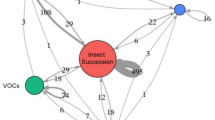
Abstract
Decomposition studies of vertebrate remains primarily focus on data that can be seen with the naked eye, such as arthropod or vertebrate scavenger activity, with little regard for what might be occurring with the microorganism community. Here, we discuss the necrobiome, or community of organisms associated with the decomposition of remains, specifically, the “epinecrotic” bacterial community succession throughout decomposition of vertebrate carrion. Pyrosequencing was used to (1) detect and identify bacterial community abundance patterns that described discrete time points of the decomposition process and (2) identify bacterial taxa important for estimating physiological time, a time–temperature metric that is often commensurate with minimum post-mortem interval estimates, via thermal summation models. There were significant bacterial community structure differences in taxon richness and relative abundance patterns through the decomposition process at both phylum and family taxonomic classification levels. We found a significant negative linear relationship for overall phylum and family taxon richness as decomposition progressed. Additionally, we developed a statistical model using high throughput sequencing data of epinecrotic bacterial communities on vertebrate remains that explained 94.4 % of the time since placement of remains in the field, which was within 2–3 h of death. These bacteria taxa are potentially useful for estimating the minimum post-mortem interval. Lastly, we provide a new framework and standard operating procedure of how this novel approach of using high throughput metagenomic sequencing has remarkable potential as a new forensic tool. Documenting and identifying differences in bacterial communities is key to advancing knowledge of the carrion necrobiome and its applicability in forensic science.



Similar content being viewed by others
References
Benbow M, Lewis A, Tomberlin J, Pechal J (2013) Seasonal necrophagous insect community assembly during vertebrate carrion decomposition. J Med Entomol 50(2):440–450
Michaud J-P, Moreau G (2009) Predicting the visitation of carcasses by carrion-related insects under different rates of degree-day accumulation. For Sci Int 185:78–83
Byrd JH, Allen JC (2001) The development of the black blow fly, Phormia regina (Meigen). For Sci Int 120(1–2):79–88
Byrd JH, Butler JF (1996) Effects of temperature on Cochliomyia macellaria (Diptera: Calliphoridae) development. J Med Entomol 33:901–905
Byrd JH, Butler JF (1997) Effects of Temperature on Chrysomya rufifacies (Diptera: Calliphoridae) development. J Med Entomol 34:353–358
Payne JA (1965) A summer carrion study of the baby pig Sus scrofa Linnaeus. Ecology 46(5):592–602
Schoenly K, Reid W (1987) Dynamics of heterotrophic succession in carrion arthropod assemblages: discrete seres or a continuum of change? Oecologia 73:192–202
Amendt J, Campobasso CP, Gaudry E, Reiter C, LeBlanc HN, Hall MJR (2007) Best practice in forensic entomology—standards and guidelines. Int J Legal Med 121(2):90–104
Byrd JH, Castner JL (2001) Forensic entomology: the utility of arthropods in legal investigations. CRC Press, Boca Raton, Florida
Villet MH (2011) African carrion ecosystems and their insect communities in relation to forensic entomology. Pest Technol 5(1):1–15
Greenberg B (1991) Flies as forensic indicators. J Med Entomol 28:565–577
Zimmerman KA, Wallace JR (2008) The potential to determine a postmortem submersion interval based on algal/diatom diversity on decomposing mammalian carcasses in brackish ponds in Delaware. J For Sci 53(4):935–941
Dickson GC, Poulter RTM, Maas EW, Probert PK, Kieser JA (2011) Marine bacterial succession as a potential indicator of postmortem submersion interval. For Sci Int 209(1–3):1–10
Hitosugi M, Ishii K, Yaguchi T, Chigusa Y, Kurosu A, Kido M, Nagai T, Tokudome S (2006) Fungi can be a useful forensic tool. Legal Medicine 8:240–242
Ronaghi M, Uhlén M, Nyrén P (1998) A sequencing method based on real-time pyrophosphate. Science 281(5375):363–365
Rothberg JM, Leamon JH (2008) The development and impact of 454 sequencing. Nat Biotechnol 26(10):1117–1124
Hudson ME (2008) Sequencing breakthroughs for genomic ecology and evolutionary biology. Mol Ecol Resour 8(1):3–17
Turnbaugh PJ, Quince C, Faith JJ, McHardy AC, Yatsunenko T, Niazi F, Affourtit J, Egholm M, Henrissat B, Knight R, Gordon JI (2010) Organismal, genetic, and transcriptional variation in the deeply sequenced gut microbiomes of identical twins. Proc Natl Acad Sci U S A 107(16):7503–7508
Edwards RA, Rodriguez-Brito B, Wegley L, Haynes M, Breitbart M, Peterson DM, Saar MO, Alexander S, Alexander EC, Rohwer F (2006) Using pyrosequencing to shed light on deep mine microbial ecology. BMC Genomics 7(57):1–7
The Human Microbiome Jumpstart Reference Strains Consortium (2010) A catalog of reference genomes from the human microbiome. Science 328(5981):994–999
Mahowald MA, Rey FE, Seedorf H, Turnbaugh PJ, Fulton RS, Wollam A, Shah N, Wang C, Magrini V, Wilson RK, Cantarel BL, Coutinho PM, Henrissat B, Crock LW, Russell A, Verberkmoes NC, Hettich RL, Gordon JI (2009) Characterizing a model human gut microbiota composed of members of its two dominant bacterial phyla. Proc Nat Acad Sci U S A 106:5859–5864
Price LB, Liu CM, Melendez JH, Frankel YM, Engelthaler D, Aziz M, Bowers J, Rattray R, Ravel J, Kingsley C, Keim PS, Lazarus GS, Zenilman JM (2009) Community analysis of chronic wound bacteria using 16S rRNA gene-based pyrosequencing: impact of diabetes and antibiotics on chronic wound microbiota. PLoS One 4(7):e6462
Petrosino JF, Highlander S, Luna RA, Gibbs RA, Versalovic J (2009) Metagenomic pyrosequencing and microbial identification. Clin Chem 55(5):856–866
Costello EK, Lauber CL, Hamady M, Fierer N, Gordon JI, Knight R (2009) Bacterial community variation in human body habitats across space and time. Science 326(5960):1694–1697
Schoenly KG, Haskell NH, Hall RD, Gbur JR (2007) Comparative performance and complementarity of four sampling methods and arthropod preference tests from human and porcine remains at the forensic anthropology center in Knoxville, Tennessee. J Med Entomol 44(5):881–894
Lewis AJ, Benbow ME (2011) When entomological evidence crawls away: Phormia regina en masse larval dispersal. J Med Entomol 48(6):1112–1119
Megyesi MS, Nawrocki SP, Haskell NH (2005) Using accumulated degree-days to estimate the postmortem interval from decomposed human remains. J For Sci 50(3):618–626
Michaud JP, Moreau G (2011) A statistical approach based on accumulated degree-days to predict decomposition-related processes in forensic studies. J For Sci 56(1):229–232
Catts EP (1992) Problems in estimating the postmortem interval in death investigations. J Agricul Entomol 9(4):245–255
Dowd SE, Wolcott RD, Sun Y, McKeehan T, Smith E, Rhoads D (2008) Polymicrobial nature of chronic diabetic foot ulcer biofilm infections determined using bacterial tag encoded FLX amplicon pyrosequencing (bTEFAP). PLoS One 3(10):e3326
Sen R, Ishak HD, Estrada D, Dowd SE, Hong E, Mueller UG (2009) Generalized antifungal activity and 454-screening of Pseudonocardia and Amycolatopsis bacteria in nests of fungus-growing ants. Proc Natl Acad Sci U S A 106(42):17805–17810
Handl S, Dowd SE, Garcia-Mazcorro JF, Steiner JM, Suchodolski JS (2011) Massive parallel 16S rRNA gene pyrosequencing reveals highly diverse fecal bacterial and fungal communities in healthy dogs and cats. FEMS Microbiol Ecol 76(2):301–310
Andreotti R, Perez de Leon AA, Dowd SE, Guerrero FD, Bendele KG, Scoles GA (2011) Assessment of bacterial diversity in the cattle tick Rhipicephalus (Boophilus) microplus through tag-encoded pyrosequencing. BMC Microbiol 11(1):1–6
Gontcharova V, Youn E, Wolcott RD, Hollister EB, Gentry TJ, Dowd SE (2010) Black Box Chimera Check (B2C2): a Windows-based software for batch depletion of chimeras from bacterial 16S rRNA gene datasets. Open Microbiol J 4:6
Dowd SE, Callaway TR, Wolcott RD, Sun Y, McKeehan T, Hagevoort RG, Edrington TS (2008) Evaluation of the bacterial diversity in the feces of cattle using 16S rDNA bacterial tag-encoded FLX amplicon pyrosequencing (bTEFAP). BMC Microbiol 8:125
Wolcott RD, Gontcharova V, Sun Y, Dowd SE (2009) Evaluation of the bacterial diversity among and within individual venous leg ulcers using bacterial tag-encoded FLX and titanium amplicon pyrosequencing and metagenomic approaches. BMC Microbiol 9:226
Suchodolski JS, Dowd SE, Westermarck E, Steiner JM, Wolcott RD, Spillmann T, Harmoinen JA (2009) The effect of the macrolide antibiotic tylosin on microbial diversity in the canine small intestine as demonstrated by massive parallel 16S rRNA gene sequencing. BMC Microbiol 9:210
Li W, Dowd SE, Scurlock B, Acosta-Martinez V, Lyte M (2009) Memory and learning behavior in mice is temporally associated with diet-induced alterations in gut bacteria. Physiol Behav 96(4–5):557–567
Ishak HD, Plowes R, Sen R, Kellner K, Meyer E, Estrada DA, Dowd SE, Mueller UG (2011) Bacterial diversity in Solenopsis invicta and Solenopsis geminata ant colonies characterized by 16S amplicon 454 pyrosequencing. Microb Ecol 61(4):821–831
Nawrocki E, Kolbe D, Eddy S (2009) Infernal 1.0: inference of RNA alignments. Bioinformatics 25:1335–1337
Nawrocki EP, Eddy SR (2007) Query-dependent banding (QBD) for faster RNA similarity searches. PLoS Comput Biol 3:540–554
Garrity GM, Bell JA, Lilburn TG (2004) Taxonomic outline of the prokaryotes. In: Garrity GM (ed) Bergey's manual of systematic bacteriology, vol 2, 2nd edn. Springer, New York, New York, pp 4–23
Wang Q, Garrity GM, Tiedje JM, Cole JR (2007) Naïve Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl Environ Microbiol 73:5261–5267
R Development Core Team (2010) R: a language and environment for statistical computing. Vienna, Austria ISBN 3-900051-07-0: http://www.R-project.org
Anderson MJ (2001) A new method for non-parametric multivariate analysis of variance. Austral Ecol 26(1):32–46
McCune B, Mefford MJ (2006) PC-ORD
McCune B, Grace JB (2002) Analysis of ecological communities. MjM Software Design, Gleneden Beach, Oregon
Biondini ME, Bonham CD, Redente EF (1985) Secondary successional patterns in a sagebrush (Artemisia tridentata) community as they relate to soil disturbance and soil biological activity. Vegetation 60(1):25–36
Hastie T, Tibshirani R (1990) Generalized additive models. Chapman and Hall ⁄ CRC, Boca Raton
Panikov NS, Sizova MV (2007) Growth kinetics of microorganisms isolated from Alaskan soil and permafrost in solid media frozen down to −35 °C. FEMS Microbiol Ecol 59(2):500–512
Prescott LM, Harley JP, Klein DA (2004) Microbiology. 6th edition edn. McGraw-Hill Science/Engineering/Math.
Byrd JH, Castner JL (2010) Insects of forensic importance. In: Byrd JH, Castner JL (eds) Forensic entomology: the utility of arthropods in legal investigations, 2nd edn. CRC Press, Boca Raton, Florida, pp 39–126
Wilson EE, Wolkovich EM (2011) Scavenging: how carnivores and carrion structure communities. Trends Ecol Evol 26(3):129–135
Stadler S, Stefanuto P-H, Brokl M, Forbes SL, Focant J-F (2012) Characterization of volatile organic compounds from human analogue decomposition using thermal desorption coupled to comprehensive two-dimensional gas chromatography—time-of-flight mass spectrometry. Anal Chem 85(2):998–1005
Dekeirsschieter J, Verheggen FJ, Gohy M, Hubrecht F, Bourguignon L, Lognay G, Haubruge E (2009) Cadaveric volatile organic compounds released by decaying pig carcasses (Sus domesticus L.) in different biotopes. Forensic Science International 189(1-3):46–53
Grice EA, Kong HH, Renaud G, Young AC, Bouffard GG, Blakesley RW, Wolfsberg TG, Turner ML, Segre JA, NISC Comparative Sequencing Program (2008) A diversity profile of the human skin microbiota. Genome Res 18(7):1043–1050
Sibley C, Church D, Surette M, Dowd S, Parkins M (2012) Pyrosequencing reveals the complex polymicrobial nature of invasive pyogenic infections: microbial constituents of empyema, liver abscess, and intracerebral abscess. European Journal of Clinical Microbiology & Infectious Diseases 31(10):2679–2691
Ursell LK, Clemente JC, Rideout JR, Gevers D, Caporaso JG, Knight R (2012) The interpersonal and intrapersonal diversity of human-associated microbiota in key body sites. J Allerg Clin Immunol 129(5):1204–1208
Dewhirst FE, Chen T, Izard J, Paster BJ, Tanner ACR, Yu W-H, Lakshmanan A, Wade WG (2010) The human oral microbiome. J Bacteriol 192(19):5002–5017
Carter DO, Tibbett M (2003) Taphonomic mycota: fungi with forensic potential. J For Sci 48(1):168–171
Mille–Lindblom C, Fischer H, Tranvik LJ (2006) Antagonism between bacteria and fungi: substrate competition and a possible tradeoff between fungal growth and tolerance towards bacteria. Oikos 113(2):233–242
Boer W, Folman LB, Summerbell RC, Boddy L (2006) Living in a fungal world: impact of fungi on soil bacterial niche development. FEMS Microbiol Rev 29(4):795–811
Ferrandon D, Imler J-L, Hetru C, Hoffmann JA (2007) The Drosophila systemic immune response: sensing and signalling during bacterial and fungal infections. Nat Rev Immunol 7(11):862–874
Gottar M, Gobert V, Michel T, Belvin M, Duyk G, Hoffmann JA, Ferrandon D, Royet J (2002) The Drosophila immune response against Gram-negative bacteria is mediated by a peptidoglycan recognition protein. Nature 416(6881):640–644
Gerardo NM, Altincicek B, Anselme C, Atamian H, Barribeau SM, Md V, Duncan EJ, Evans JD, Gabaldón T, Ghanim M, Heddi A, Kaloshian I, Latorre A, Moya A, Nakabachi A, Parker BJ, Pérez-Brocal V, Pignatelli M, Rahbé Y, Ramsey JS, Spragg CJ, Tamames J, Tamarit D, Tamborindeguy C, Vincent-Monegat C, Vilcinskas A (2010) Immunity and other defenses in pea aphids. Acyrthosiphon pisum Genome Biol 11:R21
Wilson-Rich N, Spivak M, Fefferman NH, Starks PT (2009) Genetic, individual, and group facilitation of disease resistance in insect societies. Annual Rev Entomol 54:405–423
Suwannapong G, Benbow M, Nieh J (2011) Biology of Thai honeybees: natural history and threats. In: Florio R (ed) Bees: biology, threats and colonies. Nova Science Publishers, Inc, Hauppauge, NY, pp 1–101
Rozen DE, Engelmoer DJP, Smiseth PT (2008) Antimicrobial strategies in burying beetles breeding on carrion. Proc Natl Acad Sci U S A 105(46):17890–17895
Kerridge A, Lappin-Scott H, Stevens JR (2005) Antibacterial properties of larval secretions of the blowfly, Lucilia sericata. Med Vet Entomol 19:333–337
Nigam Y, Bexfield A, Thomas S, Ratcliffe NA (2006) Maggot therapy: the science and implication for CAM—Part II—maggots combat infection. Evidence-Based Complement Alter Med 3(3):303–308
Sherman RA, Hall MJR, Thomas S (2000) Medicinal maggots: an ancient remedy for some contemporary afflictions. Ann Rev Entomol 45(1):55–81
Mumcuoglu KY, Miller J, Mumcuoglu M, Friger M, Tarshis M (2001) Destruction of bacteria in the digestive tract of the maggot of Lucilia sericata (Diptera: Calliphoridae). J Med Entomol 38(2):161–166
Tomberlin JK, Crippen TL, Tarone AM, Singh B, Adams K, Rezenom YH, Benbow ME, Flores M, Longnecker M, Pechal JL, Russell DH, Beier RC, Wood TK (2012) Interkingdom responses of flies to bacteria mediated by fly physiology and bacterial quorum sensing. Anim Behav 84(6):1449–1456
Ma Q, Fonseca A, Liu W, Fields AT, Pimsler ML, Spindola AF, Tarone AM, Crippen TL, Tomberlin JK, Wood TK (2012) Proteus mirabilis interkingdom swarming signals attract blow flies. The ISME Journal:1–11
Ieno EN, Amendt J, Fremdt H, Saveliev AA, Zuur AF (2010) Analysing forensic entomology data using additive mixed effects modelling. In: Current Concepts in Forensic Entomology. Springer, Dordrecht, pp 139–162. ISBN 978-1-4020-9683-9
Tarone AM, Foran DR (2008) Generalized additive models and Lucilia sericata growth: assessing confidence intervals and error rates in forensic entomology. J For Sci 53(4):942–948
Wells J, LaMotte LR (1995) Estimating maggot age from weight using inverse prediction. J For Sci 40:585–585
Clark K, Evans L, Wall R (2006) Growth rates of the blowfly, Lucilia sericata, on different body tissues. For Sci Int 156(2–3):145–149
Donovan SE, Hall MJR, Turner BD, Moncrieff CB (2006) Larval growth rates of the blowfly, Calliphora vicina, over a range of temperatures. Med Vet Entomol 20(1):106–114
Nabity PD, Higley LG, Heng-Moss TM (2006) Effects of temperature on development of Phormia regina (Diptera: Calliphoridae) and use of developmental data in determining time intervals in forensic entomology. J Med Entomol 43(6):1276–1286
Tarone AM, Foran DR (2011) Gene Expression During Blow Fly Development: Improving the Precision of Age Estimates in Forensic Entomology. J For Sci 56:S112–S122
Michaud JP, Majka CG, Prive JP, Moreau G (2010) Natural and anthropogenic changes in the insect fauna associated with carcasses in the North American Maritime lowlands. Forensic Sci Int 202(1–3):64–70
Shendure J, Ji H (2008) Next-generation DNA sequencing. Nat Biotechnol 26(10):1135–1145
Richards CS, Villet MH (2009) Data quality in thermal summation development models for forensically important blowflies. Med Vet Entomol 23(3):269–276
Richards C, Paterson I, Villet M (2008) Estimating the age of immature Chrysomya albiceps (Diptera: Calliphoridae), correcting for temperature and geographical latitude. Int J Legal Med 122(4):271–279
Boatright SA, Tomberlin JK (2010) Effects of temperature and tissue type of Cochliomyia macellaria (Diptera: Calliphoridae). Journal of Medical Entomology 37(5):917–923
Acknowledgments
We thank A. Lewis, T. Blair, and J. White for their help during the study. The Blair family is gratefully acknowledged for allowing access to their property for this research. We thank B. Singh for assistance in the metagenomic data sequence classification processing. Financial support was given to JLP from the Department of Entomology and the Whole Systems Genome Initiative at Texas A&M University. AMT and JKT would like to thank the Department of Entomology at Texas A&M University and Texas A&M University AgriLife Research for financial support of this project. MEB was supported by the University of Dayton Research Council and the Department of Biology.
Disclaimer
This project was funded (TLC, MEB, AMT, and JKT), in part, by the National Institute of Justice, Office of Justice Programs, US Department of Justice through Grant 2010-DN-BX-K243. Points of view in this document are those of the authors and do not necessarily represent the official position or policies of the U.S. Department of Justice. Mention of trade names, companies, or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement of the products by the US Department of Agriculture
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. S1
(DOCX 229 kb)
Fig. S2
(DOCX 5086 kb)
Fig. S3
(DOCX 164 kb)
Fig. S4
(DOCX 240 kb)
Table S1
(DOCX 52 kb)
Table S2
(DOCX 85 kb)
Table S3
(DOCX 42 kb)
Table S4
(DOCX 44 kb)
Table S5
(DOCX 45 kb)
Table S6
(DOCX 39 kb)
Table S7
(DOCX 42 kb)
Table S8
(DOCX 44 kb)
Table S9
(DOCX 74 kb)
Rights and permissions
About this article
Cite this article
Pechal, J.L., Crippen, T.L., Benbow, M.E. et al. The potential use of bacterial community succession in forensics as described by high throughput metagenomic sequencing. Int J Legal Med 128, 193–205 (2014). https://doi.org/10.1007/s00414-013-0872-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00414-013-0872-1