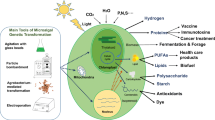
Abstract
Recombinant proteins are widely used for industrial, nutritional, and medical applications. Green microalgae have attracted considerable attention recently as a biomanufacturing platform for the production of recombinant proteins for a number of reasons. These photosynthetic eukaryotic microorganisms are safe, scalable, easy to genetically modify through transformation, mutagenesis, or breeding, and inexpensive to grow. Many microalgae species are genetically transformable, but the green alga Chlamydomonas reinhardtii is the most widely used host for recombinant protein expression. An extensive suite of molecular genetic tools has been developed for C. reinhardtii over the last 25 years, including a fully sequenced genome, well-established methods for transformation, mutagenesis and breeding, and transformation vectors for high levels of recombinant protein accumulation and secretion. Here, we review recent successes in the development of C. reinhardtii as a biomanufacturing host for recombinant proteins, including antibodies and immunotoxins, hormones, industrial enzymes, an orally-active colostral protein for gastrointestinal health, and subunit vaccines. In addition, we review the biomanufacturing potential of other green algae from the genera Dunaliella and Chlorella.

Similar content being viewed by others
References
Alper H, Fischer C, Nevoigt E, Stephanopoulos G (2005) Tuning genetic control through promoter engineering. Proc Natl Acad Sci USA 102:12678–12683. doi:10.1073/pnas.0504604102
Bai L-L, Yin W-B, Chen Y-H et al (2013) A new strategy to produce a defensin: stable production of mutated NP-1 in nitrate reductase-deficient Chlorella ellipsoidea. PLoS One 8:e54966. doi:10.1371/journal.pone.0054966
Beg QK, Kapoor M, Mahajan L, Hoondal GS (2001) Microbial xylanases and their industrial applications: a review. Appl Microbiol Biotechnol 56:326–338
Belhaj K, Chaparro-Garcia A, Kamoun S, Nekrasov V (2013) Plant genome editing made easy: targeted mutagenesis in model and crop plants using the CRISPR/Cas system. Plant Methods 9:39. doi:10.1186/1746-4811-9-39
Beligni MV, Yamaguchi K, Mayfield SP (2004) The translational apparatus of Chlamydomonas reinhardtii chloroplast. Photosynth Res 82:315–325. doi:10.1007/s11120-004-2440-5
Breiman A, Fawcett TW, Ghirardi ML, Mattoo AK (1992) Plant organelles contain distinct peptidylprolyl cis, trans-isomerases. J Biol Chem 267:21293–21296
Brooks SA (2004) Appropriate glycosylation of recombinant proteins for human use: implications of choice of expression system. Mol Biotechnol 28:241–255. doi:10.1385/MB:28:3:241
Celik E, Calık P (2012) Production of recombinant proteins by yeast cells. Biotechnol Adv 30:1108–1118. doi:10.1016/j.biotechadv.2011.09.011
Cerutti H, Johnson AM, Gillham NW, Boynton JE (1997) Epigenetic silencing of a foreign gene in nuclear transformants of Chlamydomonas. Plant Cell 9:925–945. doi:10.1105/tpc.9.6.925
Chen Y, Wang Y, Sun Y et al (2001) Highly efficient expression of rabbit neutrophil peptide-1 gene in Chlorella ellipsoidea cells. Curr Genet 39:365–370
Corchero JL, Gasser B, Resina D et al (2013) Unconventional microbial systems for the cost-efficient production of high-quality protein therapeutics. Biotechnol Adv 31:140–153. doi:10.1016/j.biotechadv.2012.09.001
Danon A, Mayfield SP (1994) Light-regulated translation of chloroplast messenger RNAs through redox potential. Science 266:1717–1719
Dauvillée D, Delhaye S, Gruyer S et al (2010) Engineering the chloroplast targeted malarial vaccine antigens in Chlamydomonas starch granules. PLoS One 5:e15424. doi:10.1371/journal.pone.0015424
De Wilde C, Van Houdt H, De Buck S et al (2000) Plants as bioreactors for protein production: avoiding the problem of transgene silencing. Plant Mol Biol 43:347–359
Demain AL, Vaishnav P (2009) Production of recombinant proteins by microbes and higher organisms. Biotechnol Adv 27:297–306
Demurtas OC, Massa S, Ferrante P et al (2013) A chlamydomonas-derived human papillomavirus 16 E7 vaccine induces specific tumor protection. PLoS One 8:e61473. doi:10.1371/journal.pone.0061473
DePalma A (2009) Removing impediments in downstream processing. Genet Eng Biotechnol News 29:1–2
Dreesen IAJ, Charpin-El Hamri G, Fussenegger M (2010) Heat-stable oral alga-based vaccine protects mice from Staphylococcus aureus infection. J Biotechnol 145:273–280. doi:10.1016/j.jbiotec.2009.12.006
Eichler-Stahlberg A, Weisheit W, Ruecker O, Heitzer M (2009) Strategies to facilitate transgene expression in Chlamydomonas reinhardtii. Planta 229:873–883. doi:10.1007/s00425-008-0879-x
Evers P (2010) The Future of the Biologicals Market: Market overview, innovations and company profiles. Bus. Insights
FDA (2009) SUMMARY REPORT on antimicrobials sold or distributed for use in food-producing animals
Feng S, Feng W, Zhao L et al (2013) Preparation of transgenic Dunaliella salina for immunization against white spot syndrome virus in crayfish. Arch Virol. doi:10.1007/s00705-013-1856-7
Ferrer-Miralles N, Domingo-Espín J, Corchero JL et al (2009) Microbial factories for recombinant pharmaceuticals. Microb Cell Fact 8:17. doi:10.1186/1475-2859-8-17
Gaj T, Gersbach CA, Barbas CF (2013) ZFN, TALEN, and CRISPR/Cas-based methods for genome engineering. Trends Biotechnol 31:397–405. doi:10.1016/j.tibtech.2013.04.004
Geng D, Wang Y, Wang P et al (2003) Stable expression of hepatitis B surface antigen gene in Dunaliella salina (Chlorophyta). J Appl Phycol 15:451–456. doi:10.1023/B:JAPH.0000004298.89183.e5
Georgianna DR, Hannon MJ, Marcuschi M et al (2013) Production of recombinant enzymes in the marine alga Dunaliella tertiolecta. Algal Res 2:2–9
Gong Y, Hu H, Gao Y et al (2011) Microalgae as platforms for production of recombinant proteins and valuable compounds: progress and prospects. J Ind Microbiol Biotechnol 38:1879–1890. doi:10.1007/s10295-011-1032-6
Gregory JA, Li F, Tomosada LM et al (2012) Algae-produced Pfs25 elicits antibodies that inhibit malaria transmission. PLoS One 7:e37179. doi:10.1371/journal.pone.0037179
Gregory JA, Topol AB, Doerner DZ, Mayfield S (2013) Alga-produced cholera toxin-Pfs25 fusion proteins as oral vaccines. Appl Environ Microbiol 79:3917–3925. doi:10.1128/AEM.00714-13
Harris EH, Stern DB, Witman GB (2009) The chlamydomonas sourcebook, 2nd edn. Null. doi:10.1016/B978-0-12-370873-1.00002-2
Hawkins RL, Nakamura M (1999) Expression of human growth hormone by the eukaryotic alga, Chlorella. Curr Microbiol 38:335–341
He D-M, Qian K-X, Shen G-F et al (2007) Recombination and expression of classical swine fever virus (CSFV) structural protein E2 gene in Chlamydomonas reinhardtii chroloplasts. Colloids Surf B Biointerfaces 55:26–30. doi:10.1016/j.colsurfb.2006.10.042
Hou Q, Qiu S, Liu Q et al (2013) Selenoprotein-transgenic Chlamydomonas reinhardtii. Nutrients 5:624–636. doi:10.3390/nu5030624
Jinek M, Chylinski K, Fonfara I et al (2012) A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science 337:816–821. doi:10.1126/science.1225829
Jones CS, Mayfield SP (2013) Steps toward a globally available malaria vaccine: harnessing the potential of algae for future low cost vaccines. Bioengineered 4:164–167. doi:10.4161/bioe.22577
Jones CS, Luong T, Hannon M et al (2013) Heterologous expression of the C-terminal antigenic domain of the malaria vaccine candidate Pfs48/45 in the green algae Chlamydomonas reinhardtii. Appl Microbiol Biotechnol 97:1987–1995. doi:10.1007/s00253-012-4071-7
Keam SJ, Harper DM (2008) Human papillomavirus types 16 and 18 vaccine (recombinant, AS04 adjuvanted, adsorbed) [Cervarix]. Drugs 68:359–372
Kim D-H, Kim YT, Cho JJ et al (2002) Stable integration and functional expression of flounder growth hormone gene in transformed microalga, Chlorella ellipsoidea. Mar Biotechnol (NY) 4:63–73. doi:10.1007/s1012601-0070-x
Kindle KL, Schnell RA, Fernández E, Lefebvre PA (1989) Stable nuclear transformation of Chlamydomonas using the Chlamydomonas gene for nitrate reductase. J Cell Biol 109:2589–2601
Larson MA, Wei SH, Weber A et al (2003) Human serum amyloid A3 peptide enhances intestinal MUC3 expression and inhibits EPEC adherence. Biochem Biophys Res Commun 300:531–540. doi:10.1016/S0006-291X(02)02901-7
Mack DR, McDonald TL, Larson MA et al (2003) The conserved TFLK motif of mammary-associated serum amyloid A3 is responsible for up-regulation of intestinal MUC3 mucin expression in vitro. Pediatr Res 53:137–142. doi:10.1203/00006450-200301000-00023
Mansfield E, Amlot P, Pastan I, FitzGerald DJ (1997) Recombinant RFB4 immunotoxins exhibit potent cytotoxic activity for CD22-bearing cells and tumors. Blood 90:2020–2026
Manuell AL, Beligni MV, Elder JH et al (2007a) Robust expression of a bioactive mammalian protein in Chlamydomonas chloroplast. Plant Biotechnol J 5:402–412. doi:10.1111/j.1467-7652.2007.00249.x
Manuell AL, Quispe J, Mayfield SP (2007b) Structure of the chloroplast ribosome: novel domains for translation regulation. PLoS Biol 5:e209. doi:10.1371/journal.pbio.0050209
Maul JE (2002) The Chlamydomonas reinhardtii plastid chromosome: Islands of genes in a sea of repeats. Plant Cell Online 14:2659–2679. doi:10.1105/tpc.006155
Mayfield SP, Kindle KL (1990) Stable nuclear transformation of Chlamydomonas reinhardtii by using a Chlamydomonas reinhardtii gene as the selectable marker. Proc Natl Acad Sci USA 87:2087–2091
Mayfield SP, Franklin SE, Lerner RA (2003) Expression and assembly of a fully active antibody in algae. Proc Natl Acad Sci USA 100:438–442. doi:10.1073/pnas.0237108100
Merchant SS, Prochnik SE, Vallon O et al (2007) The Chlamydomonas genome reveals the evolution of key animal and plant functions. Science 318:245–250. doi:10.1126/science.1143609
Molnár A, Schwach F, Studholme DJ et al (2007) miRNAs control gene expression in the single-cell alga Chlamydomonas reinhardtii. Nature 447:1126–1129. doi:10.1038/nature05903
Polizeli MLTM, Rizzatti ACS, Monti R et al (2005) Xylanases from fungi: properties and industrial applications. Appl Microbiol Biotechnol 67:577–591. doi:10.1007/s00253-005-1904-7
Popescu CE, Lee RW (2007) Mitochondrial genome sequence evolution in Chlamydomonas. Genetics 175:819–826. doi:10.1534/genetics.106.063156
Qin X, Qian J, Yao G et al (2011) GAP promoter library for fine-tuning of gene expression in Pichia pastoris. Appl Environ Microbiol 77:3600–3608. doi:10.1128/AEM.02843-10
Rasala BA, Mayfield SP (2011) The microalga Chlamydomonas reinhardtii as a platform for the production of human protein therapeutics. Bioeng Bugs 2:50–54. doi:10.4161/bbug.2.1.13423
Rasala BA, Muto M, Lee PA et al (2010) Production of therapeutic proteins in algae, analysis of expression of seven human proteins in the chloroplast of Chlamydomonas reinhardtii. Plant Biotechnol J 8:719–733. doi:10.1111/j.1467-7652.2010.00503.x
Rasala BA, Lee PA, Shen Z et al (2012) Robust expression and secretion of Xylanase1 in Chlamydomonas reinhardtii by fusion to a selection gene and processing with the FMDV 2A peptide. PLoS One 7:e43349. doi:10.1371/journal.pone.0043349
Rasala BA, Barrera DJ, Ng J et al (2013) Expanding the spectral palette of fluorescent proteins for the green microalga Chlamydomonas reinhardtii. Plant J 74:545–556. doi:10.1111/tpj.12165
Rayman MP, Infante HG, Sargent M (2008) Food-chain selenium and human health: spotlight on speciation. Br J Nutr 100:238–253. doi:10.1017/S0007114508922522
Sanchez S, Demain AL (2011) Enzymes and bioconversions of industrial, pharmaceutical, and biotechnological significance. Org Process Res Dev 15:224–230. doi:10.1021/op100302x
Schroda M (2004) The Chlamydomonas genome reveals its secrets: chaperone genes and the potential roles of their gene products in the chloroplast. Photosynth Res 82:221–240. doi:10.1007/s11120-004-2216-y
Schroda M (2006) RNA silencing in Chlamydomonas: mechanisms and tools. Curr Genet 49:69–84. doi:10.1007/s00294-005-0042-1
Shen G-L, Li J-L, Ghetie M-A et al (1988) Evaluation of four CD22 antibodies as ricin a chain-containing immunotoxins for thein vivo therapy of human B-cell leukemias and lymphomas. Int J Cancer 42:792–797. doi:10.1002/ijc.2910420527
Sizova I, Greiner A, Awasthi M et al (2013) Nuclear gene targeting in Chlamydomonas using engineered zinc-finger nucleases. Plant J 73:873–882. doi:10.1111/tpj.12066
Specht EA, Mayfield SP (2014) Algae-based oral recombinant vaccines. Front Microbiol (in press)
Specht E, Miyake-Stoner S, Mayfield S (2010) Micro-algae come of age as a platform for recombinant protein production. Biotechnol Lett 32:1373–1383. doi:10.1007/s10529-010-0326-5
Su Z-L, Qian K-X, Tan C-P et al (2005) Recombination and heterologous expression of allophycocyanin gene in the chloroplast of Chlamydomonas reinhardtii. Acta Biochim Biophys Sin (Shanghai) 37:709–712
Sun M, Qian K, Su N et al (2003) Foot-and-mouth disease virus VP1 protein fused with cholera toxin B subunit expressed in Chlamydomonas reinhardtii chloroplast. Biotechnol Lett 25:1087–1092
Surzycki R, Greenham K, Kitayama K et al (2009) Factors effecting expression of vaccines in microalgae. Biologicals 37:133–138. doi:10.1016/j.biologicals.2009.02.005
Swiech K, Picanço-Castro V, Covas DT (2012) Human cells: new platform for recombinant therapeutic protein production. Protein Expr Purif 84:147–153. doi:10.1016/j.pep.2012.04.023
Tang Z, Zhao F, Qin S et al (2004) The production of rAPC and the study of its inhibition effect on H22 tumor. High Technol Lett 3:83–86
Tisch R, Yang XD, Singer SM et al (1993) Immune response to glutamic acid decarboxylase correlates with insulitis in non-obese diabetic mice. Nature 366:72–75. doi:10.1038/366072a0
Tran M, Zhou B, Pettersson PL et al (2009) Synthesis and assembly of a full-length human monoclonal antibody in algal chloroplasts. Biotechnol Bioeng 104:663–673. doi:10.1002/bit.22446
Tran M, Henry RE, Siefker D et al (2013a) Production of anti-cancer immunotoxins in algae: ribosome inactivating proteins as fusion partners. Biotechnol Bioeng. doi:10.1002/bit.24966
Tran M, Van C, Barrera DJ et al (2013b) Production of unique immunotoxin cancer therapeutics in algal chloroplasts. Proc Natl Acad Sci USA 110:E15–E22. doi:10.1073/pnas.1214638110
Van Hulten MC, Witteveldt J, Snippe M, Vlak JM (2001) White spot syndrome virus envelope protein VP28 is involved in the systemic infection of shrimp. Virology 285:228–233. doi:10.1006/viro.2001.0928
Walsh G (2010) Biopharmaceutical benchmarks 2010. Nat Biotechnol 28:917–924. doi:10.1038/nbt0910-917
Wang X, Brandsma M, Tremblay R et al (2008) A novel expression platform for the production of diabetes-associated autoantigen human glutamic acid decarboxylase (hGAD65). BMC Biotechnol 8:87. doi:10.1186/1472-6750-8-87
Wiley SR, Schooley K, Smolak PJ et al (1995) Identification and characterization of a new member of the TNF family that induces apoptosis. Immunity 3:673–682
Wu-Scharf D (2000) Transgene and transposon silencing in Chlamydomonas reinhardtii by a DEAH-Box RNA helicase. Science (80–) 290:1159–1162. doi:10.1126/science.290.5494.1159
Yang Z, yinü Li, Chen F et al (2006) Expression of human soluble TRAIL in Chlamydomonas reinhardtii chloroplast. Chinese Sci Bull 51:1703–1709. doi:10.1007/s11434-006-2041-0
Yoon S-M, Kim SY, Li KF et al (2011) Transgenic microalgae expressing Escherichia coli AppA phytase as feed additive to reduce phytate excretion in the manure of young broiler chicks. Appl Microbiol Biotechnol 91:553–563. doi:10.1007/s00253-011-3279-2
Zhang X-Y, Wang P, Zhao S-M et al (2006a) Expression of rabbit neutrophile peptide-1 in nitrate reductase-deficient mutant of Chlorella ellipsoidea. Yi Chuan 28:1580–1584
Zhang Y-K, Shen G-F, Ru B-G (2006b) Survival of human metallothionein-2 transplastomic Chlamydomonas reinhardtii to ultraviolet B exposure. Acta Biochim Biophys Sin (Shanghai) 38:187–193
Zhao T, Li G, Mi S et al (2007) A complex system of small RNAs in the unicellular green alga Chlamydomonas reinhardtii. Genes Dev 21:1190–1203. doi:10.1101/gad.1543507
Zhu J (2012) Mammalian cell protein expression for biopharmaceutical production. Biotechnol Adv 30:1158–1170
Zorin B, Hegemann P, Sizova I (2005) Nuclear-gene targeting by using single-stranded DNA avoids illegitimate DNA integration in Chlamydomonas reinhardtii. Eukaryot Cell 4:1264–1272. doi:10.1128/EC.4.7.1264-1272.2005
Acknowledgments
This work was supported the by the Department of Energy (DE-EE0003373), the California Energy Commission (CILMSF #500-10-039) and Triton Algae Innovations. We thank Elizabeth Specht and Miller Tran for reviewing and editing the manuscript. Stephen Mayfield and Beth Rasala are founders of Triton Algae Innovations, a company that may potentially benefit from the commercialization of green algae for the production of recombinant proteins.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rasala, B.A., Mayfield, S.P. Photosynthetic biomanufacturing in green algae; production of recombinant proteins for industrial, nutritional, and medical uses. Photosynth Res 123, 227–239 (2015). https://doi.org/10.1007/s11120-014-9994-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-014-9994-7