Abstract
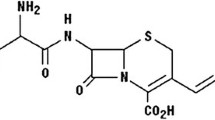
The optimization of the radiolabeling yield of cefazolin with 99mTc was described. Dependence of the labeling yield of 99mTc-cefazolin complex on the amounts of cefazolin and SnCl2·2H2O, pH and reaction time was studied. Cefazolin was labeled with 99mTc with a labeling yield of 89.5 % by using 1 mg cefazolin, 5 μg SnCl2·2H2O at pH 4 and 30 min reaction time. The radiochemical purity of 99mTc-cefazolin was evaluated with ITLC. The formed 99mTc-cefazolin complex was stable for a time up to 3 h, after that the labeling yield decreased 64.0 % at 8 h. Biological distribution of 99mTc-cefazolin complex was investigated in experimentally induced inflammation mice, in the left thigh, using Staphylococcus aureus (bacterial infection model) and turpentine oil (sterile inflammation model). Both thighs of the mice were dissected and counted and the ratio of bacterial infected thigh/contralateral thigh was then evaluated. In case of bacterial infection, T/NT for 99mTc-cefazolin complex was 8.57 ± 0.4 after 0.5 h, which was higher than that of the commercially available 99mTc-ciprofloxacin under the same experimental conditions. The ability of 99mTc-cefazolin to differentiate between septic and aseptic inflammation indicates that 99mTc-cefazolin could undergo further clinical trials to be used for imaging sites of infection.







Similar content being viewed by others
References
Lima JET, Maliska C, Goncalves MRB, Lima RC, Corbo R (2004) World J Nucl Med 3:284
Britton KE, Vinjamuri S, Hall AV (1997) Eur J Nucl Med 24:553–555
Rennen HJM, Boerman OC, Oyen WJG, Corstens FHM (2004) Med Chem Rev 1:27–30
Das SS, Hall AV, Wareham DDW, Britton KE (2002) Braz Arch Biol Technol 45:25
Lupetti A, Welling MM, Paulwels EKJ, Nibbering PH (2003) Lancet Infect Dis 3:223–226
Seabold JE, Palestro CJ, Brown ML (1997) J Nucl Med 38:994–997
Seabold JE, Forstrom LA, Schauwecher DS (1997) J Nucl Med 38:997–999
Schrijver MD, Streule K, Senekowitsch R (1987) Nucl Med Commun 8:895–897
Buscombe JR, Miller RF, Lui D (1991) Nucl Med Commun 12:583–587
McAfee JG, Gagne G, Subramanian G (1991) J Nucl Med 32:2126
Akhtar MS, Qaisar A, Irfanullah J (2005) J Nucl Med 46:567–568
Akhtar MS, Iqbal J, Khan MA (2004) J Nucl Med 45:849–851
Nibbering PH, Welling MM, Paulusma-Annema A (2004) J Nucl Med 45:321–325
Rennen HJ, Boerman OC, Oyen WJ, Corstens FH (2001) Eur J Nucl Med 28:241–244
Boerman OC, Laverman P, Oyen WJ, Corstens FH, Storm G (2000) Prog Lipid Res 39:461–463
Erdogan S, Ozer AY, Ercan MT, Hincal AA (2000) J Microencapsul 17:459–461
Singh AK, Verma J, Bhatnagar A, Ali AW (2003) J Nucl Med 2:103–106
Martin-Comin J, Soroa V, Rabiller G, Galli R, Cuesta L, Roca M (2004) Rev Esp Med Nucl 23:357–358
Gomes Barreto V, Rabiller G, Iglesias F, Soroa V, Tubau F, Roca M (2005) Rev Esp Med Nucl 24:312–315
Motaleb MA (2007) J Radioanal Nucl Chem 272:95–97
Welling MM, Paulusma-Annema A, Balter HS, Pauwels EKJ, Nibbering PH (2000) Eur J Nucl Med 27:292
Fournier B, Zhao X, Lu T, Drlica K, Hooper DC (2000) Antimicrob Agents Chemother 44:2160
Sonmezoglu K, Sonmezoglu M, Halac M (2001) J Nucl Med 42:567
Yapar Z, Kibar M, Yapar AF, Togrul E, Kayaselcuk U (2001) Eur J Nucl Med 28:822–825
Larikka MJ, Ahonen AK, Niemela O (2002) Nucl Med Commun 23:167–169
Dumarey N, Blocklet D, Appelboom T, Tant L, Schoutens A (2002) Eur J Nucl Med 29:530–535
Welling MM, Lupetti A, Balter HS (2001) J Nucl Med 42:788–790
Sarda L, Saleh-Mghir A, Peker C, Meulemans A, Cremieux AC, Leguludec D (2002) J Nucl Med 43:239–240
Rien HS, Huub JR, OTTO CB, Rudi D, Guido S (2004) J Nucl Med 42:2088–2090
Seung JO, Jin SR, Joong WS, Eun JY, Hyun JH (2002) Appl Radiat Isot 57:193–195
Sarda L, Cremieux AC, Lebellec Y, Meulemans A, Lebtahi R (2003) J Nucl Med 44:920–925
El-Ghany EA, Amine AM, El-Kawy OA, Amin M (2007) J Label Compd Radiopharm 50:25–29
El-Ghany EA, El-Kolaly MT, Amine AM, El-Sayed AS, Abdel-Gelil F (2005) J Radioanal Nucl Chem 266:131–135
Ibrahim IT, Motaleb MA, Attalah KM (2010) J Radioanal Nucl Chem 285(3):431–436
Motaleb MA (2007) J Radioanal Nucl Chem 272:167–171
Motaleb MA (2009) J Label Compd Radiopharm 52:415–418
Yurt Lambrecht F, Durkan K, Unak P (2008) J Radioanal Nucl Chem 275:161–166
Shah SQ, Khan MR, Khan AU (2011) Radiochim Acta 99:53–58
Mostafa M, Motaleb MA, Sakr TM (2010) Appl Radiat Isot 68:1959–1963
Motaleb MA, Alabdullah ES, Zaghary WA (2011) J Radioanal Nucl Chem 287:61–67
Barreto VG, Iglesias F, Roca M, Tubau F, Martin Comin J (2000) Rev Esp Med Nucl 19:479
Roohi S, Mushtaq A, Jehangir M, Malik SA (2006) J Radioanal Nucl Chem 267:561
Vallee F, Lebel M (1991) J Antimicrob Agents Chemother 35:2057
Mirshojaei SF, Gandomkar M, Najafi R, Sadat Ebrahimi SE, Babaei MH, Shafiei A, Talebi MH (2011) J Radioanal Nucl Chem 287:21–25
Motaleb MA (2010) J Label Compd Radiopharm 53:104–109
Ibrahim IT, Motaleb MA, Attalah KM (2010) J Label Compd Radiopharm 50:25–29
Shah SQ, Khan AU, Khan MR (2010) Appl Radiat Isot 68:2255–2260
Meral T, Erean T, Isil SU (1992) J Nucl Med Biol 19:802–806
Imen E, Wafa G, Nadia MS, Mouldi S (2010) J Nucl Med Biol 37:821–829
Acknowledgments
This work was made by the generous and encouragement of the staff of Radioisotopes Production Division, Hot Laboratories Center, Atomic Energy Authority, Cairo-Egypt. The author would like to thank the referees and the Editor of the Journal of Radioanalytical and Nuclear Chemistry for their valuable notices. Many thanks to Prof. K. Farah for her review and advices.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
El-Tawoosy, M. Preparation and biological distribution of 99mTc-cefazolin complex, a novel agent for detecting sites of infection. J Radioanal Nucl Chem 298, 1215–1220 (2013). https://doi.org/10.1007/s10967-013-2593-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-013-2593-9