Abstract
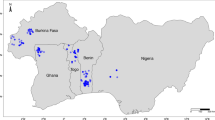
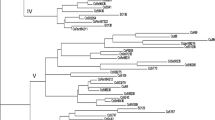
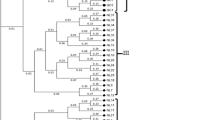
Twenty landraces of cowpea (Vigna unguiculata (L.) Walp.) scattered throughout Algeria were compared through morphological and genetic characterization. At the morphological level, for qualitative characters there was no intra-landrace variation and for quantitative characters the variations were low except for landrace NAG2 Three different cultigroups were located in Algeria: Biflora that was dominant in the Sahara, Melanophtalmus in the North and Unguiculata including one landrace in Kabylia and two in Sahara. The AMOVA analysis indicated that the genetic variation was lower within than among agro-ecological regions. A Mantel test, revealed a correlation between the qualitative morphological data and the geographical data (R = 0.28; P < 0.01), indicating that the degree of morphological change among landraces was roughly proportional to the geographical distances separating them. Genetic diversity was analyzed by using 11 random amplified polymorphic DNA (RAPD) and 12 inter-simple sequence repeat (ISSR) markers. No intra-landrace variability was found. The eleven RAPD primers yielded 77 bands, of which 45 (58.44%) were polymorphic; the genetic similarity ranged from 66.0 to 96.7%. The twelve ISSR primers provided a total of 104 bands, of which 65 (62.5%) were polymorphic; the genetic similarity ranged from 62.8 to 97.8%. cluster analysis showed a good match between genetic background and geographical distribution, which was confirmed by the results of the Mantel test. In particular, geographical data and genetic data were found to be correlated: (R = 0.33; P < 0.01) for RAPD, (R = 0.37; P < 0.01) for ISSR, and (R = 0.33; P < 0.01) for a combined RAPD-ISSR dataset. Moreover, despite the absence of significant correlation between morphological and RAPD data (R = 0.14; P = 0.14), significant correlations between morphological data and both ISSR (R = 0.27, P < 0.05) and a combined RAPD-ISSR dataset (R = 0.22, P < 0.05) were noted. ISSR markers were better linked to morphological variation than were RAPD markers. However, despite this, genetic distances among these landraces were found to be essentially the same no matter which markers were used.



Similar content being viewed by others
References
Ajibade SR, Weeden NF, Chite SM (2000) Inter simple sequence repeat analysis of genetic relationships in the genus Vigna. Euphytica 111:47–55
Anoun N, Echikh N (1990) Étude biosystématique d’une légumineuse saharienne: le Tadelaght. Mémoire DES. Univ. Sci. Technol. Houari Boumedienne. Bab Ezzouar, Algeria, 101 pp
Ba FS, Pasquet RS, Gepts P (2004) Genetic diversity in cowpea [Vigna unguiculata (L.) Walp.] as revealed by RAPD markers. Genet Resour Crop Evol 51:539–550
Badiane FA, Diouf D, Sané D, Diouf O, Goudiaby V, Diallo N (2004) Screening cowpea (Vigna unguiculata (L.) Walp.) varieties by inducing water deficit and RAPD analyses. Afr J Biotechnol 3:174–178
Blair MW, Panaud O, McCouch SR (1999) Inter-simple sequence repeat (ISSR) amplification for analysis of microsatellite motif frequency and fingerprinting in rice (Oryza sativa L.). Theor Appl Genet 98:780–792
Charters YM, Wilkinson MJ (2000) The use of self-pollinated progenies as ‘in-groups’ for the genetic characterization of cocoa germplasm. Theor Appl Genet 100:160–166
Chevalier A (1932) Productions végétales du Sahara. Rev Bot Appl Agric Trop 12:133–134
Chevalier A (1944) Le dolique de Chine en Afrique. Son histoire, ses affinités, les formes sauvages et cultivées. Son rôle dans l’alimentation indigène et en agriculture tropicale et sub-tropicale. Rev Bot Appl Agric Trop 24:128–152
Depeiges A, Goubely C, Lenoir A, Cocherel S, Picard G, Raynal M, Grellet F, Delseny M (1995) Identification of the most represented repeat motifs in Arabidopsis thaliana microsatellite loci. Theor Appl Genet 91:160–168
Dikshit HK, Jhang T, Singh NK, Koundal KR, Bansal KC, Chandra N, Tickoo JL, Sharma TR (2007) Genetic differentiation of Vigna species by RAPD, URP and SSR markers. Biol Plant 51:451–457
Diouf D, Hilu KW (2005) Microsatellites and RAPD markers to study genetic relationships among cowpea breeding lines and local varieties in Senegal. Genet Resour Crop Evol 52:1057–1067
Doebley J (1989) Isozymic evidence and the evolution of crop plants. In: Soltis DE, Soltis PS (eds) Isoenzymes in plant biology. Dioscorides Press, Portland, pp 165–191
Echikh N (2000) Organisation du pool génique de formes sauvages et cultivées d’une légumineuse alimentaire, Vigna unguiculata (L.) Walp. Ph.D. thesis, Faculté Universitaire des Sciences Agronomiques de Gembloux, Gembloux, Belgium, 307 pp
Ehlers JD, Hall AE (1997) Cowpea (Vigna unguiculata (L.) Walp.). Field Crops Res 53:187–204
Eloward HOA, Hall AE (1987) Influence of early and late nitrogen fertilization on yield and nitrogen fixation of cowpea under well-watered and dry field conditions. Field Crops Res 15:229–244
Fall L, Diouf D, Fall-Ndiaye MA, Badiane FA, Gueye M (2003) Genetic diversity in cowpea [Vigna unguiculata (L.) Walp.] varieties determined by ARA and RAPD techniques. Afr J Biotechnol 2:48–50
Fang DQ, Roose ML (1997) Identification of closely related citrus cultivars with inter-simple sequence repeat markers. Theor Appl Genet 95:408–417
Gillaspie AG, Hopkins MS, Dean RE (2005) Determining genetic diversity between lines of Vigna unguiculata subspecies by AFLP and SSR markers. Genet Resour Crop Evol 52:245–247
Goulao L, Valdiviesso T, Santana C, Oliveira CM (2001) Comparison between phenetic characterisation using RAPD and ISSR markers and phenotypic data of cultivated chestnut (Castanea sativa Mill.). Genet Resour Crop Evol 48:329–338
Hall AE, Patel PN (1985) Breeding of resistance to drought and heat. In: Singh SR, Rachie KO (eds) Cowpea research. Production and utilization. Wiley, New York, pp 137–151
International Board for Plant Genetic Resources (IBPGR) (1983) Cowpea descriptors. IBPGR, Rome (34 pp)
Jones CJ, Edwards KJ, Castaglione S, Winfield MO, Sala F, Van De Wiel C (1997) Reproducibility testing of RAPD, AFLP and SSR markers in plants by a network of European laboratories. Mol Breeding 3:381–390
Kwapata MB, Hall AE (1985) Effects of moisture regime and phosphorus on mycorrhizal infection, nutrient uptake and growth of cowpeas (Vigna unguiculata (L.) Walp.). Field Crops Res 12:241–250
Lakhanpaul S, Chadha S, Bhat KV (2000) Random amplified polymorphic DNA (RAPD) analysis in Indian mungbean [Vigna radiata (L.) Wilczek] cultivars. Genetica 109:227–234
Leakey CLA (1988) Genotypic and phenotypic markers in common bean. In: Gepts P (ed) Genetic resources of Phaseolus bean. Kluwer, Dordrecht, pp 245–327
Li CD, Fatokun CA, Ubi B, Singh BB, Scoles GJ (2001) Determining genetic similarities and relationships among cowpea breeding lines and cultivars by microsatellite markers. Crop Sci 41:189–197
Mantel N (1967) The detection of disease clustering and generalized regression approach. Cancer Res 27:209–220
Marotti I, Bonetti A, Minelli M, Catizone P, Dinelli G (2007) Characterization of some Italian common bean (Phaseolus vulgaris L.) landraces by RAPD, semi-random and ISSR molecular markers. Genet Resour Crop Evol 54:175–188
Massawe FJ, Roberts JA, Azam-Ali SN, Daved MR (2002) Genetic diversity in bambara groundnut (Vigna subterranea (L.) Verdc.) landraces assessed by random amplified polymorphic DNA (RAPD) markers. Genet Resour Crop Evol 50:737–741
Menendez CM, Hall AE, Gepts P (1997) A genetic linkage map of cowpea (Vigna unguiculata) developed from a cross between two inbred, domesticated lines. Theor Appl Genet 95:1210–1217
Muthusamy S, Kanagarajan S, Ponnusamy S (2008) Efficiency of RAPD and ISSR markers system in accessing genetic variation of rice bean (Vigna umbellata) landraces. Electr J Biotech 11(3): 10 pp. doi: 10.2225/vol11-issue3-fulltext-8
Ng NQ, Marechal R (1985) Cowpea taxonomy, origin and germplasm. In: Singh SR, Rachie KO (eds) Cowpea research, production and utilization. Wiley, New York, pp 11–21
Nkongolo KK (2003) Genetic characterization of Malawian cowpea (Vigna unguiculata (L.) Walp.) landraces: diversity and gene flow among accessions. Euphytica 129:219–228
Ouédraogo JT, Gowda BS, Jean M, Close TJ, Ehlers JD, Hall AE, Gillaspie AG, Roberts PA, Ismail AM, Bruening G, Gepts P, Timko MP, Belzile FJ (2002) An improved genetic linkage map for cowpea (Vigna unguiculata L.) combining AFLP, RFLP, RAPD, biochemical markers, and biological resistance traits. Genome 45:175–188
Padulosi S (1993) Genetic diversity, taxonomy and ecogeographic survey of the wild relatives of cowpea (Vigna unguiculata (L.) Walp.). Ph.D. dissertation, Universite’ catholique Louvain-la- Neuve, Belgium, 477 p
Pasquet R (1994) Organisation génétique et évolutive des formes spontanées et cultivées du niébé, Vigna unguiculata (L.) Walp. biosystématique et processus de domestication. Ph.D. thesis, Paris Grignon, I.N.A. Paris-Grignon-ORSTOM, France, 284 p
Pasquet RS (1998) Morphological study of cultivated cowpea Vigna unguiculata (L.) Walp. Importance of ovule number and definition of cv. gr Melanophthalmus. Agronomie 18:61–70
Pasquet RS (1999) Genetic relationships among subspecies of Vigna unguiculata (L.) Walp. based on allozyme variation. Theor Appl Genet 98:1104–1119
Pasquet RS (2000) Allozyme diversity of cultivated cowpea Vigna unguiculata (L.) Walp. Theor Appl Genet 101:211–219
Qian W, Ge S, Hong DY (2001) Genetic variation within and among populations of a wild rice Oryza granulata from China detected by RAPD and ISSR markers. Theor Appl Genet 102:440–449
Raina SN, Rani V, Kojima T, Ogihara Y, Singh KP, Devarumath RM (2001) RAPD and ISSR fingerprints as useful genetic markers for analysis of genetic diversity, varietal identification, and phylogenetic relationships in peanut (Arachis hypogaea) cultivars and wild species. Genome 44:763–772
Ranade R, Vaidya UJ, Kotwal SA, Bhagwat A, Gopalakrishna T (2000) Hybrid seed genotyping and plant varietal identification using DNA markers. In: DAE-BRNS symposium on the use of nuclear and molecular techniques in crop improvement, 6–8 December 2000, Mumbai, India, pp 338–345
Rosenberg MS (2001) Passage. pattern analysis, spatial statistics, and geographic exegesis. Version 1.1. Department of Biology, Arizona State University, Tempe
Sarutayophat T, Nualsri C, Santipracha Q, Saereeprasert V (2007) Characterization and genetic relatedness among 37 yardlong bean and cowpea accessions based on morphological characters and RAPD analysis. Songklanakarin J Sci Technol 29:591–600
Schneider S, Roessli D, Excoffier L (2000) Arlequin ver 2.000; a software for population genetics data analysis. Genetics and biometry laboratory, Dept of Anthropology and ecology, University of Geneva, Suisse
Singh SP, Nodari R, Gepts P (1991) Genetic diversity in cultivated common bean. l. Allozymes. Crop Sci 31:19–23
Singh BB, Chambliss OL, Sharma B (1997) Recent advances in cowpea breeding. In: Singh BB, Mohanraj DR, Dashiell KE, Jackai LEN (eds) Advances in cowpea research. IITA-JIRCAS, Ibadan, pp 30–49
Singh S, Reddy KS, Jawali N (2000) PCR analysis of mungbean genotypes using anchored simple sequence repeat primers. In: DAE-BRNS symposium on the use of nuclear and molecular techniques in crop Improvement. BARC, Mumbai, India, pp 359–369
Souframanien J, Gopalakrishna T (2004) A comparative analysis of genetic diversity in blackgram genotypes using RAPD and ISSR markers. Theor Appl Genet 109:1687–1693
Tosti N, Negri V (2002) Efficiency of three PCR-based markers in assessing genetic variation among cowpea (Vigna unguiculata subsp. unguiculata) landraces. Genome 45:268–275
Trabut A (1935) Répertoire des noms indigènes des plantes spontanées cultivées et utilisées dans le nord de l’Afrique. Algiers, Algeria, 355 pp
Van de Peer Y, de Wachter Y (1994) TREECON for windows: a software package for the construction and drawing of evolutionary trees for the microsoft environment. Comput Appl Biosci 10:569–570
Welsh J, Mc Clelland M (1990) Fingerprinting genomes using PCR with arbitrary primers. Nucleic Acids Res 18:7213–7218
Westphal E (1974) Pulses in Ethiopia: their taxonomy and agriculture significance. Agricultural research report 815. Center for Agricultural Publishing and Documentation, Wageningen
Xue-Jun G, Yan Y, Yong-Ming Y, Hong-Wen H, Cheng Y (2005) Genetic diversity and geographic differentiation in endangered Ammopiptanthus (Leguminosae) populations in desert regions of northwest china as revealed by ISSR analysis. Ann Bot 95:843–851
Zietkiewicz E, Rafalski A, Labuda D (1994) Genome fingerprinting by simple sequence repeat (SSR)-anchored polymerase chain reaction amplification. Genomics 20:176–183
Acknowledgments
The authors are grateful to the Algerian Ministry of Higher Education and Scientific Research (Ministère de l’Enseignement Supérieur et de la Recherche Scientifique) for its financial support. We thank the “Laboratory of Molecular Biology of Walloon Agricultural Research Centre of Gembloux” for valuable technical assistance and Dr Bernard China for his critical reading of the manuscript. N. Ghalmi thanks the “Institut Technique des Cultures Maraîchères” of Staouéli for its contribution to the morphological assay.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ghalmi, N., Malice, M., Jacquemin, JM. et al. Morphological and molecular diversity within Algerian cowpea (Vigna unguiculata (L.) Walp.) landraces. Genet Resour Crop Evol 57, 371–386 (2010). https://doi.org/10.1007/s10722-009-9476-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10722-009-9476-5