Abstract
Main conclusion
Successful application of the photosynthesis-to-fuels approach requires a high product-to-biomass carbon-partitioning ratio. The work points to the limiting amounts of heterologous terpene synthase in cyanobacteria as a potential barrier in the yield of terpene hydrocarbons via photosynthesis.
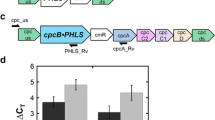
Cyanobacteria like Synechocystis sp. can be exploited as platforms in a photosynthesis-to-fuels process for the generation of terpene hydrocarbons. Successful application of this concept requires maximizing photosynthesis and attaining a high endogenous carbon partitioning toward the desirable product. The work addressed the question of the regulation of β-phellandrene synthase transgene expression in relation to product yield from the terpenoid biosynthetic pathway of cyanobacteria. The choice of strong alternative transcriptional and translational cis-regulatory elements and the choice of the Synechocystis genomic DNA loci for transgene insertion were investigated. Specifically, the β-phellandrene synthase transgene was expressed under the control of the endogenous psbA2 promoter, or under the control of the Ptrc promoter from Escherichia coli with the translation initiation region of highly expressed gene 10 from bacteriophage T7. These heterologous elements allowed for constitutive transgene expression. In addition, the β-phellandrene synthase construct was directed to replace the Synechocystis cpc operon, encoding the peripheral phycocyanin rods of the phycobilisome antenna. Results showed that a 4-fold increase in the cellular content of the β-phellandrene synthase was accompanied by a 22-fold increase in β-phellandrene yield, suggesting limitations in rate and yield by the amount of the transgenic enzyme. The work points to the limiting amount of transgenic terpene synthases as a potential barrier in the heterologous generation of terpene products via the process of photosynthesis.









Similar content being viewed by others
Abbreviations
- APC:
-
Allophycocyanin
- Chl:
-
Chlorophyll
- cmR:
-
Chloramphenicol resistance
- cpc:
-
Operon encoding the phycocyanin peripheral rods
- dcw:
-
Dry cell weight
- DB:
-
Downstream box
- GC–MS:
-
Gas chromatography–mass spectrometry
- OD:
-
Optical density
- PC:
-
Phycocyanin
- PHL:
-
β-Phellandrene
- PHLS:
-
β-Phellandrene synthase
- psbA2:
-
Gene encoding the D1/32 kD reaction center protein of PSII
- Ptrc:
-
Hybrid promoter made with consensus sequences from E. coli Ptrp and Plac
- RBS:
-
Ribosome-binding site
- T7g10:
-
Gene 10 from bacteriophage T7
References
Adir N (2005) Elucidation of the molecular structures of components of the phycobilisome: reconstructing a giant. Photosynth Res 85:15–32
Amann E, Ochs B, Abel KJ (1988) Tightly regulated tac promoter vectors useful for the expression of unfused and fused protein in Escherichia coli. Gene 69:301–315
Angermayr SA, Paszota M, Hellingwerf KJ (2012) Engineering a cyanobacterial cell factory for production of lactic acid. Appl Environ Microb 78:7098–7106
Atsumi S, Higashide W, Liao JC (2009) Direct photosynthetic recycling of carbon dioxide to isobutyraldehyde. Nat Biotechnol 27:1177–1180
Bailey S, Grossman A (2008) Photoprotection in cyanobacteria: regulation of light harvesting. Photochem Photobiol 84:1410–1420
Bentley FK, Melis A (2012) Diffusion-based process for carbon dioxide uptake and isoprene emission in gaseous/aqueous two-phase photobioreactors by photosynthetic microorganisms. Biotechnol Bioeng 109:100–109
Bentley FK, García-Cerdán JG, Chen HC, Melis A (2013) Paradigm of monoterpene (β-phellandrene) hydrocarbons production via photosynthesis in cyanobacteria. BioEnergy Res 6:917–929
Bohlmann J, Keeling CI (2008) Terpenoid biomaterials. Plant J 54:656–669
Booker H, Evans LK, Gillam AE (1940) The effect of molecular environment on the absorption spectra of organic compounds in solution. Part I. Conjugated dienes. J Chem Soc 1453–1463. doi:10.1039/JR9400001453
Demissie ZA, Sarker LS, Mahmoud SS (2011) Cloning and functional characterization of β-phellandrene synthase from Lavandula angustifolia. Planta 233:685–696
Deng MD, Coleman JR (1999) Ethanol synthesis by genetic engineering in cyanobacteria. Appl Environ Microbiol 65:523–528
Dexter J, Fu P (2009) Metabolic engineering of cyanobacteria for ethanol production. Energy Environ Sci 2:857
Ducat DC, Avelar-Rivas JA, Way JC, Silver PA (2012) Rerouting carbon flux to enhance photosynthetic productivity. Appl Environ Microbiol 78:2660–2668
Eaton-Rye JJ (2011) Construction of gene interruptions and gene deletions in the cyanobacterium Synechocystis sp. strain PCC 6803. Methods Mol Biol 684:295–312
Formighieri C, Franck F, Bassi R (2012) Regulation of the pigment optical density of an algal cell: filling the gap between photosynthetic productivity in the laboratory and in mass culture. J Biotechnol 162:115–123
Gao Z, Zhao H, Li Z, Tan X, Lu X (2012) Photosynthetic production of ethanol from carbon dioxide in genetically engineered cyanobacteria. Energy Environ Sci 5:9857
Gershenzon J, Dudareva N (2007) The function of terpene natural products in the natural world. Nat Chem Biol 3:408–414
Glazer AN, Melis A (1987) Photochemical reaction centers: structure, organization and function. Annu Rev Plant Physiol 38:11–45
Gross KP, Schnepp O (1978) Absorption and circular dichroism spectra of the cis- and trans- butadiene chromophores α- and β-phellandrene. J Chem Phys 68:2647–2657
Grossman AR, Schaefer MR, Chiang GG, Collier JL (1993) The phycobilisome, a light-harvesting complex responsive to environmental conditions. Microbiol Rev 57:725–749
Guerrero F, Carbonell V, Cossu M, Correddu D, Jones PR (2012) Ethylene synthesis and regulated expression of recombinant protein in Synechocystis sp. PCC 6803. PLoS One 7:e50470
Hellingwerf KJ, Teixeira de Mattos MJ (2009) Alternative routes to biofuels: light-driven biofuel formation from CO2 and water based on the “photanol” approach. J Biotechnol 142:87–90
Horie Y, Ito Y, Ono M, Moriwaki N, Kato H, Hamakubo Y, Amano T, Wachi M, Shirai M, Asayama M (2007) Dark-induced mRNA instability involves RNase E/G-type endoribonuclease cleavage at the AU-box and SD sequences in cyanobacteria. Mol Genet Genomics 278:331–346
Huang HH, Camsund D, Lindblad P, Heidorn T (2010) Design and characterization of molecular tools for a synthetic biology approach towards developing cyanobacterial biotechnology. Nucleic Acids Res 38:2577–2593
Imashimizu M, Fujiwara S, Tanigawa R, Tanaka K, Hirokawa T, Nakajima Y, Higo J (2003) Thymine at -5 is crucial for cpc promoter activity of Synechocystis sp. strain PCC 6714. J Bacteriol 185:6477–6480
Jansson C, Salih G, Eriksson J, Wiklund R, Ghebramedhin H (1998) Use of Synechocystis 6803 to study expression of a psbA gene family. Methods Enzymol 297:166–182
Kirilovsky D, Kerfeld CA (2012) The orange carotenoid protein in photoprotection of photosystem II in cyanobacteria. Biochim Biophys Acta 1817:158–166
Kuroda H, Maliga P (2001a) Complementarity of the 16S rRNA penultimate stem with sequences downstream of the AUG destabilizes the plastid mRNAs. Nucleic Acids Res 29:970–975
Kuroda H, Maliga P (2001b) Sequences downstream of the translation initiation codon are important determinants of translation efficiency in chloroplasts. Plant Physiol 125:430–436
Lan EI, Liao JC (2012) ATP drives direct photosynthetic production of 1-butanol in cyanobacteria. Proc Natl Acad Sci USA 109:6018–6023
Li H, Liao JC (2013) Engineering a cyanobacterium as the catalyst for the photosynthetic conversion of CO2 to 1,2-propanediol. Microb Cell Fact 12:4
Lichtenthaler HK (2000) Sterols and isoprenoids. Biochem Soc Trans 28:785–789
Lindberg P, Park S, Melis A (2010) Engineering a platform for photosynthetic isoprene production in cyanobacteria, using Synechocystis as the model organism. Metab Eng 12:70–79
Liu X, Sheng J, Curtiss R (2011) Fatty acid production in genetically modified cyanobacteria. Proc Natl Acad Sci USA 108:6899–6904
MacBeth AK, Smith GE, West TF (1938) β-Phellandrene. J Chem Soc 119–123. doi:10.1039/JR9380000119
MacColl R (1998) Cyanobacterial phycobilisomes. J Struct Biol 124:311–334
McNevin D, von Caemmerer S, Graham Farquhar G (2006) Determining RuBisCO activation kinetics and other rate and equilibrium constants by simultaneous multiple non-linear regression of a kinetic model. J Exp Bot 57:3883–3900
Meeks JK, Castenholz RW (1971) Growth and photosynthesis in an extreme thermophile, Synechococcus lividus (Cyanophyta). Arch Microbiol 78:25–41
Melis A (1999) Photosystem-II damage and repair cycle in chloroplasts: what modulates the rate of photodamage in vivo?. Trends Plant Sci 4:130–135
Melis A (2009) Solar energy conversion efficiencies in photosynthesis: minimizing the chlorophyll antennae to maximize efficiency. Plant Sci 177:272–280
Melis A (2012) Photosynthesis-to-fuels: from sunlight to hydrogen, isoprene, and botryococcene production. Energy Environ Sci 5:5531–5539
Melis A (2013) Carbon partitioning in photosynthesis. Curr Opin Cheml Biol 17:453–456
Melis A, Neidhardt J, Benemann JR (1999) Dunaliella salina (Chlorophyta) with small chlorophyll antenna sizes exhibit higher photosynthetic productivities and photon use efficiencies than normally pigmented cells. J Appl Phycol 10:515–525
Mohamed A, Eriksson J, Osiewacz HD, Jansson C (1993) Differential expression of the psbA genes in the cyanobacterium Synechocystis 6803. Mol Gen Genet 238:161–168
Mulo P, Sakurai I, Aro EM (2012) Strategies for psbA gene expression in cyanobacteria, green algae and higher plants: from transcription to PSII repair. Biochim Biophys Acta 1817:247–257
Münch R, Hiller K, Grote A, Scheer M, Klein J, Schobert M, Jahn D (2005) Virtual footprint and PRODORIC: an integrative framework for regulon prediction in prokaryotes. Bioinformatics 21:4187–4189
Nakajima Y, Ueda R (1997) Improvement of photosynthesis in dense microalgal suspension by reduction of light harvesting pigments. J Appl Phycol 9:503–510
Nakajima Y, Ueda R (1999) Improvement of microalgal photosynthetic productivity by reducing the content of light harvesting pigment. J Appl Phycol 11:195–201
Nakajima Y, Fujiwara S, Sawai H, Imashimizu M, Tsuzuki M (2001) A phycocyanin-deficient mutant of Synechocystis PCC 6714 with a single-base substitution upstream of the cpc operon. Plant Cell Physiol 42:992–998
Nixon P, Jansson C (1996) Cyanobacterial transformation and gene regulation. In: Andersson B, Salter AH, Barber J (eds) Molecular genetics of photosynthesis. Oxford University Press, Oxford, UK, pp 197–224
Oliver JWK, Machado IMP, Yoneda H, Atsumi S (2013) Cyanobacterial conversion of carbon dioxide to 2,3-butanediol. Proc Natl Acad Sci USA 110:1249–1254
Peralta-Yahya PP, Ouellet M, Chan R, Mukhopadhyay A, Keasling JD, Lee TS (2011) Identification and microbial production of a terpene-based advanced biofuel. Nat Commun 2:483
Rakhimberdieva MG, Elanskaya IV, Vermaas WF, Karapetyan NV (2010) Carotenoid-triggered energy dissipation in phycobilisomes of Synechocystis sp. PCC 6803 diverts excitation away from reaction centers of both photosystems. Biochim Biophys Acta 1797:241–249
Reinsvold RE, Jinkerson RE, Radakovits R, Posewitz MC, Basu C (2011) The production of the sesquiterpene β-caryophyllene in a transgenic strain of the cyanobacterium Synechocystis. J Plant Physiol 168:848–852
Sakurai I, Stazic D, Eisenhut M, Vuorio E, Steglich C, Hess WR, Aro EM (2012) Positive regulation of psbA gene expression by cis-encoded antisense RNAs in Synechocystis sp. PCC 6803. Plant Physiol 160:1000–1010
Sasaki K, Ohara K, Yazaki K (2005) Gene expression and characterization of isoprene synthase from Populus alba. FEBS Lett 579:2514–2518
Sprengart ML, Fuchs E, Porter AG (1996) The downstream box: an efficient and independent translation initiation signal in Escherichia coli. EMBO J 15:665–674
Takahama K, Matsuoka M, Nagahama K, Ogawa T (2003) Construction and analysis of a recombinant cyanobacterium expressing a chromosomally inserted gene for an ethylene-forming enzyme at the psbAI locus. J Biosci Bioeng 95:302–305
Tan X, Yao L, Gao Q, Wang W, Qi F, Lu X (2011) Photosynthesis driven conversion of carbon dioxide to fatty alcohols and hydrocarbons in cyanobacteria. Metab Eng 13:169–176
Tian L, van Stokkum IHM, Koehorst RBM, van Amerongen H (2012) Light harvesting and blue-green light induced non-photochemical quenching in two different c-phycocyanin mutants of Synechocystis PCC 6803. J Phys Chem 117:11000–11006
Van Wagoner RM, Drummond AK, Wright JLC (2007) Biogenetic diversity of cyanobacterial metabolites. Adv Appl Microbiol 61:89–217
Varman AM, Xiao Y, Pakrasi HB, Tang YJ (2013) Metabolic engineering of Synechocystis sp. strain PCC 6803 for isobutanol production. Appl Environ Microbiol 79:3908–3914
Wang B, Pugh S, Nielsen DR, Zhang W, Meldrum DR (2013) Engineering cyanobacteria for photosynthetic production of 3-hydroxybutyrate directly from CO2. Metab Eng 16:68–77
Wijffels RH, Kruse O, Hellingwerf KJ (2013) Potential of industrial biotechnology with cyanobacteria and eukaryotic microalgae. Curr Opin Biotechnol 24:405–413
Williams JGK (1988) Construction of specific mutations in photosystem II photosynthetic reaction center by genetic engineering methods in Synechocystis 6803. Methods Enzymol 167:766–778
Wilson A, Ajlani G, Verbavatz JM, Vass I, Kerfeld CA, Kirilovsky D (2006) A soluble carotenoid protein involved in phycobilisome-related energy dissipation in cyanobacteria. Plant Cell 18:992–1007
Zhou J, Zhang H, Zhang Y, Li Y, Ma Y (2012) Designing and creating a modularized synthetic pathway in cyanobacterium Synechocystis enables production of acetone from carbon dioxide. Metab Eng 14:394–400
Zurbriggen A, Kirst H, Melis A (2012) Isoprene production via the mevalonic acid pathway in Escherichia coli (Bacteria). BioEnergy Res 5:814–828
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Formighieri, C., Melis, A. Regulation of β-phellandrene synthase gene expression, recombinant protein accumulation, and monoterpene hydrocarbons production in Synechocystis transformants. Planta 240, 309–324 (2014). https://doi.org/10.1007/s00425-014-2080-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-014-2080-8