Abstract
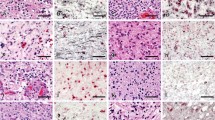
Doublecortin (DCX) is required for neuroblastic migration during the development of the cerebral cortex. DCX is a microtubule-associated protein that plays a role in cellular motility. These facts led us to hypothesize that DCX is increased in invasive brain tumors. DCX expression was assessed in 69 paraffin-embedded brain tumors of neuroepithelial origin. In addition, mouse brain sections of the subventricular zone and dentate gyrus were used as positive controls for immunostaining, and specificity of antibody staining was demonstrated by peptide neutralization. DCX was highly expressed in both high-grade invasive tumors (glioblastoma, n=11; anaplastic astrocytoma/oligoastrocytoma, n=7; and medulloblastoma/PNET, n=6) and low-grade invasive tumors (oligodendroglioma, n=3; and astrocytoma/oligoastrocytoma, n=5). However, DCX was less intensely expressed in the circumscribed group of tumors (pilocytic astrocytoma, n=6; ependymoma/subependymoma, n=7; dysembryoplastic neuroepithelial tumor, n=4; ganglioglioma, n=2; meningioma, n=9; and schwannoma, n=9). By the Cochran-Mantel-Haenszel statistical test, the circumscribed group was significantly different from both the high-grade invasive group (P<0.0001) and the low-grade invasive group (P<0.0001). We conclude that DCX is preferentially expressed in invasive brain tumors. In addition, DCX immunostaining was stronger at the margin of the tumor than at the center. For a subset of these tumors, we also detected DCX mRNA and protein by Northern and Western blotting. DCX mRNA and protein was detected in glioma cell lines by Northern blotting, immunofluorescence microscopy and Western blotting. Collectively, the immunohistochemistry, Western blots and Northern blots conclusively demonstrate expression of DCX by human brain tumors.






Similar content being viewed by others
References
Almqvist PM, Mah R, Lendahl U, Jacobsson B, Hendson G (2002) Immunohistochemical detection of nestin in pediatric brain tumors. J Histochem Cytochem 50:147–158
Bachoo RM, Maher EA, Ligon KL, Sharpless NE, Chan SS, You MJ, Tang Y, DeFrances J, Stover E, Weissleder R, Rowitch DH, Louis DN, DePinho RA (2002) Epidermal growth factor receptor and Ink4a/Arf. Convergent mechanisms governing terminal differentiation and transformation along the neural stem cell to astrocyte axis. Cancer Cell 1:269–277
Bai J, Ramos RL, Ackman JB, Thomas AM, Lee RV, LoTurco JJ (2003) RNAi reveals doublecortin is required for radial migration in rat neocortex. Nat Neurosci 6:1277–1283
Becker AJ, Klein H, Baden T, Aigner L, Normann S, Elger CE, Schramm J, Wiestler OD, Blumcke I (2002) Mutational and expression analysis of the reelin pathway components CDK5 and doublecortin in gangliogliomas. Acta Neuropathol 104:403–408
Branle F, Lefranc F, Camby I, Jeuken J, Geurts-Moespot A, Sprenger S, Sweep F, Kiss R, Salmon I (2002) Evaluation of the efficiency of chemotherapy in in vivo orthotopic models of human glioma cells with and without 1p19q deletions and in C6 rat orthotopic allografts serving for the evaluation of surgery combined with chemotherapy. Cancer 95:641–655
Brown JP, Couillard-Despres S, Cooper-Kuhn CM, Winkler J, Aigner L, Kuhn HG (2003) Transient expression of doublecortin during adult neurogenesis. J Comp Neurol 467:1–10
Dahlstrand J, Collins VP, Lendahl U (1992) Expression of the class VI intermediate filament nestin in human central nervous system tumors. Cancer Res 52:5334–5341
Dai C, Celestino JC, Okada Y, Louis DN, Fuller GN, Holland EC (2001) PDGF autocrine stimulation dedifferentiates cultured astrocytes and induces oligodendrogliomas and oligoastrocytomas from neural progenitors and astrocytes in vivo. Genes Dev 15:1913–1925
Feng Y, Walsh CA (2001) Protein-protein interactions, cytoskeletal regulation and neuronal migration. Nat Rev Neurosci 2:408–416
Francis F, Koulakoff A, Boucher D, Chafey P, Schaar B, Vinet MC, Friocourt G, McDonnell N, Reiner O, Kahn A, McConnell SK, Berwald-Netter Y, Denoulet P, Chelly J (1999) Doublecortin is a developmentally regulated, microtubule-associated protein expressed in migrating and differentiating neurons. Neuron 23:247–256
Friocourt G, Koulakoff A, Chafey P, Boucher D, Fauchereau F, Chelly J, Francis F (2003) Doublecortin functions at the extremities of growing neuronal processes. Cereb Cortex 13:620–626
Gdalyahu A, Ghosh I, Levy T, Sapir T, Sapoznik S, Fishler Y, Azoulai D, Reiner O (2004) DCX, a new mediator of the JNK pathway. EMBO J 23:823–32
Giese A, Bjerkvig R, Berens ME, Westphal M (2003) Cost of migration: invasion of malignant gliomas and implications for treatment. J Clin Oncol 21:1624–1636
Gladson CL, Pijuan-Thompson V, Olman MA, Gillespie GY, Yacoub IZ (1995) Up-regulation of urokinase and urokinase receptor genes in malignant astrocytoma. Am J Pathol 146:1150–1160
Gladson CL, Wilcox JN, Sanders L, Gillespie GY, Cheresh DA (1995) Cerebral microenvironment influences expression of the vitronectin gene in astrocytic tumors. J Cell Sci 108:947–956
Gleeson JG, Lin PT, Flanagan LA, Walsh CA (1999) Doublecortin is a microtubule-associated protein and is expressed widely by migrating neurons. Neuron 23:257–271
Holland EC, Hively WP, DePinho RA, Varmus HE (1998) A constitutively active epidermal growth factor receptor cooperates with disruption of G1 cell-cycle arrest pathways to induce glioma-like lesions in mice. Genes Dev 12:3675–3685
Holland EC, Celestino J, Dai C, Schaefer L, Sawaya RE, Fuller GN (2000) Combined activation of Ras and Akt in neural progenitors induces glioblastoma formation in mice. Nat Genet 25:55–57
Horesh D, Sapir T, Francis F, Wolf SG, Caspi M, Elbaum M, Chelly J, Reiner O (1999) Doublecortin, a stabilizer of microtubules. Hum Mol Genet 8:1599–1610
Jang T, Litofsky NS, Smith TW, Ross AH, Recht LD (2004) Aberrant nestin expression during ethylnitrosourea-(ENU)-induced neurocarcinogenesis. Neurobiol Dis 15:544–552
Kato M, Dobyns WB (2003) Lissencephaly and the molecular basis of neuronal migration. Hum Mol Genet 12:R89–96
Khoshyomn S, Penar PL, Wadsworth MP, Taatjes DJ (1997) Localization of CD44 at the invasive margin of glioblastomas by immunoelectron microscopy. Ultrastruct Pathol 21:517–525
Kizhatil K, Wu YX, Sen A, Bennett V (2002) A new activity of doublecortin in recognition of the phospho-FIGQY tyrosine in the cytoplasmic domain of neurofascin. J Neurosci 22:7948–7958
Kleihues P, Cavenee WK (2000) Tumours of the nervous system. International Agency for Research on Cancer, Lyon
Lee A, Maldonado M, Baybis M, Walsh CA, Scheithauer B, Yeung R, Parent J, Weiner HL, Crino PB (2003) Markers of cellular proliferation are expressed in cortical tubers. Ann Neurol 53:668–673
Maher EA, Furnari FB, Bachoo RM, Rowitch DH, Louis DN, Cavenee WK, DePinho RA (2001) Malignant glioma: genetics and biology of a grave matter. Genes Dev 15:1311–1333
Mantel N (1963) Chi-square tests with one degree of freedom: extensions of the Mantel-Haenszel procedure. J Am Statist Assoc 58:690–700
Miyata H, Chute DJ, Fink J, Villablanca P, Vinters HV (2004) Lissencephaly with agenesis of corpus callosum and rudimentary dysplastic cerebellum: a subtype of lissencephaly with cerebellar hypoplasia. Acta Neuropathol 107:69–81
Mizuguchi M, Qin J, Yamada M, Ikeda K, Takashima S (1999) High expression of doublecortin and KIAA0369 protein in fetal brain suggests their specific role in neuronal migration. Am J Pathol 155:1713–1721
Nacher J, Crespo C, McEwen BS (2001) Doublecortin expression in the adult rat telencephalon. Eur J Neurosci 14:629–644
Oliver TG, Wechsler-Reya RJ (2004) Getting at the root and stem of brain tumors. Neuron 42:885–888
Olson EC, Walsh CA (2002) Smooth, rough and upside-down neocortical development. Curr Opin Genet Dev 12:320–327
Recht L, Jang T, Savarese T, Litofsky NS (2003) Neural stem cells and neuro-oncology: Quo vadis? J Cell Biochem 88:11–19
Rich JN, Bigner DD (2004) Development of novel targeted therapies in the treatment of malignant glioma. Nat Rev Drug Discov 3:430–446
Rich JN, Hans C, Jones B, Iversen ES, McLendon RE, Rasheed BK, Dobra A, Dressman HK, Bigner DD, Nevins JR, West M (2005) Gene expression profiling and genetic markers in glioblastoma survival. Cancer Res 65:4051–4058
Salajegheh M, Rudnicki A, Smith TW (2005) Expression of urokinase-type plasminogen activator receptor (uPAR) in primary central nervous system neoplasms. Appl Immunohistochem Mol Morphol 13:184–189
Schaffner W, Weismann C (1973) A rapid, sensitive, and specific method for the determination of protein in dilute solution. Anal Biochem 56:502–514
Steiner B, Kronenberg G, Jessberger S, Brandt MD, Reuter K, Kempermann G (2004) Differential regulation of gliogenesis in the context of adult hippocampal neurogenesis in mice. Glia 46:41–52
Tanaka T, Serneo FF, Higgins C, Gambello MJ, Wynshaw-Boris A, Gleeson JG (2004) Lis1 and doublecortin function with dynein to mediate coupling of the nucleus to the centrosome in neuronal migration. J Cell Biol 165:709–721
Taylor KR, Holzer AK, Bazan JF, Walsh CA, Gleeson JG (2000) Patient mutations in doublecortin define a repeated tubulin-binding domain. J Biol Chem 275:34442–34450
Tohyama T, Lee VM, Rorke LB, Marvin M, McKay RD, Trojanowski JQ (1992) Nestin expression in embryonic human neuroepithelium and in human neuroepithelial tumor cells. Lab Invest 66:303–313
Uhrbom L, Dai C, Celestino JC, Rosenblum MK, Fuller GN, Holland EC (2002) Ink4a-Arf loss cooperates with KRas activation in astrocytes and neural progenitors to generate glioblastomas of various morphologies depending on activated Akt. Cancer Res. 62:5551–5558
Vallee RB, Tai C, Faulkner NE (2001) LIS1: cellular function of a disease-causing gene. Trends Cell Biol 11:155–160
Yang HK, Sundholm-Peters NL, Goings GE, Walker AS, Hyland K, Szele FG (2004) Distribution of doublecortin expressing cells near the lateral ventricles in the adult mouse brain. J Neurosci Res 76:282–295
Zagzag D, Zhong H, Scalzitti JM, Laughner E, Simons JW, Semenza GL (2000) Expression of hypoxia-inducible factor 1a in brain tumors: association with angiogenesis, invasion, and progression. Cancer 88:2606–2618
Acknowledgements
We thank Li Li for her discussions of her studies of DCX expression in neural precursor cells and Qin Liu for her help with the statistical analyses. This study was supported by NIH grants CA-10737 and NS-21716 to AHR.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Daou, MC., Smith, T.W., Litofsky, N.S. et al. Doublecortin is preferentially expressed in invasive human brain tumors. Acta Neuropathol 110, 472–480 (2005). https://doi.org/10.1007/s00401-005-1070-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00401-005-1070-0