Abstract
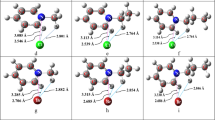
O-H…X and O-H…O H-bonds as well as C-X…X dihalogen and C-X…O halogen bonds have been investigated in halomethanol dimers (bromomethanol dimer, iodomethanol dimer, difluorobromomethanol…bromomethanol complex and difluoroiodomethanol…iodomethanol complex). Structures of all complexes were optimized at the counterpoise-corrected MP2/cc-pVTZ level and single-point energies were calculated at the CCSD(T)/aug-cc-pVTZ level. Energy decomposition for the bromomethanol dimer complex was performed using the DFT-SAPT method based on the aug-cc-pVTZ basis set. OH…O and OH…X H-bonds are systematically the strongest in all complexes investigated, with the former being the strongest bond. Halogen and dihalogen bonds, being of comparable strength, are weaker than both H-bonds but are still significant. The strongest bonds were found in the difluoroiodomethanol…iodomethanol complex, where the O-H…O H-bond exceeds 7 kcal mol-1, and the halogen and dihalogen bonds exceed 2.5 and 2.3 kcal mol-1, respectively. Electrostatic energy is dominant for H-bonded structures, in halogen bonded structures electrostatic and dispersion energies are comparable, and, finally, for dihalogen structures the dispersion energy is clearly dominant.

Competition of hydrogen, halogen, and dihalogen bonding in the bromomethanol dimer are investigated


Similar content being viewed by others
References
Metrangolo, P, Resnati G (eds.) (2008) Halogen bonding with dihalogens and interhalogens, in halogen bonding. In: Fundamentals and applications. Springer, Berlin, p 126
Politzer P, Lane P, Concha MC, Ma Y, Murray JSJ (2007) Mol Model 13:305–311
Brinck T, Murray JS, Politzer P (1992) Int J Quant Chem Quant Biol Symp 19:57–64
Politzer P, Murray JS, Concha MCJ (2007) Mol Model 13:643–650
Auffinger P, Hays FA, Westhof E, Ho PS (2004) Proc Natl Acad Sci U S A 101:16789–16794
Awwadi F, Willett RD, Twamley B (2011) Cryst Growth Des 11:5316–5323
Politzer P, Murray JS, Concha MCJ (2008) Mol Model 14:659–665
Rezac J, Riley KE, Hobza PJ (2011) Chem Theory Comput 7:2427–2438
Riley KE, Murray JS, Politzer P, Concha MC, Hobza PJ (2009) Chem Theory Comput 5:155–163
Riley KE (2009) Int J Quant Chem 110:1833–1841
Riley KE, Pitonak M, Jurecka P, Hobza P (2010) Chem Rev 110:5023–5063
Jeziorski B, Moszynski R, Szalewicz K (1994) Chem Rev 94:1887–1930
Jansen G, Hesselmann AJ (2001) Phys Chem A 105:11156–11157
Dabkowska I, Jurecka P, Hobza PJ (2005) Chem Phys 122:204322
Řezáč J, Hobza PJ (2011) Chem Theory Comput 7:685–689
Bader RFW, Carroll MT, Cheeseman JR, Chang CJ (1987) Am Chem Soc 109:7968–7979
Bulat FA, Toro-Labbe A, Brinck T, Murray JS, Politzer PJ (2010) Mol Model 16:1679–1691
Werner HJ, Knowles PJ, Lindh R, Manby FR, Sch€utz M, Celani P, Korona T, Rauhut G, Amos RD, Bernhardsson A, Berning A, Cooper DL, Deegan MJO, Dobbyn AJ, Eckert F, Hampel C, Hetzer, G, Lloyd AW, McNicholas SJ, Meyer W, Mura ME, Nicklaß A, Palmieri P, Pitzer P, Schumann U, Stoll H, Stone AJ, Tarroni R, Thorsteinsson T (2008) MOLPRO, version 2010.1
Riley KE, Murray JS, Fanfrlik J, Rezac J, Sola RJ, Concha MC, Ramos FM, Politzer PJ (2012) Mol Model doi:10.1007/s00894-012-1428-x
Acknowledgments
This work was part of the Research Project RVO: 61388963 of the Institute of Organic Chemistry and Biochemistry, Academy of Sciences of the Czech Republic. The support of Praemium Academiae, Academy of Sciences of the Czech Republic, awarded to P.H. in 2007 is also acknowledged. This work was also supported by the Czech Science Foundation [P208/12/G016] and the operational program Research and Development for Innovations of European Social Fund (CZ 1.05/2.1.00/03/0058).
Author information
Authors and Affiliations
Corresponding author
Additional information
Dedicated to the 75th birthday of our distinguished colleague and friend, Peter Politzer
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 2328 kb)
Rights and permissions
About this article
Cite this article
Riley, K.E., Řezáč, J. & Hobza, P. Competition between halogen, dihalogen and hydrogen bonds in bromo- and iodomethanol dimers. J Mol Model 19, 2879–2883 (2013). https://doi.org/10.1007/s00894-012-1727-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00894-012-1727-2