Abstract
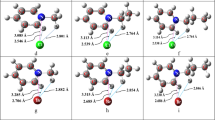
The molecular interactions between sulfoximine (SOI) and hypohalous acids HOX molecules (X=F, Cl and Br) were investigated using the MP2 method at 6-311++G(2d,2p) basis set. Two different patterns including hydrogen bond (HB; H···N, H···O) and halogen bond (XB; N···X, O···X) interactions were found for complex formation between SOI and HOX molecules. In general stability of the hydrogen bond complexes are greater than other one. Quantum theories of atoms in molecules and natural bond orbitals methods have been applied to analyze the intermolecular interactions. Good correlations have been found between the interaction energies (SE), the second-order perturbation energies E(2), and the charge transfer qCT in the studied systems.





Similar content being viewed by others
References
Scheiner S (1997) Hydrogen bonding: a theoretical perspective. Oxford University Press, New York
Scheiner S (1997) Molecular interactions: from van der Waals to strongly bound complexes. Wiley, England
Hadzi D (1997) Theoretical treatment of hydrogen bonding. Wiley, England
Metrangolo P, Resnati G (2008) Halogen bonding: fundamental and applications. Springer, Berlin
Metrangolo P, Neukirch H, Pilati T, Resnati G (2005) Acc Chem Res 38:386–395
Metrangolo P, Resnati G (2008) Science 321:918–919
Metrangolo P, Meyer F, Pilati T, Resnati G, Terraneo G (2008) Angew Chem Int Ed 47:6114–6127
Auffinger P, Hays FA, Westhof E, Ho PS (2004) Proc Natl Sci USA 101:16789–16794
Aakeroy CB, Fasulo M, Schultheiss N, Desper J, Moor C (2007) J Am Chem Soc 129:13772–13773
Lu YX, Zou JW, Wang HQ, Yu QS, Zhang HS, Jiang YJ (2007) J Phys Chem A 111:10781–10788
Li QZ, Lin QQ, Li WZ, Chen JB, Sou JZ (2008) Chem Phys Chem 9:2265–2269
Alkorta I, Blanco F (2008) J Phys Chem A 112:10856–10863
Zabardasti A, Kakanejadifard A, Ghasemian M (2012) Comput. Theor Chem 989:1–6
Huang SL, Swern D (1976) Phosphorus Sulfur Relat Elem 1:309–314
Kennewell PD, Taylor JB (1980) Chem Soc Rev 9:477–498
Reggelin M, Zur C (2000) Synthesis 1:1–64
Trost BM, Matsuoka RT (1992) Synlett 1:27–30
Okamura H, Bolm C (2004) Chem Lett 33:482–487
Okamura H, Bolm C (2004) Org Lett 6(8):1305–1307
Hackenberger CPR, Raabe G, Bolm C (2004) Chem Eur J 10:2942–2952
Frings M, Thomé I, Bolm C (2012) Beilstein J Org Chem 8:1443–1451
Jeanguenat A (2006) Patent WO 2006/032462 A1, March 30, 2006
Huang JX, Zhu Y, Rogers RB (2008) Patent US 2008/0108665 A1, May 8, 2008
Yu H, Qin Z, Dai H, Zhang X (2008) J Agric Food Chem 56:11356–11360
Jeschke P, Thielert W (2010) Patent WO 2010/022897 A2, March 4, 2010
Kim CU, Luh BY, Misco PF (1991) Nucleosides Nucleotides 10:483–484
Dolle RE, McNair D (1993) Tetrahedron Lett 34:133–136
Kawanishi H, Morimoto H, Nakano T (1998) Heterocycles 49:169–189
Jautelat R, Lücking U, Siemeister G (2010) Patent WO 046035 A1, April 29, 2010
Chen XY, Park SJ, Buschmann H (2012) Bioorg Med Chem Lett 22:4307–4309
Thomas E (1979) Infect Immun 23:522–531
Pattison D, Davies M (2001) J Chem Res Toxicol 14:1453–1464
Pattison D, Hawkins C (2003) Chem Res Toxicol 16:439–449
Solimannejad M, Alkorta I (2008) Chem Phys Lett 454:201–206
Berski S, Lundell J, Latajka Z, Leszczynski J (1998) J Phys Chem A 102:10768–10776
Berski B, Silvi Z, Latajka J, Leszczynski J (1999) J Chem Phys 111:2542–2555
Bil A, Latajka Z (2004) Chem Phys 303:43–45
Panek JJ, Berski S (2008) Chem Phys Lett 467:42–45
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Montgomery JA, Vreven T, Kudin KN, Burant JC, Millam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Knox JE, Hratchian HP, Cross JB, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Gonzalez C, Pople JA (2003) Gaussian 03. Gaussian Inc., Pittsburgh
Boys SF, Bernardi F (1970) Mol Phys 19:553–566
Bader RFW (1990) Atoms in molecules: a quantum theory. Oxford University Press, Oxford
Reed AE, Curtiss LA, Weinhold F (1988) Chem Rev 88:899–926
Kumar PS, Bharatam PV (2005) Tetrahedron 61:5633–5639
Bharatam PV, Gupta A, Kaur A (2002) Tetrahedron 58:1759–1764
Senning A (1972) Sulfur in organic and inorganic chemistry. Marcel Dekker, New York
Pyne SG, Dong A (1995) Tetrahedron Lett 36:3029–3030
Pyne SG, Davis DH, Dong Z (1998) Tetrahedron Lett 39:8499–8502
Harmata H, Glaser R, Chen GS (1995) Tetrahedron Lett 36:9145–9148
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kakanejadifard, A., Japelaghi, S., Ghasemian, M. et al. Theoretical study of molecular interactions of sulfoximine with hypohalous acids HOF, HOCl, and HOBr. Struct Chem 26, 23–33 (2015). https://doi.org/10.1007/s11224-014-0461-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-014-0461-z