 2,4-D on transmembrane proton gradient: a mechanism for their antagonistic interaction. Plant Physiol 77:107–114
Stefania F, Fabio DP, Francesco C, Maurizio S (2002) Effect of 2,4-D and 4-CPPU on somatic embryogenesis from stigma and style transverse thin cell layers of citrus. Plant Cell Tissue Organ Cult 68:57–63
Wang HL, Lee PD, Chen WL, Huang DJ, Su JC (2000) Osmotic stress-induced changes of sucrose metabolism in cultured sweet potato cells. J Exp Bot 51:1991–1999
Xiong L, Zhu JK (2002) Molecular and genetic aspects of plant responses to osmotic stress. Plant Cell Environ 25:131–139
Yang QS, Wang YQ, Zhang JJ, Shi WP, Qian CM, Peng XX (2007) Identification of aluminum-responsive proteins in rice roots by a proteomic approach: cysteine synthase as a key player in Al response. Proteomics 7:737–749
Yoshiba Y, Kiyosue T, Nakashima K, Yamaguchi-Shinozaki K, Shinozaki K (1997) Regulation of levels of proline as an osmolyte in plants under water stress. Plant Cell Physiol 38:1095–1102
Youssefian S, Nakamura M, Orudgev E, Kondo N (2001) Increased cysteine biosynthesis capacity of transgenic tobacco overexpressing an o-acetylserine(thiol) lyase modifies plant responses to oxidative stress. Plant Physiol 126:1001–1011
Zhang JE, Guo WW, Deng XX (2006) Relationship between ploidy variation of citrus calli and competence for somatic embryogenesis. Acta Genet Sin 33:647–654
Acknowledgments
We thank Dr. Xiao Shunyuan (Center for Biosystems Research, University of Maryland Biotechnology Institute, USA) and Dr Xu Qiang for their suggestions on manuscript preparation. The research was financially supported by the National Natural Science Foundation of China (No. 30570973, 30830078, 30921002), and the Ministry of Education of China (IRT0548).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pan, Z., Zhu, S., Guan, R. et al. Identification of 2,4-D-responsive proteins in embryogenic callus of Valencia sweet orange (Citrus sinensis Osbeck) following osmotic stress. Plant Cell Tiss Organ Cult 103, 145–153 (2010). https://doi.org/10.1007/s11240-010-9762-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-010-9762-0