Abstract
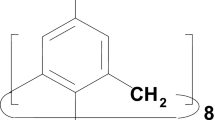
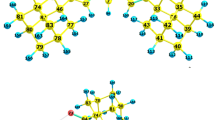
The cone conformation of C 4 symmetry is shown by the Hartree-Fock method (3-21G basis) to be the predominant conformer of calix[4]arene; the compressed cone of C 2 symmetry is the major conformer of calix[6]arene. Using quantum chemical methods we calculated hydrogen bond cleavage energies for calix[4]-(ab initio and density functional methods) and calix[6]arene (ab initio), and also for the complex of calix[4]arene with carbon disulfide. These energies along with structural data point to the cooperative effect of hydrogen bonds. The results of these studies provided an explanation to the greater conformational lability of calix[6]arene compared with calix[4]arene molecules. It is also predicted that the nucleophilic substitution reaction involving calix[6]arene in the presence of weak bases and in aprotic solvents, as well as in the gas phase, will occur via diastereomeric transition states.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
P. D. J. Grootenhuis, P. A. Kollman, L. C. Groenen, et al., J. Am. Chem. Soc., 112, 4165–4176 (1990).
S. Fischer, P. D. J. Grootenhuis, L. C. Groenen, et al., ibid., 117, 1611–1620 (1995).
W. P. van Hoorn, F. C. J. M. van Veggel, and D. N. Reinhoudt, J. Org. Chem., 61, No. 20, 7180–7184 (1996).
I. Thondorf and J. Brenn, J. Mol. Struct. (Theochem), 398/399, 307–314 (1997).
W. P. van Hoorn, M. G. H. Morshuis, F. C. J. M. van Veggel, and D. N. Reinhoudt, J. Phys. Chem. A, 102, 1130–1138 (1998).
R. J. Bernardino and B. J. C. Cabral, ibid., 103, 9080–9085 (1999).
J. Schatz, A. C. Backes, and H.-U. Siehl, J. Chem. Soc., Perkin Trans. 2, 609/610 (2000).
B. P. Hay, J. B. Nicholas, and D. Feller, J. Am. Chem. Soc., 122, 10083–10089 (2000).
R. J. Bernardino and B. J. C. Cabral, J. Mol. Struct. (Theochem), 549, 253–260 (2001).
L. Frkanec, A. Visnjevac, B. Kojic-Prodic, and M. Zinic, Chem. Eur. J., 6, No. 3, 442–453 (2000).
D. M. Rudkevich, ibid., No. 15, 2679–2686.
A. N. Novikov, V. A. Bacherikov, and A. I. Gren, Zh. Strukt. Khim., 42, No. 6, 1806–1096 (2001).
A. N. Novikov, V. A. Bacherikov, and A. I. Gren, Zh. Obshch. Khim., 72, No. 9, 1481–1486 (2002).
M. I. Ogden, A. L. Rohl, and J. D. Gale, Chem. Commun., 1626/1627 (2001).
N. D. Sokolov (ed.), The Hydrogen Bond [in Russian], Nauka, Moscow (1981).
P. Lipkowski, A. Koll, A. Karpfen, and P. Wolschann, Chem. Phys. Lett., 360, 256–263 (2002).
M. Lozynski, D. Rusinska-Roszak, and H.-G. Mack, J. Phys. Chem. A, 101, No. 8, 1542–1548 (1997).
A. D. Becke, Phys. Rev. A, 38, 3098 (1998).
P. M. W. Gill, Mol. Phys., 89, 433 (1996).
C. Lee, W. Yang, and R. G. Parr, Phys. Rev. B, 37, 785 (1988).
J. P. Perdew, K. Burke, and M. Ernzerhof, Phys. Rev. Lett., 77, 3865 (1996).
M. W. Schmidt, K. K. Baldridge, J. A. Boatz, et al., J. Comput. Chem., 14, No. 11, 1347–1363 (1993).
A. Granovskii, PC GAMESS Software Package [in Russian], Versions 4.3, 6.3, and 6.4.
P. F. Flükiger, Molekel, Molecular Visualization Software, University of Geneva (http://igc.ethz.ch/molekel).
J. Rak, P. Skurski, M. Gutowski, et al., J. Phys. Chem. A, 101, 283–292 (1997).
N. Iki, N. Morohashi, T. Suzuki, et al., Tetrahedron Lett., 41, 2587–2590 (2000).
R. Ludwig, Chem. Phys. Chem., No. 1, 53–56 (2000).
P. Schuster and P. Wolschann, Universität Wien (www.tbi.univie.ac.at/∼pks/PUBL/99-pks-007.pdf).
K. Ebert and H. Ederer, in: Computer Applications in Chemistry, VCH, Weinheim (1989).
A. S. Dneprovskii and T. I. Temnikova, Theoretical Principles of Organic Chemistry [in Russian], Khimiya, Leningrad (1991).
N. S. Akhmetov, General and Inorganic Chemistry [in Russian], Vysshaya Shkola, Moscow (1988).
H. Becker, W. Berger, G. Domschke, E. Fanghanel, et al., Organicum, VEB Deutscher Verlag der Wissenschaften, Berlin (1977).
L. V. Gurvich, G. V. Karachevtsev, V. N. Kondratiev, et al., Ionization Potentials, in: Chemical Bond Cleavage Energies. Ionization Potentials and Electron Affinities [in Russian], Nauka, Moscow (1974).
M. Nogradi, Stereochemistry. Basic Concepts and Applications, Pergamon, New York (1980).
Author information
Authors and Affiliations
Additional information
Original Russian Text Copyright © 2006 by A. N. Novikov, V. A. Bacherikov, Yu. E. Shapiro, and A. I. Gren
__________
Translated from Zhurnal Strukturnoi Khimii, Vol. 47, No. 6, pp. 1015–1027, November–December, 2006.
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Novikov, A.N., Bacherikov, V.A., Shapiro, Y.E. et al. Ab initio and density functional studies of cooperative hydrogen bonding in calix[4]-and calix[6]arenes. J Struct Chem 47, 1003–1015 (2006). https://doi.org/10.1007/s10947-006-0419-0
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/s10947-006-0419-0