Abstract
We investigate how synchrony can be generated or induced in networks of electrically coupled integrate-and-fire neurons subject to noisy and heterogeneous inputs. Using analytical tools, we find that in a network under constant external inputs, synchrony can appear via a Hopf bifurcation from the asynchronous state to an oscillatory state. In a homogeneous net work, in the oscillatory state all neurons fire in synchrony, while in a heterogeneous network synchrony is looser, many neurons skipping cycles of the oscillation. If the transmission of action potentials via the electrical synapses is effectively excitatory, the Hopf bifurcation is supercritical, while effectively inhibitory transmission due to pronounced hyperpolarization leads to a subcritical bifurcation. In the latter case, the network exhibits bistability between an asynchronous state and an oscillatory state where all the neurons fire in synchrony. Finally we show that for time-varying external inputs, electrical coupling enhances the synchronization in an asynchronous network via a resonance at the firing-rate frequency.


















Similar content being viewed by others
Notes
Clearly, the amount of charge transferred during any portion of the spike is proportional to γ gap . Here we deliberately treat β gap and γ gap as independent parameters to be able to consider the case γ gap = 0 while β gap ≠ 0.
References
Abbott, L. F., & van Vreeswijk, C. (1993). Asynchronous states in a network of pulse-coupled oscillators. Physical Review, E, 48, 1483–1490.
Amit, D., & Brunel, N. (1997). Model of global spontaneous activity and local structured delay activity during delay periods in the cerebral cortex. Cerebral Cortex, 7, 237–252.
Beierlein, M., Gibson, J. R., & Connors, B. (2000). A network of electrically coupled interneurons drives synchronized inhibition in neocortex. Nature Neuroscience, 3, 904–909
Bem, T., Feuvre, Y. L., Rinzel, J., & Meyrand, P. (2005). Electrical coupling induces bistability of rhythms in networks of inhibitory spiking neurons. European Journal of Neuroscience, 22, 2661–2668.
Bennett, M., & Zukin, R. (2004). Electrical coupling and neuronal synchronization in the mammalian brain. Neuron, 41, 495–511.
Brunel, N. (2000). Dynamics of sparsely connected networks of excitatory and inhibitory spiking neurons. Journal of Computational Neuroscience, 8, 183–208.
Brunel, N., Chance, F., Fourcaud, N., & Abbott, L. (2001). Effects of synaptic noise and filtering on the frequency response of spiking neurons. Physical Review Letters, 86, 2186–2189.
Brunel, N., & Hakim, V. (1999). Fast global oscillations in networks of integrate-and-fire neurons with low firing rates. Neural Computation, 11, 162–1671.
Brunel, N., & Hakim, V. (2008). Sparsely synchronized neuronal oscillations. Chaos, 18, 015113.
Brunel, N., & Hansel, D. (2006). How noise affects the synchronization properties of reccurent networks of inhibitory neurons. Neural Computation, 18, 1066–1110.
Chow, C. C., & Kopell, N. (2000). Dynamics of spiking neurons with electrical coupling. Neural Computation, 12, 1643–1678.
Connors, B., & Long, M. (2004). Electrical synapses in the mammalian brain. Annual Review of Neuroscience, 27, 393–418.
Coombes, S., & Zachariou, M. (2008). Gap junctions and emergent rhythms. In J. Rubin, R. Matias (Ed.), Coherent behavior in neuronal networks, computational neuroscience series. New York: Springer.
Coombes, S. (2008). Neuronal networks with gap junctions: A study of piece-wise linear planar neuron models. SIAM Journal on Applied Dynamical Systems, 7, 1101–1129.
Draguhn, A., Traub, R., Schmitz, D., & Jefferys, J. (1998). Electrical coupling underlies high-frequency oscillations in the hippocampus in vitro. Nature, 394, 189–192.
Dugué G., Brunel N., Hakim V., Schwartz E., Chat M., Levesque M., et al. (2008). Electrical coupling mediates tunable low-frequency oscillations and resonance in the cerebellar Golgi network. Neuron (in press).
Fourcaud-Trocmé, N., Hansel, D., van Vreeswijk, C., & Brunel, N. (2003). How spike generation mechanisms determine the neuronal response to fluctuating inputs. Journal of Neuroscience, 23, 11628–11640.
Fukuda, T., & Kosaka, T. (2000). Gap-junction coupling linking the dendritic network of gabaergic neurons in the hippocampus. Journal of Neuroscience, 20, 1519–1528.
Galarreta, M., Erdelyi, F., Szabo, G., & Hestrin, S. (2004). Electrical coupling among irregular-spiking gabaergic interneurons expressing cannabinoid receptors. Journal of Neuroscience, 24, 9770–9778.
Galarreta, M., & Hestrin, S. (1999). A network of fast-spiking cells in the neocortex connected by electrical synapses. Nature, 402, 72–75.
Galarreta, M., & Hestrin, S. (2001a). Electrical synapses between gaba-releasing interneurons. Nature Reviews, Neuroscience, 2, 425–433.
Galarreta, M., & Hestrin, S. (2001b). Spike transmission and synchrony detection in networks of gabaergic interneurons. Science, 292, 2295–2299.
Galarreta, M., & Hestrin, S. (2002). Electrical and chemical synapses among parvalbumin fast-spiking gabaergic interneurons in adult mouse neocortex. Proceedings of the National Academy of Sciences of the United States of America, 00, 12438–12443.
Gerstner, W., & van Hemmen, J. L. (1993). Coherence and incoherence in a globally coupled ensemble of pulse-emitting units. Physical Review Letters, 71, 312–315.
Gibson, J. R., Beierlein, M., & Connors, B. (1999). Two networks of electrically coupled inhibitory neurons in neocortex. Nature, 402, 75–79.
Hestrin, S., & Galarreta, M. (2005). Electrical synapses define networks of neocortical gabaergic neurons. Trends in Neuroscience, 28, 304–309.
Kopell, N., & Ermentrout, B. (2004). Chemical and electrical synapses perform complementary roles in the synchronization of interneuronal networks. Proceedings of the National Academy of Sciences of the United States of America, 101, 15482–15487.
Landisman, C., Long, M., Beierlein, M., Deans, M., Paul, D. & Connors, B. (2002). Electrical synapses in the thalamic reticular nucleus. Journal of Neuroscience, 22, 1002–1009.
LeBeau, F., Traub, R., Monyer, H., Whittington, M., & Buhl, E. (2003). The role of electrical signaling via gap junctions in the generation of fast network oscillations. Brain Research Bulletin, 62, 3–13.
Lewis, T. J., & Rinzel, J. (2003). Dynamics of spiking neurons connected by both inhibitory and electrical coupling. Journal of Computational Neuroscience, 14, 283–309.
Mann-Metzer, P., & Yarom, Y. (1999). Electrotonic coupling interacts with intrinsic properties to generate synchronized activity in cerebellar networks of inhibitory interneurons. Journal of Neuroscience, 19, 3298–3306.
Pfeuty, B., Mato, G., Golomb, D., & Hansel, D. (2003). Electrical synapses and synchrony: The role of intrinsic currents. Journal of Neuroscience, 23, 6280–6294.
Pfeuty, B., Mato, G., Golomb, D., & Hansel, D. (2005). The combined effects of inhibitory and electrical synapses in synchrony. Neural Computation, 17, 633–670.
Risken, H. (1984). The Fokker Planck equation: methods of solution and applications. New York: Springer.
Schneider A. R., Lewis T. J., & Rinzel J. (2006). Effects of correlated input and electrical coupling on synchrony in fast-spiking cell networks. Neurocomputing, 69, 1125–1129
Sherman, A., & Rinzel, J. (1992). Proceedings of the National Academy of Sciences of the United States of America, 89, 2471–2474.
Skinner F. K., Zhang L., Perez Velazquez J.L., & Carlen P. L. (1999). Bursting in Inhibitory Interneuronal Networks: A Role for Gap-Junctional Coupling. Journal of Neurophysiology, 81, 1274–1283.
Tamas, G., Buhl, E., Lorincz, A., & Somogyi, P. (2000). Proximally targeted gabaergic synapses and gap junctions synchronize cortical interneurons. Nature Neuroscience, 3, 366–371.
Timme, M., Wolf, F., & Geisel, T. (2002). Coexistence of regular and irregular dynamics in complex networks of pulse-coupled oscillators. Physical Review Letters, 89, 258701.
Traub, R., Kopell, N., Bibbig, A., Buhl, E., LeBeau, F., & Whittington, M. (2001). Gap junctions between interneuron dendrites can enhance synchrony of gamma oscillations in distributed networks. Journal of Neuroscience, 21, 9478–86.
Tuckwell, H. (1988). Introduction to theoretical neurobiology. Cambridge: Cambridge University Press.
Venance, L., Rozov, A., Blatow, M., Burnashev, N., Feldmeyer, D., & Monyer, H. (2000). Connexin expression in electrically coupled postnatal rat brain neurons. Proceedings of the National Academy of Sciences of the United States of America, 97, 10260–10265.
Acknowledgements
This work was supported by the Agence Nationale de la Recherche grant ANR-05-NEUR-030.
Author information
Authors and Affiliations
Corresponding author
Additional information
Action Editor: Alain Destexhe
Appendix
Appendix
1.1 A Networks of exponential integrate-and-fire neurons
In order to study the dynamics analytically, we used a model based on leaky integrate-and-fire (LIF) neurons. An important caveat of this approach is that the voltage trace during the action potential is not modeled. To account for electrotonic coupling during the action potential, we assumed that the transmission of the depolarizing part is instantaneous. While this approximation is supported by experimental measurements in fast-spiking cells (Galarreta and Hestrin 2001a), it is important to show that the obtained results, and in particular the bistability between synchrony and asynchrony, are not artifacts of this approximation. In this section, we consider the exponential integrate-and-fire model, which includes a spike-generating sodium current, and thus produces full traces of action potentials. We briefly consider the spikelets produced in this model by the transmission of the action potentials through electrical synapses, and then show that bistability between synchrony and asynchrony is still found in this model.
The exponential integrate-and-fire model is a non-linear integrate-and-fire model that has been shown to approximate remarkably well the action potential shape, and the dynamic properties of more complex conductance-based models such as the Hodgkin-Huxley model (Fourcaud-Trocmé et al. 2003). The dy namics of the membrane potential of neuron i are given by
This model includes an exponential spike-generating current: once the membrane potential crosses the threshold V T , it diverges to infinity in finite time. This divergence represents the firing of an action potential. After the divergence, in the original EIF model, the membrane potential is reset instantaneously to V r . Since this gives an unrealistic spike shape, we reset the membrane potential to V r following a linear trajectory during a refractory period τ rp , as was done in Dugué et al. (2008).
One advantage of the EIF model over more complicated models is that it provides a way of modulating the shape of the generated action potential by modifying a single parameter Δ T . The parameter Δ T determines the sharpness of spike-initiation: the larger Δ T , the slower the spike initiation, and the wider the full spike trace. The width of the hyperpolarizing part of the spike can be controlled via the refractory period τ rp . In Fig. 19 we illustrate the effect on a postsynaptic cell of spikes of different width: a wide spike leads to a predominantly excitatory spikelet, while a narrow one elicits a mostly inhibitory spikelet. Note that the shapes of the spikelets are very similar to the ones obtained in the simplified, leaky integrate-and-fire model (cf. Fig. 1).
Spikelets elicited in a post-synaptic cell by the transmission through an electrical synapse of a pre-synaptic spike, obtained from the exponential integrate-and-fire model for two different sets of coupling parameters. (A) A wide presynaptic spike leads to a dominantly excitatory spikelet (V T = 5.1 mV, Δ T = 4 mV, τ rp = 3 ms, V r = − 10 mV, holding potential 3 mV); (B) a narrow presynaptic spike leads to a dominantly inhibitory spikelet (Δ T = 1 mV, τ rp = 1 ms, V r = − 10 mV, holding potential 0 mV)
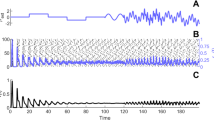
In Fig. 20, we illustrate the fact that bistability between synchrony and asynchrony is generically found in networks of exponential integrate-and-fire neurons. We display the activity of a network consisting of N = 50 neurons. In such a small system, the activity switches spontaneously between the asynchronous and the synchronous state. In this particular example, the firing of neurons in the synchronous state is not as highly coordinated as in the case of LIF neurons displayed in Fig. 9, but the two states can be clearly distinguished.
Bistability in a network of exponential integrate-and-fire neurons: spontaneous transitions between synchrony and asynchrony in a network of N = 50 neurons. (A) Spike raster of the full network; (B) instantaneous population firing rate ν(t) computed in 1 ms bins. The parameters used in the simulation are: μ ext = 3 mV, σ = 0.47 mV, g c = 0.5, V T = 5.1 mV, Δ T = 1 mV, V r = − 10 mV, τ rp = 1.7 ms
1.2 B Linear stability of the asynchronous state in the homogeneous case
The probability distribution P(V,t) obeys
where \(\mu_{syn}=g_c\langle V \rangle+\beta \tau \nu(t)\). The normalization and boundary conditions are:
Multiplying Eq. (37) by V and integrating, we obtain an equation for the evolution of the mean membrane potential \(\langle V \rangle\):
From this point on, the analysis follows the same steps as in Brunel and Hakim (1999). We use the rescaled variables v(t), n(t) and m syn (t) defined by:
With these notations, Eq. (37) becomes
where the linear operator \(\mathcal{L}\) is defined as
and the boundary conditions read
1.2.1 B.1 Stationary state
The steady-state solution of Eq. (46) is given by
The stationary mean membrane potential V 0 is given by Eq. (42):
The stationary firing rate ν 0 is then obtained from the normalization condition Eq. (38):
As y r and y th depend on ν 0, Eq. (52) is an implicit equation which can be solved self-consistently for ν 0.
1.2.2 B.2 Linear stability
In the following, we expand the time varying quantities perturbatively at successive orders:
At first order Eq. (46) becomes
The eigenmodes of Eq. (54) are of the form
As v(t) and n(t) are linearly related via Eq. (42)
and
with
\(\hat Q_1\) therefore obeys the ordinary differential equation
together with the boundary conditions
similar conditions at y r , and the integrability requirement
The general solution of Eq. (58) can be written as
where
and ϕ 1,2 are two independent solutions of the homogeneous equation
Solutions of Eq. (63) are obtained in terms of confluent hypergeometric functions (Brunel and Hakim 1999):
The boundary conditions Eq. (59) give expressions for \(\alpha_1^{+},\alpha_1^{-},\beta_1^{+}\) and \(\beta_1^{-}\) as functions of λ:
where
The eigenvalues are determined by the requirement (60) that \(\hat Q_1(y,\lambda)\) be integrable, which corresponds to \(\alpha_1^{-}=0\), i.e.
or
with
1.2.3 B.3 Weakly non-linear analysis
Pushing the development in Eq. (53) to higher orders, it is possible to determine the non-linear contribution to the oscillations in the vicinity of the bifurcation. The n-th order terms obey inhomogeneous linear equations with forcing terms formed by quadratic combinations of lowest-order terms. The dynamics of the first-order oscillation amplitude \(\hat n_1\) is obtained from a self-consistancy condition on the third order terms, and read
The procedure is identical to the one followed in Brunel and Hakim (1999), we therefore only give here the expressions necessary for evaluating B, and for more details refer the reader to Brunel and Hakim (1999). The results of Brunel and Hakim (1999) are recovered (in the fully connected case H = 0) by replacing below \(\bar R_g(\lambda)\) with \(-G_c e^{-\delta \lambda}\).
In the following, we write
B.3.1 Second order
The second order terms oscillate at frequencies 0 and 2ω c , and can be written as:
\(\hat Q_{2,2}(y)\) can be expressed as
where:
Similarly, \(\hat Q_{2,0}(y)\) can be expressed as
where:
B.3.2 Third order
The third order terms oscillate at frequencies ω c and 3 ω c :
The part \(\hat Q_{3,1}(y)\) of Q 3(y,t) oscillating at frequency ω c can be written as
with
where
and
B.3.3 Final expressions
The constants A and B determining the non-linear dynamics of \(\hat n_1\) read:
Note that the denominator in these expressions can be rewritten as
1.3 C The heterogeneous case
We consider a distribution of input currents such that neurons in the network receive a mean external current \(\mu_{ext}=\bar \mu_{ext} + z\, \delta \mu\) with probability η(z). Taking into account the interactions, the total current received by a neuron becomes
where \({\bar \nu}\) and \(\bar{\langle V \rangle}\) are the mean firing rate and membrane potential in the network, averaged over the heterogeneity. We denote by \(\langle \, \rangle\) the average over neurons with the same z, and by a bar the average over different z.
1.3.1 C.1 Stationary state
In the stationary state, the firing rate of the neuron with total input \(\mu_{tot, 0}(z)={\bar \mu}_{ext} + z\, \delta \mu + \beta \tau \bar \nu_0 +g_c \bar V_0\) is given by
where μ tot, 0(z) depends on the stationary average firing rate \(\bar \nu_0\) and mean membrane potential \(\bar V_0\), which are unknown at this stage.
Averaging over heterogeneity, we get
These equations can be solved self-consistently for \(\bar \nu_0\) and \(\bar V_0\). The distribution ρ of firing rates in the network is then obtained as
1.3.2 C.2 Linear stability
To determine the linear stability of the asynchronous state, we follow the same steps as in Appendix A. We start by linearizing all the time-dependent quantities around their stationary value:
where \({\bar n(t)}=\frac{1}{{\bar \nu_0}}\int dz\,\eta(z)\nu_0(z)n(z,t)\).
At first order, after expanding Q 1, v, n and \(\bar{n}\) in eigenmodes, the Foker-Planck equation becomes
with boundary conditions
The integrability condition for each z reads:
where \(R_n(\lambda,z)= \frac{\tau \nu_0(z)}{\sigma}\frac{1}{1+\lambda} \frac{\frac{\partial U}{\partial y}(y_{th},\lambda)-\frac{\partial U}{\partial y}(y_{r},\lambda)}{U(y_{th})-U(y_{r})}\)
Multiplying both sides of Eq. (112) by η(z) ν 0(z), and integrating over z, we get
with
1.4 D Response to external oscillations
We consider the situation where the external current varies in time as μ ext (t) = μ ext,0 + μ ext,1(t), with \(\mu_{ext,1}(t)=\tilde m_{ext}(\lambda)e^{\lambda t/\tau}+c.c.\), and λ = iω. In that case
so that Eq. (58) becomes
The condition \(\alpha_1^{-}\) then gives
Rearranging the terms, we obtain
Rights and permissions
About this article
Cite this article
Ostojic, S., Brunel, N. & Hakim, V. Synchronization properties of networks of electrically coupled neurons in the presence of noise and heterogeneities. J Comput Neurosci 26, 369–392 (2009). https://doi.org/10.1007/s10827-008-0117-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10827-008-0117-3