Abstract
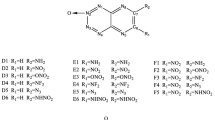
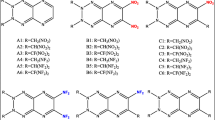
The heats of formation (HOFs), electronic structure, energetic properties, and thermal stabilities for a series of 1,4-bis(1-azo-2,4-dinitrobenzene)-iminotetrazole derivatives with different substituents and substitution positions and numbers of nitrogen atoms in the nitrobenzene rings were studied using the DFT-B3LYP method. All the substituted compounds have higher HOFs than their parent compounds. As the number of nitrogen atoms in the nitrobenzene ring increases, the HOFs of the derivatives with the same substituent rise gradually. Replacing carbon atoms in the nitrobenzene with nitrogen atoms to form N–N bonds is very helpful in improving their HOFs. Most of the substituted compounds have higher HOMO–LUMO gaps than the corresponding unsubstituted compounds. Substitution of the –NO2, –NF2, or –ONO2 group and an increase in the number of nitrogen atoms in the nitrobenzene rings are useful for enhancing their detonation performance. The substituents’ substitution is not favorable for improving thermal stability. Considering detonation performance and thermal stability, five compounds may be considered potential candidates for high energy density compounds (HEDCs).




Similar content being viewed by others

References
Fried LE, Manaa MR, Pagoria PF, Simpson RL (2001) Annu Rev Mater Res 31:291–321
Pagoria PF, Lee GS, Mitchell AR, Schmidt (2002) Thermochim Acta 384:187–204
Xu XJ, Xiao HM, Ju XH, Gong XD, Zhu WH (2006) J Phys Chem A 110:5929–5933
Wei T, Zhu WH, Zhang XW, Li YF, Xiao HM (2009) J Phys Chem A 113:9404–9412
Gutowski KE, Rogers RD, Dixon DA (2006) J Phys Chem A 110:11890–11897
Wei T, Zhu WH, Zhang JJ, Xiao HM (2010) J Hazard Mater 179:581–590
Zhang CC, Zhu WH, Xiao HM (2010) Comput Theor Chem 967:257–264
Pan Y, Zhu WH, Xiao HM (2012) J Mol Model 18:3125–3138
Joo YH, Shreeve JM (2010) Angew Chem Int Ed 49:7320–7323
Qiu L, Xiao HM, Gong XD, Ju XH, Zhu WH (2006) J Phys Chem A 110:3797–3807
Wei T, Wu JZ, Zhang CC, Zhu WH, Xiao HM (2012) J Mol Model 18:3467–3479
Joo YH, Shreeve JM (2009) Angew Chem Int Ed 48:564–567
Stierstorfer J, Tarantik KR, Klapöke TM (2009) Chem Eur J 15:5775–5792
Zhao GZ, Lu M (2012) J Mol Model 18:2443–2451
Agrawal JP (2005) Propell Explos Pyrot 5:316–328
Nair UR, Gore GM, Sivabalan R, Pawar SJ, Asthana SN, Venugopalan S (2007) J Hazard Mater 143:500–505
Zhang XW, Zhu WH, Xiao HM (2010) J Phys Chem A 114:603–612
Zhu WH, Zhang CC, Wei T, Xiao HM (2011) J Comput Chem 32:2298–2312
Zhang XW, Zhu WH, Xiao HM (2010) Int J Quantum Chem 110:1549–1558
Ghule VD, Radhakrishnan S, Jadhav PM, Pandey RK (2011) J Mol Model 17:2927–2937
Thottempudi V, Gao HX, Shreeve JM (2011) J Am Chem Soc 133:6464–6471
Zhou Y, Long XP, Shu YJ (2010) J Mol Model 16:1021–1027
Zhu WH, Zhang CC, Wei T, Xiao HM (2011) Struct Chem 22:149–159
Zhu WX, Wong NB, Wang WZ, Zhou G, Tian A (2004) J Phys Chem A 108:97–106
Pan Y, Li JS, Cheng BB, Zhu WH, Xiao HM (2012) Comput Theor Chem 992:110–119
Ravi P, Gore GM, Tewari SP, Sikder AK (2012) Propell Explos Pyrot 37:52–58
Fan XW, Ju XH, Xiao HM, Qiu L (2006) J Mol Struct (THEOCHEM) 801:55–62
Fan XW, Ju XH (2008) J Comput Chem 29:505–513
Muthurajan H, Sivabalan R, Talawar MB, Anniyappan M, Venugopalan S (2006) J Hazard Mater 133:30–45
Chen ZX, Xiao JM, Xiao HM, Chiu YN (1999) J Phys Chem A 103:8062–8066
Xiao HM, Chen ZX (2000) The modern theory for tetrazole chemistry, 1st edn. Science, Beijing
Hahre WJ, Radom L, Schleyer PVR, Pole JA (1986) Ab initio molecular orbital theory. Wiley-Interscience, New York
Atkins PW (1982) Physical chemistry. Oxford University Press, Oxford
Politzer P, Lane P, Murray JS (2011) Cent Eur J Energ Mat 8:39–52
Politzer P, Murray JS (2011) Cent Eur J Energ Mat 8:209–220
Politzer P, Murray JS, Grice ME, DeSalvo M, Miller E (1997) Mol Phys 91:923–928
Byrd EFC, Rice BM (2006) J Phys Chem A 110:1005–1013
Bulat FA, Toro-Labbe A, Brinck T, Murray JS, Politzer P (2010) J Mol Model 16:1679–1691
Jaidann M, Roy S, Abou-Rachid H, Lussier LS (2010) J Hazard Mater 176:165–173
Kamlet MJ, Jacobs SJ (1968) J Chem Phys 48:23–35
Politzer P, Martinez J, Murray JS, Concha MC, Toro-Labbé A (2009) Mol Phys 107:2095–2101
Benson SW (1976) Thermochemical kinetic, 2nd edn. Wiley-Interscience, New York
Mills I, Cvitas T, Homann K, Kallay N, Kuchitsu K (1988) Quantities, units, and symbols in physical chemistry. Blackwell, Oxford
Blanksby SJ, Ellison GB (2003) Accounts Chem Res 36:255–263
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Zakrzewski VG, Montgomery JA, Stratmann RE, Burant JC, Dapprich S, Millam JM, Daniels AD, Kudin KN, Strain MC, Farkas O, Tomasi J, Barone V, Cossi M, Cammi R, Mennucci B, Pomelli C, Adamo C, Clifford S, Ochterski J, Petersson GA, Ayala PY, Cui Q, Morokuma K, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Cioslowski J, Ortiz JV, Baboul AG, Stefanov BB, Liu G, Liashenko, A, Piskorz P, Komaromi I, Gomperts R, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Gonzalez C, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Andres JL, Gonzalez C, Head-Gordon M, Replogle ES, Pople JA (2009) Gaussian 09, Revision A. 01. Gaussian, Inc.
Afeefy HY, Liebman JF, Stein SE (2000) Neutral thermochemical data. In: Linstrom PJ, Mallard WG (eds) NIST chemistry webbook, NIST standerd reference database number 69. National Institute of Standards and Technology, Gaitherersburg, MD, http://webbook.nist.gov
Dean JA (1999) LANGE’S handbook of chemistry, 15th edn. McGraw-Hill, New York
Scott AP, Radom L (1996) J Phys Chem 100:16502–16513
Talawar MB, Sivabalan R, Mukundan T, Muthurajan H, Sikder AK, Gandhe BR, Rao AS (2009) J Hazard Mater 161:589–607
Chung GS, Schmidt MW, Gordon MS (2000) J Phys Chem A 104:5647–5650
Acknowledgment
This work was supported by the National Natural Science Foundation of China (Grant No. 21273115).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, Q., Pan, Y., Zhu, W. et al. Computational study of energetic nitrogen-rich derivatives of 1,4-bis(1-azo-2,4-dinitrobenzene)-iminotetrazole. J Mol Model 19, 1853–1864 (2013). https://doi.org/10.1007/s00894-013-1756-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-013-1756-5