Abstract
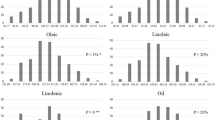
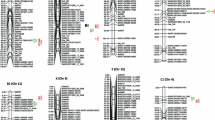
Soybean [Glycine max (L.) Merr.] is an important oilseed crop which produces about 30 % of the world’s edible vegetable oil. The quality of soybean oil is determined by its fatty acid composition. Soybean oil high in oleic and low in linolenic fatty acids is desirable for human consumption and other uses. The objectives of this study were to identify quantitative trait loci (QTLs) for unsaturated fatty acids and to evaluate the genetic effects of single QTL and QTL combinations in soybean. A population of recombinant inbred lines derived from the cross of SD02-4-59 × A02-381100 was evaluated for fatty acid content in seven environments. In total, 516 polymorphic single nucleotide polymorphism markers, 477 polymorphic simple sequence repeat markers and three GmFAD3 genes were used to genotype the mapping population. By using the composite interval mapping and/or the interval mapping method, a total of 15 QTLs for the three unsaturated fatty acids were detected in more than two environments. Two QTLs for oleic acid on linkage groups G [chromosome (Chr) 18] (qOLE-G) and J (Chr 16) (qOLE-J), three QTLs for linoleic acid on linkage groups A1 (Chr 5) (qLLE-A1) and G (Chr 18) (qLLE-G-1 and qLLE-G-2), and five QTLs for linolenic acid on linkage groups C2 (Chr 6), D1a (Chr 1), D1b (Chr 2), F (Chr 13) and G (Chr 18) were consistently detected in at least three individual environments and the average data over all environments. Significant QTL × QTL interactions were not detected. However, significant QTL × environment interactions were detected for all the QTLs which were repeatedly detected. Some QTLs reported previously were confirmed, and seven new QTLs (two for oleic acid, two for linoleic acid and three for linolenic acid) were identified in this study. Comparisons of two-locus and three-locus combinations indicated that cumulative effects of QTLs were significant for all the three unsaturated fatty acids. QTL pyramiding by molecular marker-assisted breeding would be an appropriate strategy for the improvement of unsaturated fatty acids in soybean.

Similar content being viewed by others
References
Bachlava E, Dewey RE, Burton JW, Cardinal AJ (2009) Mapping and comparison of quantitative trait loci for oleic acid seed content in two segregating soybean populations. Crop Sci 49:433–442
Beavis WD (1994) The power and deceit of QTL experiments: lessons from comparative QTL studies. In: 49th annual corn and sorghum industry research conference. American Seed Trade Association, Washington, DC
Bilyeu KD, Palavalli L, Sleper DA, Beuselinck PR (2003) Three microsomal omega-3 fatty-acid desaturase genes contribute to soybean linolenic acid levels. Crop Sci 43:1833–1838
Bilyeu K, Palavalli L, Sleper D, Beuselinck P (2005) Mutations in soybean microsomal omega-3 fatty acid desaturase genes reduce linolenic acid concentration in soybean seeds. Crop Sci 45:1830–1836
Bilyeu K, Palavalli L, Sleper D, Beuselinck P (2006) Molecular genetic resources for development of 1% linolenic acid soybeans. Crop Sci 46:1913–1918
Burton JW, Wilson RF, Brim CA (1983) Recurrent selection in soybeans: IV. Selection for increased oleic acid percentage in seed oil. Crop Sci 23:744–747
Concibido VC, Denny R, Lange DA, Orf JH, Young ND (1997) Genome mapping on soybean cyst nematode resistance genes in ‘Peking’, PI 90763 and PI 88788 using DNA markers. Crop Sci 37:258–264
Cornelious B, Chen P, Chen Y, de Leon N, Shannon JG, Wang D (2005) Identification of QTLs underlying water-logging tolerance in soybean. Mol Breed 16:103–112
Crapiste GH, Brevedan MI, Carelli AA (1999) Oxidation of sunflower oil during storage. J Am Oil Chem Soc 76:1437–1443
Diers BW, Shoemaker RC (1992) Restriction fragment length polymorphism analysis of soybean fatty acid content. J Am Oil Chem Soc 69:1242–1244
Fasoula VA, Harris DK, Boerma HR (2004) Validation and designation of quantitative trait loci for seed protein, seed oil, and seed weight from two soybean populations. Crop Sci 44:1218–1225
Fehr WR (1987) Principles of cultivar development—volume 1, theory and technique. Macmillan Publishing Company, New York
Ha B, Monteros MJ, Boerma HR (2010) Development of SNP assays associated with oleic acid QTL in N00-3350 soybean. Euphytica 176:403–415
Henderson MM (1991) Correlations between fatty acid intake and cancer incidence. In: Nelson GJ (ed) Health effects of dietary fatty acids. AOCS, Champaign, IL, p 136
Hu FB, Stampfer MJ, Manson JE, Rimm E, Colditz GA, Rosner BA, Hennekens CH, Willett WC (1997) Dietary fat intake and the risk of coronary heart disease in women. N Engl J Med 337:1491–1499
Huang N, Angeles ER, Domingo J, Magpantay G, Singh S, Zang G, Kumaravadivel N, Bennet J, Khush GS (1997) Pyramiding of bacterial blight resistance genes in rice: marker-assisted selection using RFLP and PCR. Theor Appl Genet 95:313–320
Hyten DL, Pantalone VR, Sams CE, Saxton AM, Landau-Ellis D, Stefaniak TR, Schmidt ME (2004a) Seed quality QTL in a prominent soybean population. Theor Appl Genet 109:552–561
Hyten DL, Pantalone VR, Saxton AM, Schmidt ME, Sams CE (2004b) Molecular mapping and identification of soybean fatty acid modifier quantitative trait loci. J Am Oil Chem Soc 81:1115–1118
Jiang G-L, Shi JR, Ward RW (2007) QTL analysis of resistance to Fusarium head blight in the novel wheat germplasm CJ 9306. I. Resistance to fungal spread. Theor Appl Genet 116:3–13
Lark KG, Chase K, Adler F, Mansur LM, Orf JH (1995) Interactions between quantitative trait loci in soybean in which trait variation at one locus is conditional upon a specific allele at another. Proc Natl Acad Sci USA 92:4656–4660
Li Z, Wilson RF, Rayford WE, Boerma HR (2002) Molecular mapping genes conditioning reduced palmitic acid content. Crop Sci 42:373–378
Li X, Han Y, Teng W, Zhang S, Yu K, Poysa V, Anderson T, Ding J, Li W (2010) Pyramided QTL underlying tolerance to Phytophthora root rot in mega-environment from soybean cultivars ‘conrad’ and ‘Hefeng 25’. Theor Appl Genet 121:651–658
Melchinger AE, Utz HF, Schon CC (2000) From Mendel to Fisher: the power and limits of QTL mapping for quantitative traits. Vortr Pflanzenzucht 48:132–142
Mensink RP, Temme EH, Hornstra G (1994) Dietary saturated and trans fatty acids and lipoprotein metabolism. Ann Med 26:461–464
Monteros MJ, Burton JW, Boerma HR (2008) Molecular mapping and confirmation of QTL associated with oleic content in N00-3350 soybean. Crop Sci 48:2223–2234
Mounts TL, Warner K, List GR, Kleiman R, Fehr WR, Hammond EG, Wilcox JR (1988) Effect of altered fatty acid composition on soybean oil stability. J Am Oil Chem Soc 65:624–628
Njiti VN, Johnson JE, Torto TA, Gray LE, Lightfoot DA (2001) Inoculum rate influences selection for field resistance to soybean sudden death syndrome in the greenhouse. Crop Sci 41:1726–1731
Njiti VN, Meksem K, Iqbal MJ, Johnson JE, Kassem MA, Zobrist KF, Kilo VY, Lightfoot DA (2002) Common loci underlie field resistance to soybean sudden death syndrome in Forrest, Pyramid, Essex, and Douglas. Theor Appl Genet 104:294–300
Oliva ML, Shannon JG, Sleper DA, Ellersieck MR, Cardinal AJ, Paris RL, Lee JD (2006) Stability of fatty acid profile in soybean genotypes with modified seed oil composition. Crop Sci 46:2069–2075
Orf JH, Chase K, Adler FR, Mansur LM, Lark KG (1999) Genetics of soybean agronomic traits: II. Interaction between yield quantitative trait loci in soybean. Crop Sci 39:1652–1657
Panthee DR, Pantalone VR, Saxton AM (2006) Modifier QTL for fatty acid composition in soybean oil. Euphytica 152:67–73
Pham A-T, Lee JD, Shannon JG, Bilyeu KD (2010) Mutant alleles of FAD2-1A and FAD2-1B combine to produce soybeans with the high oleic acids seed oil trait. BMC Plant Biol 10:195
Pham A-T, Shannon JG, Bilyeu KD (2012) Combinations of mutant FAD2 and FAD3 genes to produce high oleic acid and low linolenic acid soybean oil. Theor Appl Genet 125:503–515
Primomo VS, Poysa VW, Ablett GR, Jackson C, Gijzen M, Rajcan I (2005) Mapping QTL for individual and total isoflavone content in soybean seeds. Crop Sci 45:2454–2464
Reinprecht Y, Poysa VW, Yu K, Rajcan I, Ablett GR, Pauls KP (2006) Seed and agronomic QTL in low linolenic acid, lipoxygenase-free soybean (Glycine max (L.) Merrill) germplasm. Genome 49:1510–1527
Richardson KL, Vales MI, Kling JG, Mundt CC, Hayes PM (2006) Pyramiding and dissecting disease resistance QTL to barley stripe rust. Theor Appl Genet 113:485–495
Song Q, Marek LF, Shoemaker RC, Lark KG, Concibido VC, Delannay X, Specht JE, Cregan PB (2004) A new integrated genetic linkage map of the soybean. Theor Appl Genet 109:122–128
Song Q, Jia G, Zhu Y, Grant D, Nelson RT, Hwang E-Y, Hyten DL, Cregan PB (2010) Abundance of SSR motifs and development of candidate polymorphic markers (BARCSOYSSR_1.0) in soybean. Crop Sci 50:1950–1960
Tanksley SD (1993) Mapping polygenes. Annu Rev Genet 27:205–233
Tucker DM, Saghai-Maroof MA, Mideros S, Skoneczka JA, Nabati DA, Buss GR, Hoeschele I, Tyler BM, St. Martin SK, Dorrance AE (2010) Mapping quantitative trait loci for partial resistance to Phytophthora sojae in a soybean interspecific cross. Crop Sci 50:628–635
Van Ooijen JW, Voorrips RE (2001) JoinMap 3.0: software for the calculation of genetic linkage maps. Plant Research International, Wageningen, The Netherlands
Wang S, Basten CJ, Zeng ZB (2005) Windows QTL cartographer 2.5. Department of Statistics, North Carolina State University, Raleigh, NC
Wang J, Li H, Zhang L, Li C, Meng L (2011) QTL IciMapping V3.1. Institute of Crop Science and CIMMT China, Chinese Academy of Agriculture Sciences, Beijing, China
Wang X, Jiang G-L, Green M, Scott RA, Hyten DL, Cregan PB (2012) Quantitative trait locus analysis of saturated fatty acids in a population of recombinant inbred lines of soybean. Mol Breed 30:1163–1179
White HB, Quackenbush FW, Probsr AH (1961) Occurrence and inheritance of linolenic and linoleic acids in soybean seeds. J Am Oil Chem Soc 38:113–117
Wilcox JR (2004) World distribution and trade of soybean. In: Boerma RH, Specht JE (eds) Soybeans: improvement, production, and uses, 3rd edn. American Society of Agronomy, Madison, WI, pp 1–13
Willett WC (1994) Diet and health: what should we eat? Science 264:532–537
Wilson RF (2004) Seed composition. In: Boerma RH, Specht JE (eds) Soybeans: improvement, production, and uses, 3rd edn. American Society of Agronomy, Madison, WI, pp 621–669
Winter SMJ, Shelp BJ, Anderson TR, Welacky TW, Rajcan I (2007) QTL associated with horizontal resistance to soybean cyst nematode in Glycine soja PI464925B. Theor Appl Genet 114:461–472
Wolf RB, Cavins JF, Kleiman R, Black LT (1982) Effect of temperature on soybean seed constituents: oil, protein, moisture, fatty acids, amino acids and sugars. J Am Oil Chem Soc 59:230–232
Yesudas CR, Bashir R, Geisler MB, Lightfoot DA (2013) Identification of germplasm with stacked QTL underlying seed traits in an inbred soybean population from cultivars Essex and Forrest. Mol Breed 31:693–703
Yue P, Arelli PR, Sleper DA (2001) Molecular characterization of resistance to Heterodera glycines in soybean PI 438489B. Theor Appl Genet 102:921–928
Acknowledgments
This research was supported in part by USB Seed Composition Research project via USDA/ARS, and South Dakota Soybean Research and Promotion Council project. The determination of fatty acids was completed in the USDA/ARS Lab., in Peoria, IL and the Iowa State University DNA Facility in Ames, IA. Genotyping of three GmFAD3 genes was performed by Dr. Kristin Bilyeu’s Lab., USDA/ARS in Columbia, MO. The parental line A02-381100 was provided by Dr. Walter Fehr, Iowa State University. Dr. Kelly Whiting, the United Soybean Board, reviewed the manuscript and provided constructive suggestions.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, X., Jiang, GL., Green, M. et al. Quantitative trait locus analysis of unsaturated fatty acids in a recombinant inbred population of soybean. Mol Breeding 33, 281–296 (2014). https://doi.org/10.1007/s11032-013-9948-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11032-013-9948-3