Abstract
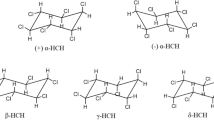
Hexachlorocyclohexane (HCH) contaminated soils were treated for a period of up to 64 days in situ (HCH dumpsite, Lucknow) and ex situ (University of Delhi) in line with three bioremediation approaches. The first approach, biostimulation, involved addition of ammonium phosphate and molasses, while the second approach, bioaugmentation, involved addition of a microbial consortium consisting of a group of HCH-degrading sphingomonads that were isolated from HCH contaminated sites. The third approach involved a combination of biostimulation and bioaugmentation. The efficiency of the consortium was investigated in laboratory scale experiments, in a pot scale study, and in a full-scale field trial. It turned out that the approach of combining biostimulation and bioaugmentation was most effective in achieving reduction in the levels of α- and β-HCH and that the application of a bacterial consortium as compared to the action of a single HCH-degrading bacterial strain was more successful. Although further degradation of β- and δ-tetrachlorocyclohexane-1,4-diol, the terminal metabolites of β- and δ-HCH, respectively, did not occur by the strains comprising the consortium, these metabolites turned out to be less toxic than the parental HCH isomers.






Similar content being viewed by others
References
Bala K, Geueke B, Miska ME, Rentsch D, Poiger T, Dadhwal M, Lal R, Holliger C, Kohler HPE (2012) Enzymatic conversion of ε-hexachlorocyclohexane and a heptachlorocyclohexane isomer, two neglected components of technical hexachlorocyclohexane. Environ Sci Technol 46:4051–4058
Dadhwal M, Singh A, Prakash O, Gupta SK, Kumari K, Sharma P, Jit S, Verma M, Holliger C, Lal R (2009) Proposal of biostimulation for hexachlorocyclohexane (HCH) decontamination and characterization of culturable bacterial community from high dose point HCH contaminated sites. J Appl Microbiol 106:381–392
Dhooge W, Arijs K, D’Haese I, Stuyvaert S, Versonnen B, Janssen C, Verstraete W, Comhaire F (2006) Experimental parameters affecting sensitivity and specificity of a yeast assay for estrogenic compounds: results of an inter laboratory validation exercise. Anal Bioanal Chem 386:1419–1428
Escher BI, Bramaz N, Mueller JF, Quayle P, Rutishauser S (2008) Toxic equivalent concentrations (TEQs) for baseline toxicity and specific modes of action as a tool to improve interpretation of ecotoxicity testing of environmental samples. J Environ Monit 10:612–621
Geueke B, Garg N, Ghosh S, Fleischmann T, Holliger C, Lal R, Kohler H-PE (2013) Metabolomics of hexachlorocyclohexane (HCH)-transformation: ratio of LinA to LinB determines metabolic fate of HCH isomers. Environ Microbiol 15:1040–1049
Girling AE, MarkarianRK Bennett D (1992) Aquatic toxicity testing of oil products—some recommendations. Chemosphere 24:1469–1472
Gupta SK, Lal D, Lata P, Sangwan N, Garg N, Holliger C, Lal R (2013) Changes in the bacterial community and lin genes diversity during biostimulation of indigenous bacterial community of hexachlorocyclohexane (HCH) dumpsite soil. Microbiol 82:234–240
Hammer Ø, Harper DAT, Ryan PD (2001) PAST: paleontological statistics software package for education and data analysis. Palaeontol Electron 4:9
Jit S, Dadhwal M, Kumari H, Jindal S, Kaur J, Lata P, Niharika N, Lal D, Garg N, Gupta SK, Sharma P, Bala K, Singh A, Vijgen J, Weber R, Lal R (2011) Evaluation of hexachlorocyclohexane contamination from the last Lindane production plant operating in India. Environ Sci Pollut Res 18:586–597
Kumari R, Subudhi S, Suar M, Dhingra G, Raina V, Dogra C, Lal S, van der Meer JR, Holliger C, Lal R (2002) Cloning and characterization of lin genes responsible for the degradation of hexachlorocyclohexane isomers by Sphingomonas paucimobilis strain B90. Appl Environ Microbiol 68:6021–6028
Kurihara N (1973) Studies on BHC isomers and related compounds V. some physiochemical properties of BHC isomers (1). Pestic Chem Physiol 2:383–390
Lal R, Pandey G, Sharma P, Kumari K, Malhotra S, Pandey R, Raina V, Kohler HPE, Holliger C, Jackson C, Oakeshott JG (2010) Biochemistry of microbial degradation of hexachlorocyclohexane and prospects for bioremediation. Microbiol Mol Biol Rev 74:58–80
Malhotra S, Sharma P, Kumari H, Singh A, Lal R (2007) Localization of HCH catabolic genes (lin genes) in Sphingobium indicumB90A. Indian J Microbiol 47:271–275
Nagata Y, Kamakura M, Endo R, Miyazaki R, Ohtsubo Y, Tsuda M (2006) Distribution of γ-hexachlorocyclohexane-degrading genes on three replicons in Sphingobium japonicum UT26. FEMS Microbiol Lett 256:112–118
Nagata Y, Endo R, Ito M, Ohtsubo Y, Tsuda M (2007) Aerobic degradation of lindane (γ-hexachlorocyclohexane) in bacteria and its biochemical and molecular basis. Appl Microbiol Biotechnol 76:741–752
Paschke A, Vrana B, Popp P, Schuurmann G (2006) Comparative application of solid-phase micro extraction fibre assemblies and semi-permeable membrane devices as passive air samplers for semi-volatile chlorinated organic compounds. A case study on the landfill “Grube Antonie” in Bitterfeld, Germany. Environ Pollut 144:414–422
Phillips TM, Lee H, Trevors JT, Seech AG (2006) Full-scale in situ bioremediation of hexachlorocyclohexane-contaminated soil. J Chem Technol Biotechnol 81:289–298
Raina V, Hauser A, Buser HR, Rentsch D, Sharma P, Lal R, Holliger C, Poiger JT, Muller MD, Kohler HPE (2007) Hydroxylated metabolites of β- and δ-hexachlorocyclohexane: bacterial formation, stereochemical configuration, and occurrence in groundwater at a former production site. Environ Sci Technol 41:4292–4298
Raina V, Saur M, Singh A, Prakash O, Dadhwal M, Gupta SK, Dogra C, Lawlor K, Lal S, van der Meer JR, Holliger C, Lal R (2008a) Enhanced biodegradation of hexachlorocyclohexane (HCH) in contaminated soils via inoculation with Sphingobium indicum B90A. Biodegradation 19:27–40
Raina V, Rentsch D, Geiger T, Sharma P, Buser HR, Holliger C, Lal R, Kohler HPE (2008b) New metabolites in the degradation of α-and γ-hexachlorocyclohexane (HCH): pentachlorocyclohexenes are hydroxylated to cyclohexenols and cyclohexenediols by the haloalkane dehalogenase LinB from Sphingobium indicum B90A. J Agric Food Chem 56:6594–6603
Routledge EJ, Sumpter JP (1996) Estrogenic activity of surfactants and some of their degradation products assessed using a recombinant yeast screen. Envrion Toxicol Chem 15:241–248
Routledge EJ, Sumpter JP (1997) Structural features of alkyl phenolic chemicals associated with estrogenic activity. J Biol Chem 272:3280–3288
Rubinos DA, Villasuso R, Muniategui S, Barral MT, Diaz Fierros F (2007) Using the landfarming technique to remediate soils contaminated with hexachlorocyclohexane isomers. Water Air Soil Pollut 181:385–399
Sharma P, Raina V, Kumari R, Malhotra S, Dogra C, Kumari H, Kohler HPE, Buser HR, Holliger C, Lal R (2006) Haloalkane dehalogenase LinB is responsible for β- and δ-hexachlorocyclohexane transformation in Sphingobium indicum B90A. Appl Environ Microbiol 72:5720–5727
Sokal RR, Rohlf JF (1995) Biometry: the principles and practices of statistics in biological sciences, 3rd edn. WH Freeman, New York
Suar M, Hauser A, Poiger T, Buser HR, Müller MD, DograC Raina V, Holliger C, van der Meer JR, Lal R, Kohler HPE (2005) Enantioselective transformation of alpha-hexachlorocyclohexane by the dehydrochlorinases LinA1 and LinA2 from the soil bacterium Sphingomonas paucimobilis B90A. Appl Environ Microbiol 71:8514–8518
UNEP (United Nations Environmental Programme) (2006) Report of the persistent organic pollutants review committee on the work of its second meeting. Addendum: Risk profile on lindane. UNEP/POPS/POPRC.2/17/Add.4. 1-25
Vijgen J, Yi LF, Forter M, Lal R, Weber R (2006) The legacy of lindane and technical HCH production. Organohalog Comp 68:899–904
Vijgen J, Abhilash PC, Li YF, Lal R, Forter M, Torres J, Singh N, Yunus M, Tian C, Schaeffer A, Weber R (2011) Hexachlorcyclohexane (HCH) as new Stockholm convention POPs- a global perspective on the management of lindane and its waste isomers. Environ Sci Pollut Res 18:152–162
Willet KL, Ulrich EM, Hites RA (1998) Differential toxicity and environmental fates of hexachlorocyclohexane isomers. Environ Sci Technol 32:2197–2207
Yamamoto S, Otsuka S, Murakami Y, Nishiyama M, Senoo K (2009) Genetic diversity of gamma-hexachlorocyclohexane-degrading sphingomonads isolated from a single experimental field. Lett Appl Microbiol 49:472–477
Acknowledgments
Part of this work was supported by grants from the Indo-Swiss Collaboration in Biotechnology (ISCB) from the Swiss Agency for Development and Cooperation (SDC), Switzerland and the Department of Biotechnology (DBT), Government of India and National Bureau of Agriculturally Important Microorganisms (NBAIM), Government of India. NG, SJ, NS, JK, NN, AD, acknowledge Council of Scientific Research and Industrial Research (CSIR), Govt. of India, AS, VD, NN and PK acknowledge University Grants Commission (UGC), Govt. of India, PL acknowledge Indian Council of Agricultural Research (ICAR), Govt. of India and AKS acknowledge Department of Biotechnology (DBT), Govt. of India for providing the research fellowships. We also thank Dr. Faizan Haider (Lucknow University, India) for his help in field trials. The authors acknowledge Rinku Pandey, Gunjan Pandey, and John Oakeshott (CSIRO, Canberra, Australia) for providing the codon optimized clone of linB. We would also like to thank the Department of Soil Science and Agricultural Chemistry, Indian Agricultural Research Institute (IARI), New Delhi for carrying out physicochemical characterization of the soil.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Garg, N., Lata, P., Jit, S. et al. Laboratory and field scale bioremediation of hexachlorocyclohexane (HCH) contaminated soils by means of bioaugmentation and biostimulation. Biodegradation 27, 179–193 (2016). https://doi.org/10.1007/s10532-016-9765-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10532-016-9765-6