Abstract
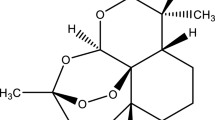
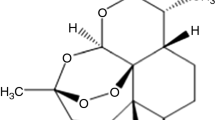
This paper presents the results obtained after the investigation of two artemisinin derived sesquiterpenes, namely artesunate and artemether, currently studied for their antitumor properties. The chosen methods of analysis included UATR–FTIR spectroscopy, characterization of the thermal behavior (TG/DTG/HF) in oxidative dynamic atmosphere, and a complete kinetic analysis. The latter was realized using two integral methods (Kissinger–Akahira–Sunose and Flynn–Wall–Ozawa), a differential one (Friedman), and was later completed with the modified NPK method. The study showed that both compounds show similar thermal stability in terms of apparent activation energies and the degradation processes occur in two parallel steps for each compound, this being solely due to chemical transformations.





Similar content being viewed by others
Abbreviations
- T :
-
Time
- T :
-
Temperature
- Α :
-
Conversion degree
- f(α):
-
The differential conversion function
- R :
-
The universal gas constant
- g(α):
-
The integral conversion function
- β :
-
The heating rate
- k(T):
-
The temperature dependence function
- A :
-
The pre-exponential factor
- E a :
-
The apparent activation energy given by the Arrhenius equation
References
Brunton LL, Lazo JS, Parker K, editors. Goodman and Gilman’s the pharmacological basis of therapeutics. 11th ed. New York: McGraw-Hill; 2006.
Aftab T. Artemisia annua—pharmacology and biotechnology (2016). https://doi.org/10.1007/978-3-642-41027-7.
Ho WE, Peh HY, Chan TK, Wong WSF. Artemisinins: pharmacological actions beyond anti-malarial. Pharmacol Ther. 2014;142(1):126–39.
World Heath Organisation. The international pharmacopoeia. 6th ed. Geneva: World Heath Organisation; 2006.
Luo X-D, Yeh HJC, Brossi A, Flippen-Anderson JL, Gillardi R. The chemistry of drugs part IV, configurations of antimalarials derived from qinghaosu: dihydroqingyhaosu, artemether, and artesunic acid. Helv Chim Acta. 1984;67(6):1515–22.
Butcher RJ, Jasinski JP, Yathirajan HS, Bindya S, Narayana B. α-Artemether. Acta Crystallogr Sect E: Struct Rep Online. 2007;63(7):o3291–2.
Wang Z, Chen J, Li L, Zhou Z, Geng Y, Sun T. Detailed structural study of β-artemether: density functional theory (DFT) calculations of infrared, Raman spectroscopy, and vibrational circular dichroism. J Mol Struct. 2015;1097(71963):61–8.
https://pubchem.ncbi.nlm.nih.gov/compound/16394563#section=Top. Accessed 23 Oct 2017.
Lisgarten J, Potter B, Palmer R, Chimanuka B, Aymami J. Structure, absolute configuration, and conformation of the antimalarial drug artesunate. J Chem Crystallogr. 2002;32(1–2):43–8.
Crespo-Ortiz MP, Wei MQ. Antitumor activity of artemisinin and its derivatives: from a well-known antimalarial agent to a potential anticancer drug. J Biomed Biotechnol. 2012;2012:1–18.
Efferth T, Dunstan H, Sauerbrey A, Miyachi H, Chitambar CR. The anti-malarial artesunate is also active against cancer. Int J Oncol. 2001;18(4):767–73.
Efferth T, Sauerbrey A, Olbrich A, Gebhart E, Rauch P, Weber HO, Hengstler JG, Halatsch M-E, Volm M, Tew KD, Ross DD, Funk JO. Molecular modes of action of artesunate in tumor cell lines. Mol Pharmacol. 2003;64(2):382–94.
Gabriëls M, Plaizier-Vercammen JA. Densitometric thin-layer chromatographic determination of artemisinin and its lipophilic derivatives, artemether and arteether. J Chromatogr Sci. 2003;41(7):359–66.
Farsam V, Hassan ZM, Zavaran-Hosseini A, Noori S, Mahdavi M, Ranjbar M. Antitumor and immunomodulatory properties of artemether and its ability to reduce CD4 + CD25 + FoxP3 + T reg cells in vivo. Int Immunopharmacol. 2011;11(11):1802–8.
Raica M, Cimpean AM, Popovici RA, Balica AR, Vladau M, Gaje PN. Mast cells stimulate lymphangiogenesis in the gingiva of patients with periodontal disease. Vivo. 2015;29(1):29–34.
Podariu AC, Popovici AR, Rosianu RS, Oancea R. Comparative study on nickel and chromium salivary concentration in patients with prosthetic restorations on metallic frame. Rev Chim. 2013;64(9):971–3.
Dai Y-FF, Zhou W-WW, Meng J, Du X-LL, Sui Y-PP, Dai L, Wang P-QQ, Huo H-RR, Sui F. The pharmacological activities and mechanisms of artemisinin and its derivatives: a systematic review. Med Chem Res. 2017;26(5):867–80.
Popovici AR, Vlase G, Vlase T, Suta LM, Popoiu C, Ledeti I, Iovanescu G, Fulias A. Local anesthetic agents: III. Study of solid dosage forms with pharmaceutical excipients. Rev Chim. 2015;66(7):1046–51.
Motoc O, Popovici R, Onisei D, Podariu AC. Toxicological activity of some compounds with application on dentistry field experimental study. Rev Chim. 2015;66(7):1024–6.
Oliva A, Llabrés M, Fariña JB. Data analysis of kinetic modelling used in drug stability studies: isothermal versus nonisothermal assays. Pharm Res. 2006;23(11):2595–602.
Chavan RB, Shastri NR. Polymorphic transformation as a result of atovaquone incompatibility with selected excipients. J Therm Anal Calorim. 2017;131(3):1–11.
De Lima Gomes EC, Ercole de Carvalho I, Fialho SL, Barbosa J, Yoshida MI, da Silva Cunha Júnior A. Mixing method influence on compatibility and polymorphism studies by DSC and statistical analysis. J Therm Anal Calorim. 2018;131(3):2123–8.
Veiga A, Oliveira PR, Bernardi LS, Mendes C, Silva MAS, Sangoi MS, Janissek PR, Murakami FS. Solid-state compatibility studies of a drug without melting point. J Therm Anal Calorim. 2018;131(3):3201–9.
Wang X, You J. Study on the thermal decomposition of sofosbuvir. J Anal Appl Pyrolysis. 2017;123(2):376–84.
Sun Y, Ren H, Jiao Q. Comparison of thermal behaviors and decomposition kinetics of NEPE propellant before and after storage. J Therm Anal Calorim. 2018;131(1):101–11.
De Souza SMM, Leles MIG, Da Conceição EC. Evaluation of thermal stability of enalapril maleate tablets using thermogravimetry and differential scanning calorimetry. J Therm Anal Calorim. 2016;123(3):1943–9.
Ledeti I, Bolintineanu S, Vlase G, Circioban D, Dehelean C, Suta LM, Caunii A, Ledeti A, Vlase T, Murariu M. Evaluation of solid-state thermal stability of donepezil in binary mixtures with excipients using instrumental techniques. J Therm Anal Calorim. 2017;130(1):425–31.
Ledeti A, Vlase G, Vlase T, Circioban D, Dehelean C, Ledeti I. Kinetic study for solid-state degradation of mental disorder therapeutic agents. J Therm Anal Calorim. 2016;131:155–65.
Ledeti A, Vlase G, Ledeti I, Vlase T, Matusz P, Dehelean C, Circioban D, Stelea L, Suta LM. Thermal stability of desipramine and imipramine. Rev Chim. 2016;67(2):336–8.
Shah PP, Mashru RC. Palatable reconstitutable dry suspension of artemether for flexible pediatric dosing using cyclodextrin inclusion complexation. Pharm Dev Technol. 2010;15(3):276–85.
Ansari MT, Karim S, Ranjha NM, Shah NH, Muhammad S. Physicochemical characterization of artemether solid dispersions with hydrophilic carriers by freeze dried and melt methods. Arch Pharm Res. 2010;33(6):901–10.
Mahgoub RA, Awad MH, Elkhidr HE. Establishment of simple colorimetric method of analysis artesunate in tablets. IOSR J Pharm Biol Sci Ver I. 2015;10(5):2319–7676.
Ansari MT, Hussain A, Nadeem S, Majeed H, Saeed-Ul-Hassan S, Tariq I, Mahmood Q, Khan AK, Murtaza G. Preparation and characterization of solid dispersions of artemether by freeze-dried method. Biomed Res Int. 2015. https://doi.org/10.1155/2015/109563.
Pawar JN, Shete RT, Gangurde AB, Moravkar KK, Javeer SD, Jaiswar DR, Amin PD. Development of amorphous dispersions of artemether with hydrophilic polymers via spray drying: physicochemical and in silico studies. Asian J Pharm Sci. 2016;11(3):385–95.
Drugbank profile of artemether. https://www.drugbank.ca/drugs/DB06697. Accessed 20 Nov 2017.
Drugbank profile of artesunate. https://www.drugbank.ca/drugs/DB09274. Accessed 20 Nov 2017.
Ledeţi I, Murariu M, Vlase G, Vlase T, Doca N, Ledeţi A, Şuta L-M, Olariu T. Investigation of thermal-induced decomposition of iodoform. J Therm Anal Calorim. 2017;127(1):565–70.
Ledeti I, Vlase G, Vlase T, Murariu M, Trandafirescu C, Soica C, Suta LM, Dehelean C, Ledeti A. Non-isothermal isoconversional kinetic study regarding the degradation of albendazole. Rev Chim. 2016;67(3):549–52.
Suta LM, Vlase G, Ledeti A, Vlase T, Matusz P, Trandafirescu C, Circioban D, Olariu S, Ivan C, Murariu MS, Stelea L, Ledeti I. Solid-state thermal behaviour of cholic acid. Rev Chim. 2016;67(2):329–31.
Buda V, Andor M, Ledeti A, Ledeti I, Vlase G, Vlase T, Cristescu C, Voicu M, Suciu L, Tomescu M. Comparative solid-state stability of perindopril active substance vs. pharmaceutical formulation. Int J Mol Sci. 2017;18(1):164.
Serra R, Sempere J, Nomen R. A new method for the kinetic study of thermoanalytical data. Thermochim Acta. 1998;316(1):37–45.
Sempere J, Nomen R, Serra R, Soravilla J. The NPK method. Thermochim Acta. 2002;388(1–2):407–14.
Vlase T, Vlase G, Birta N, Doca N. Comparative results of kinetic data obtained with different methods for complex decomposition steps. J Therm Anal Calorim. 2007;88(3):631–5.
Ozawa T. A new method of analyzing thermogravimetric data. Bull Chem Soc Jpn. 1965;38(11):1881–6.
Ozawa T. Kinetic analysis of derivative curves in thermal analysis. J Therm Anal. 1970;2(3):301–24.
Flynn JH, Wall LA. A quick, direct method for the determination of activation energy from thermogravimetric data. J Polym Sci Part B Polym Lett. 1966;4(5):323–8.
Kissinger HE. Reaction kinetics in differential thermal analysis. Anal Chem. 1957;29(11):1702–6.
Akahira T, Sunose T. Research report, trans joint convention of four electrical institutes. Chiba Inst Technol (Sci Technol). 1971;16:22–31.
Friedman HL. New methods for evaluating kinetic parameters from thermal analysis data. J Polym Sci Part B Polym Lett. 1969;7(1):41–6.
Sempere J, Nomen R, Serra R. Progress in non-parametric kinetics. J Therm Anal Calorim. 1999;56(2):843–9.
Sestak J, Berggren G. Study of the kinetics of the mechanism of solid-state reactions at increasing temperatures. Thermochim Acta. 1971;3(1):1–12.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Circioban, D., Ledeţi, I., Vlase, G. et al. Kinetics of heterogeneous-induced degradation for artesunate and artemether. J Therm Anal Calorim 134, 749–756 (2018). https://doi.org/10.1007/s10973-018-7257-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-018-7257-0