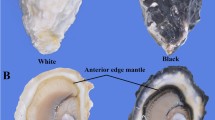
Abstract
The molecular mechanisms of shell formation and pigmentation are issues of great interest in molluscan studies due to the unique physical and biological properties of shells. The Yesso scallop, Patinopecten yessoensis, is one of the most important maricultural bivalves in Asian countries, and its shell color shows polymorphism. To gain more information about the underlying mechanisms of shell formation and pigmentation, this study presents the first analyses of histological and transcriptional differences between different mantle regions of the Yesso scallop, which are thought to be responsible for the formation of different shell layers. The results showed major microstructural differences between the edge and central mantles, which were closely associated with their functions. Different biomineralization-related GO functions, which might participate in the formation of different shell layers, were significantly enriched in the different mantle regions, indicating the different molecular functions of the two mantle regions in shell formation. The melanogenesis pathway, which controls melanin biosynthesis, was the most significantly enriched pathway in the DEGs between the two mantle regions, indicating its important role in shell pigmentation. Tyr, the key and rate-limiting gene in melanogenesis, was expressed at a remarkably high level in the central mantle, while the upstream regulatory genes included in melanogenesis were mainly upregulated in the edge mantle, suggesting the different molecular functions of the two mantle regions in shell pigmentation.






Similar content being viewed by others
References
Addadi L, Joester D, Nudelman F, Weiner S (2006) Mollusk shell formation: a source of new concepts for understanding biomineralization processes. Chem Eur J 12:980–987
Areva S, Peltola T, Säilynoja E, Laajalehto K, Linden M, Rosenholm JB (2002) Effect of albumin and fibrinogen on calcium phosphate formation on sol-gel-derived titania coatings in vitro. Chem Mater 14(4):1614–1621
Audino JA, Marian JEA, Wanninger A, Lopes SG (2015) Mantle margin morphogenesis in Nodipecten nodosus (Mollusca: Bivalvia): new insights into the development and the roles of bivalve pallial folds. BMC Dev Biol 15:22
Bai Z, Zheng H, Lin J, Wang G, Li J (2013) Comparative analysis of the transcriptome in tissues secreting purple and white nacre in the pearl mussel Hyriopsis cumingii. PLoS One 8:e53617
Belcher AM, Wu XH, Christensen RJ, Hansma PK, Stucky GD, Morse DE (1996) Control of crystal phase switching and orientation by soluble mollusc-shell proteins. Nature 381:56–58
Blank S, Arnoldi M, Khoshnavaz S, Treccani L, Kuntz M, Mann K, Grathwohl G, Fritz M (2003) The nacre protein perlucin nucleates growth of calcium carbonate crystals. J Microsc 212(3):280–291
Boettiger A, Ermentrout B, Oster G (2009) The neural origins of shell structure and pattern in aquatic mollusks. Proc Natl Acad Sci 106:6837–6842
Budd A, McDougall C, Green K, Degnan BM (2014) Control of shell pigmentation by secretory tubules in the abalone mantle. Front Zool 11:62
Chang TS (2012) Natural melanogenesis inhibitors acting through the down-regulation of tyrosinase activity. Materials 5:1661–1685
Comfort A (1949a) Acid-soluble pigments of shells. 1. The distribution of porphyrin fluorescence in molluscan shells. Biochem J 44:111–117
Comfort A (1949b) Acid-soluble pigments of molluscan shells. 2. Pigments other than porphyrins. Biochem J 45:199–204
Comfort A (1949c) Acid soluble pigments of molluscan shells. 3. The indigoid character of the blue pigment of Haliotis cracherodii Leach. Biochem J 45:204–208
Comfort A (1951) The pigmentation of molluscan shells. Biol Rev 26:285–301
Ding J, Zhao L, Chang Y, Zhao W, Du Z, Hao Z (2015) Transcriptome sequencing and characterization of Japanese scallop Patinopecten yessoensis from different shell color lines. PLoS One 10:e0116406
Dou J, Li X, Fu Q, Jiao W, Li Y, Li T, Wang Y, Hu X, Wang S, Bao Z (2016) Evaluation of the 2b-RAD method for genomic selection in scallop breeding. Sci Rep 6:19244
Fang D, Xu G, Hu Y, Pan C, Xie L, Zhang R (2011) Identification of genes directly involved in shell formation and their functions in pearl oyster, Pinctada fucata. PLoS One 6:e21860
Feng D, Li Q, Yu H, Zhao X, Kong L (2015) Comparative transcriptome analysis of the Pacific oyster Crassostrea gigas characterized by shell colors: identification of genetic bases potentially involved in pigmentation. PLoS One 10:e0145257
Force A, Lynch M, Pickett FB, Amores A, Yan YL, Postlethwait J (1999) Preservation of duplicate genes by complementary, degenerative mutations. Genetics 151:1531–1545
Fu G, Valiyaveettil S, Wopenka B, Morse DE (2005) CaCO3 biomineralization: acidic 8-kDa proteins isolated from aragonitic abalone shell nacre can specifically modify calcite crystal morphology. Biomacromolecules 6:1289–1298
Hedegaard C, Bardeau J-F, Chateigner D (2006) Molluscan shell pigments: an in situ resonance Raman study. J Molluscan Stud 72:157–162
Hou R, Bao Z, Wang S, Su H, Li Y, Du H, Hu J, Wang S, Hu X (2011) Transcriptome sequencing and de novo analysis for Yesso scallop (Patinopecten yessoensis) using 454 GS FLX. PLoS One 6:e21560
Huang J, Zhang C, Ma Z, Xie L, Zhang R (2007) A novel extracellular EF-hand protein involved in the shell formation of pearl oyster. Biochim Biophys Acta Gen Subj 1770:1037–1044
Joubert C, Piquemal D, Marie B, Manchon L, Pierrat F, Zanella-Cléon I, Cochennec-Laureau N, Montagnani C (2010) Transcriptome and proteome analysis of Pinctada margaritifera calcifying mantle and shell: focus on biomineralization. BMC Genomics 11:613
Kinoshita S, Wang N, Inoue H, Maeyama K, Okamoto K, Nagai K, Kondo H, Hirono I, Asakawa S, Watabe S (2011) Deep sequencing of ESTs from nacreous and prismatic layer producing tissues and a screen for novel shell formation-related genes in the pearl oyster. PLoS One 6:e21238
Kröger N (2009) The molecular basis of nacre formation. Science 325:1351–1352
Langmead B, Salzberg SL (2012) Fast gapped-read alignment with Bowtie 2. Nat Methods 9:357–359
Larkin MA, Blackshields G, Brown NP, Chenna R, McGettigan PA, McWilliam H, Valentin F, Wallace IM, Wilm A, Lopez R, Thompson JD, Gibson TJ, Higgins DG (2007) Clustal W and Clustal X version 2.0. Bioinformatics 23:2947–2948
Lemer S, Saulnier D, Gueguen Y, Planes S (2015) Identification of genes associated with shell color in the black-lipped pearl oyster, Pinctada margaritifera. BMC Genomics 16:568
Li Y, Zhang L, Li Y, Li W, Guo Z, Li R, Hu X, Bao Z, Wang S (2019) Dynamics of DNA methylation and DNMT expression during gametogenesis and early development of scallop Patinopecten yessoensis. Mar Biotechnol 21:196–205
Liu H, Liu S, Ge Y, Liu J, Wang X, Xie L, Zhang R, Wang Z (2007) Identification and characterization of a biomineralization related gene PFMG1 highly expressed in the mantle of Pinctada fucata. Biochemistry 46:844–851
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2− ΔΔCT method. Methods 25:402–408
Luo YJ, Takeuchi T, Koyanagi R, Yamada L, Kanda M, Khalturina M, Fujie M, S-i Y, Endo K, Satoh N (2015) The Lingula genome provides insights into brachiopod evolution and the origin of phosphate biomineralization. Nat Commun 6:8301
Lydie MAO, Golubic S, Le Campion-Alsumard T, Payri C (2001) Developmental aspects of biomineralisation in the Polynesian pearl oyster Pinctada margaritifera var. cumingii. Oceanol Acta 24:37–49
Mao J, Zhang W, Zhang X, Tian Y, Wang X, Hao Z, Chang Y (2018) Transcriptional changes in the Japanese scallop (Mizuhopecten yessoensis) shellinfested by Polydora provide insights into the molecular mechanism of shell formation and immunomodulation. Sci Rep 8:17664
Mao J, Zhang X, Zhang W, Tian Y, Wang X, Hao Z, Chang Y (2019) Genome-wide identification, characterization and expression analysis of the MITF gene in Yesso scallops (Patinopecten yessoensis) with different shell colors. Gene 688:155–162
Miyamoto H, Hamaguchi M, Okoshi K (2002) Analysis of genes expressed in the mantle of oyster Crassostrea gigas. Fish Sci 68:651–658
Naka K, Chujo Y (2001) Control of crystal nucleation and growth of calcium carbonate by synthetic substrates. Chem Mater 13:3245–3259
Pang Y, Ding J, Tian Y, Hao Z, Chang Y (2015) Analysis of shell microstructure and five surface elements of Patinopecten yessoensis at different ages. Mar Sci 39:28–34
Reindl S, Haszprunar G (1996) Fine structure of caeca and mantle of arcoid and limopsoid bivalves (Mollusca: Pteriomorpha). Veliger 39:101–116
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4(4):406–425
Samata T, Hayashi N, Kono M, Hasegawa K, Horita C, Akera S (1999) A new matrix protein family related to the nacreous layer formation of Pinctada fucata. FEBS Lett 462:225–229
Sudo S, Fujikawa T, Nagakura T, Ohkubo T, Sakaguchi K, Tanaka M, Nakashima K, Takahashi T (1997) Structures of mollusc shell framework proteins. Nature 387:563–564
Sun X, Yang A, Wu B, Zhou L, Liu Z (2015) Characterization of the mantle transcriptome of Yesso scallop (Patinopecten yessoensis): identification of genes potentially involved in biomineralization and pigmentation. PLoS One 10:e0122967
Sun X, Liu Z, Zhou L, Wu B, Dong Y, Yang A (2016) Integration of next generation sequencing and EPR analysis to uncover molecular mechanism underlying shell color variation in scallops. PLoS One 11:e0161876
Sun X, Wu B, Zhou L, Liu Z, Dong Y, Yang A (2017) Isolation and characterization of melanin pigment from Yesso scallop Patinopecten yessoensis. J Ocean Univ China 16:279–284
Suzuki M, Murayama E, Inoue H, Ozaki N, Tohse H, Kogure T, Nagasawa H (2004) Characterization of Prismalin-14, a novel matrix protein from the prismatic layer of the Japanese pearl oyster (Pinctada fucata). Biochem J 382:205–213
Suzuki M, Iwashima A, Kimura M, Kogure T, Nagasawa H (2013) The molecular evolution of the Pif family proteins in various species of mollusks. Mar Biotechnol 15:145–158
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28:2731–2739
Trapnell C, Williams BA, Pertea G, Mortazavi A, Kwan G, Van Baren MJ, Salzberg S, Wold B, Pachter L (2010) Transcript assembly and quantification by RNA-Seq reveals unannotated transcripts and isoform switching during cell differentiation. Nat Biotechnol 28:511–515
Tsukamoto D, Sarashina I, Endo K (2004) Structure and expression of an unusually acidic matrix protein of pearl oyster shells. Biochem Biophys Res Commun 320:1175–1180
Vance KW, Goding CR (2004) The transcription network regulating melanocyte development and melanoma. Pigment Cell Res 17:318–325
Wang S, Hou R, Bao Z, Du H, He Y, Su H, Zhang Y, Fu X, Jiao W, Li Y, Zhang L, Wang S, Zhang L (2013) Transcriptome sequencing of Zhikong scallop (Chlamys farreri) and comparative transcriptomic analysis with Yesso scallop (Patinopecten yessoensis). PLoS One 8:e63927
Wang S, Lv J, Zhang L, Dou J, Sun Y, Li X, Fu X, Dou H, Mao J, Hu X, Bao Z (2015) MethylRAD: a simple and scalable method for genome-wide DNA methylation profiling using methylation-dependent restriction enzymes. Open Biol 5:150130
Wang S, Zhang J, Jiao W, Li J, Xun X, Sun Y, Guo X, Huan P, Dong B, Zhang L, Hu X, Sun X, Wang J, Zhao C, Wang Y, Wang D, Huang X, Wang R, Lv J, Li Y, Zhang Z, Liu B, Lu W, Hui Y, Liang J, Zhou Z, Hou R, Li X, Liu Y, Li H, Ning X, Lin Y, Zhao L, Xing Q, Dou J, Li Y, Mao J, Guo H, Dou H, Li T, Mu C, Jiang W, Fu Q, Fu X, Miao Y, Liu J, Yu Q, Li R, Liao H, Li X, Kong Y, Jiang Z, Chourrout D, Li R, Bao Z (2017) Scallop genome provides insights into evolution of bilaterian karyotype and development. Nat Ecol Evol 1:0120
Weiss IM, Kaufmann S, Mann K, Fritz M (2000) Purification and characterization of perlucin and perlustrin, two new proteins from the shell of the mollusc Haliotis laevigata. Biochem Biophys Res Commun 267:17–21
Wheeler AP, Sikes CS (1984) Regulation of carbonate calcification by organic matrix. Am Zool 24:933–944
Wilbur KM, Saleuddin ASM (1983) Shell formation. In: Saleuddin ASM, Wilbur KM (eds) The mollusca, vol 4. Academic Press, New York, pp 235–287
Williams ST (2016) Molluscan shell colour. Biol Rev 92(2):1039–1058
Yue X, Nie Q, Xiao G, Liu B (2015) Transcriptome analysis of shell color-related genes in the clam Meretrix meretrix. Mar Biotechnol 17:364–374
Zhang C, Zhang R (2006) Matrix proteins in the outer shells of molluscs. Mar Biotechnol 8:572–586
Zhang M, Wang Y, Li Y, Li W, Li R, Xie X, Wang S, Hu X, Zhang L, Bao Z (2018) Identification and characterization of neuropeptides by transcriptome and proteome analyses in a bivalve mollusc Patinopecten yessoensis. Front Genet 9:197
Zhao X, Wang Q, Jiao Y, Huang R, Deng Y, Wang H, Du X (2012) Identification of genes potentially related to biomineralization and immunity by transcriptome analysis of pearl sac in pearl oyster Pinctada martensii. Mar Biotechnol 14:730–739
Zhao L, Li Y, Li Y, Yu J, Liao H, Wang S, Lv J, Liang J, Huang X, Bao Z (2017) A genome-wide association study identifies the genomic region associated with shell color in yesso scallop, Patinopecten yessoensis. Mar Biotechnol 19:301–309
Acknowledgments
We thank Dalian Zhangzidao Fishery Group Co., Ltd. (Dalian, China) for providing the scallop materials.
Funding
This project was supported by the National Natural Science Foundation of China (31702342), the Major Science and Technology Research Project of Liaoning Province (2017203003), and the Doctoral Startup Foundation of Liaoning Province (20170520095).
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: JM and YC. Prepared the samples: XW, JS, and DY. Performed the experiments: JM, WZ, and YT. Analyzed the data: JM, WZ, ZH, and BH. Wrote the paper: JM and YC.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
ESM 1
(DOCX 80 kb)
Rights and permissions
About this article
Cite this article
Mao, J., Zhang, W., Wang, X. et al. Histological and Expression Differences Among Different Mantle Regions of the Yesso Scallop (Patinopecten yessoensis) Provide Insights into the Molecular Mechanisms of Biomineralization and Pigmentation. Mar Biotechnol 21, 683–696 (2019). https://doi.org/10.1007/s10126-019-09913-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10126-019-09913-x