Abstract
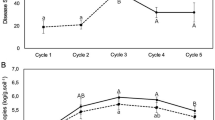
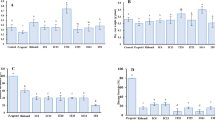
Stenotrophomonas rhizophila DSM14405T is of high biotechnological interest as plant growth stimulator, especially for salinated conditions. The objective of this study was to determine the effect of plant species (cotton, tomato, and sweet pepper) on colonisation and plant growth promotion of this beneficial bacterium in gnotobiotic systems and in non-sterile soil. All plant structures (leaves, stems, and roots) were densely colonised by DSM14405T reaching up to 109 cells g−1 fresh weight; under gnotobiotic conditions the abundances were 4–5 orders of magnitude higher than in non-sterile soil. Under non-sterile conditions and ambient humidity, tomato shoots were more densely colonised than shoots of sweet pepper and cotton. S. rhizophila DSM14405T was shown to grow endophytically and colonise the vicinity of root hairs of tomato. Plant growth promotion was particularly apparent in tomato. In general, the impact of plant species on colonisation and plant growth promotion was more pronounced in soil than under gnotobiotic conditions and likely due to the control of diseases and deleterious microorganisms. S. rhizophila DSM14405T was shown to control diseases in sweet pepper and in cotton. Molecular profiling via single strand conformation polymorphism of internal transcribed spacers and 16S rRNA genes (PCR-single strand conformational polymorphism (SSCP)) revealed that S. rhizophila DSM14405T strongly affected fungal, but not bacterial communities in the rhizosphere of tomato and sweet pepper. Major SSCP bands related to uncultured fungi and Candida subhashii, disappeared in tomato rhizosphere after Stenotrophomonas treatment. This suggests an indirect, species-specific plant growth promotion effect of S. rhizophila via the elimination of deleterious rhizosphere organisms.




Similar content being viewed by others
References
Adam H, Groenewald M, Mohan S, Richardson S, Bunn U, Gibas CF, Poutanen S, Sigler L (2009) Identification of a new species, Candida subhashii, as a cause of peritonitis. Med Mycol 47:305–311
Altschul SF, Madden TL, Schaffer AA, Zhang JH, Zhang Z, Miller W, Lipman (1997) Gapped BLAST and PSI BLAST: a new generation of protein database search programmes. Nucleic Acids Res 25:3389–3402
Amann RI, Binder BJ, Olson RJ, Chisholm SW, Devereux R, Stahl DA (1990) Combination of 16S rRNA-targeted oligonucleotide probes with flow cytometry for analyzing mixed microbial populations. Appl Environ Microbiol 56:1919–1925
Barret M, Morrissey JP, O’Gara F (2011) Functional genomics analysis of plant growth-promoting rhizobacterial traits involved in rhizosphere competence. Biol Fertils Soils 47:729–743
Bassam BJ, Caetano-Anolles G, Gresshoff PM (1991) Fast and sensitive silver staining of DNA in polyacrylamide gels. Analyt Biochem 80:81–84
Berg G (2009) Plant-microbe interactions promoting plant growth and health: perspective for controlled use of microorganisms in agriculture. Appl Microbiol Biotechnol 84:11–18
Berg G, Smalla K (2009) Plant species and soil type cooperatively shape the structure and function of microbial communities in the rhizosphere. FEMS Microbiol Ecol 68:1–13
Berg G, Zachow C (2011) PGPR interplay with rhizosphere communities and effect on plant growth and health. In: Maheshwari DK (ed) Bacteria in agrobiology: crop ecosystems. Springer, Heidelberg, pp 97–110
Berg G, Knaape C, Ballin G, Seidel D (1994) Biological control of Verticillium dahliae KLEB by naturally occurring rhizosphere bacteria. Arch Phytopathol Dis Prot 29:249–262
Berg G, Eberl L, Hartmann A (2005a) The rhizosphere as a reservoir for opportunistic human pathogenic bacteria. Environ Microbiol 7:1673–1685
Berg G, Krechel A, Ditz M, Faupel A, Ulrich A, Hallmann J (2005b) Comparison of endophytic and ectophytic potato-associated bacterial communities and their antagonistic activity against plant pathogenic fungi. FEMS Microbiol Ecol 51:215–229
Berg G, Alavi M, Schmidt CS, Zachow C, Egamberdieva D, Kamilova F, Lugtenberg (2012) Biocontrol and osmoprotection for plants under saline conditions. In: de Bruijn (ed). Molecular microbial ecology of the rhizosphere. Wiley: New York
Cardinale M, de Castro V, Jr J, Müller H, Berg G, Grube M (2008) In situ analysis of the bacterial community associated with the reindeer lichen Cladonia arbuscula reveals predominance of Alphaproteobacteria. FEMS Microbiol Ecol 66:63–71
Da Silva JC, Albuquerque MC, de Mendonça EAF, Eliane M (2006) Cotton seed performance after processing and storage. Revista Brasileira de Sementes 28:79–85
Daims H, Brühl A, Amann R, Schleifer K-H, Wagner M (1999) The domain-specific probe EUB338 is insufficient for the detection of all bacteria: development and evaluation of a more comprehensive probe set. Syst Appl Microbiol 22:434–444
Daims H, Stoecker K, Wagner M (2005) Fluorescence in situ hybridization for the detection of prokaryotes. In: Osborn AM, Smith CJ (eds) Advanced methods in molecular microbial ecology. Bios-Garland, Abingdon, UK, pp 213–239
Dubuis C, Haas D (2007) Cross-species GacA-controlled induction of antibiosis in pseudomonads. Appl Environ Microbiol 73:650–654
Dubuis C, Keel C, Haas D (2007) Dialogues of root colonizing pseudomonads. Eur J Plant Pathol 119:311–328
Dunne C, Moënne-Loccoz Y, McCarthy J, Higgins P, Powell J, Dowling DN, O’Gara F (1998) Combining proteolytic and phloroglucinol-producing bacteria for improved biocontrol of Pythium-mediated damping-off of sugar beet. Plant Pathol 47:299–307
Egamberdieva D, Kucharova Z, Davranov K, Berg G, Makarova N, Azarova T, Chebotar V, Tikhonovich I, Kamilova F, Validov SZ, Lugtenberg B (2011) Bacteria able to control foot and root rot and to promote growth of cucumber in salinated soils. Biol Fertil Soils 47:197–205
Egamberdiyeva D, Kamilova F, Validov S, Gafurova L, Kucharova Z, Lugtenberg B (2008) High incidence of plant growth-stimulating bacteria associated with the rhizosphere of wheat grown on salinated soil in Uzbekistan. Environ Microbiol 10:1–9
FAO, Rome (Italy), Land and Water Development Div.; International Programme for Technology and Research in Irrigation and Drainage, Rome (Italy). Secretariat; Centre Virtuel de l’Eau Agricole et ses Usages, Rome (Italy). Centre d’Information sur l’Eau Agricole et ses Usage (2005) Management of irrigation-induced salt-affected soils. CISEAU/FAO/IPTRID, Rome
Fujita Y, Nakahara H (2006) Variations in the microalgal structure in paddy soil in Osaka, Japan: comparison between surface and subsurface soils. Limnology 7:83–91
Fürnkranz M, Müller H, Berg G (2009) Characterization of plant growth promoting bacteria from crops in Bolivia. J Plant Dis Plant Prot 116:149–155
Gantner S, Schmid M, Duerr C, Schuhegger R, Steidle A, Hutzler P, Langebartels C, Eberl L, Hartmann A, Dazzo FB (2006) In situ quantitation of the spatial scale of calling distances and population density-independent N-acylhomoserine lactone mediated communication by rhizobacteria colonized on plant roots. FEMS Microbiol Ecol 56:188–194
Götz M, Gomes NCM, Dratwinski A, Costa R, Berg G, Peixoto R, Mendonc L, Hagler LM, Smalla K (2006) Survival of gfp-tagged antagonistic bacteria in the rhizosphere of tomato plants and their effects on the indigenous bacterial community. FEMS Microbiol Ecol 52:207–218
Gould JH, Magallenes-Cedeno M (1998) Adaptation of cotton shoot apex culture to Agrobacterium mediated transformation. Plant Mol Biol Reporter 16:1–10
Hagemann M, Hasse D, Berg G (2006) Detection of a phage genome carrying a zonula occludens like toxin gene (zot) in clinical isolates of Stenotrophomonas maltophilia. Arch Microbiol 185:449–458
Hagemann M, Ribbeck-Busch K, Klähn S, Hasse D, Steinbruch R, Berg G (2008) Synthesis of the compatible solute glucosylglycerol in Stenotrophomonas rhizophila. J Bac 190:5898–5906
Hartmann A, Schmid M, van Tuinen D, Berg G (2009) Plant-driven selection of microbes. Plant Soil 321:235–257
Howell AB, Francois L, Erwin DC (1994) Interactive effect of salinity and Verticillium albo-atrum on verticillium wilt disease severity and yield of two alfalfa cultivars. Field Crops Res 37:247–251
Jensen LE, Nybroe O (1999) Nitrogen availability to Pseudomonas fluorescens DF57 is limited during decomposition of barley straw in bulk soil in the barley rhizosphere. Appl Environ Microbiol 65:4320–4328
Ji P, Wilson M (2002) Assessment of the importance of similarity in carbon source utilization profiles between the biological control agent and the pathogen in biological control of bacterial speck of tomato. Appl Environ Microbiol 68:4383–4389
Johnson JL, Marvin W, Fawley MW, Fawley KP (2007) The diversity of Scenedesmus and Desmodesmus (Chlorophyceae) in Itasca State Park, Minnesota, USA. Phycologia 46:214–229
Kai M, Effmert U, Berg G, Piechulla B (2007) Volatiles of bacterial antagonists inhibit mycelial growth of the plant pathogen Rhizoctonia solani. Arch Microbiol 187:351–360
Kremer RJ, Kennedy AC (1996) Rhizobacteria as biocontrol agents of weeds. Weed Technol 10:601–609
Larena I, Salazar O, González V, Marıá C, Julián MC, Rubio V (1999) Design of a primer for ribosomal DNA internal transcribed spacer with enhanced specificity for ascomycetes. J Biotechnol 75:187–194
Lugtenberg B, Kamilova F (2009) Plant growth-promoting rhizobacteria. Annu Rev Microbiol 63:541–556
Manz W, Amann R, Ludwig W, Wagner M, Schleifer K-H (1992) Phylogenetic oligodeoxynucleotide probes for the major subclasses of Proteobacteria: problems and solutions. Syst Appl Microbiol 15:593–600
Mazzola M (2002) Mechanisms of natural soil suppressiveness to soilborne diseases. Antonie Van Leeuwenhoek 81:557–564
Mejri D, Gamalero E, Tombolini R, Musso C, Massa N, Berta G, Souissi T (2010) Biological control of great brome (Bromus diandrus) in durum wheat (Triticum durum): specificity, physiological traits and impact on plant growth and root architecture of the fluorescent pseudomonad strain X33d. BioControl 55:561–572
Minkwitz A, Berg G (2001) Comparison of antifungal activities and 16S ribosomal DNA sequences of clinical and environmental isolates of Stenotrophomonas maltophilia. J Clin Microbiol 39:139–145
Mo W, Li C, Zheng Y, Yue H, Li H (2006) Effect of relieving salt stress and growth-promoting bacteria on germination of cotton seed under salt stress. Nongye Gongcheng Xuebao/Trans Chin Soc Agric Eng 22:260–263
Mühling M, Woolven-Allen J, Colin Murrell J, Joint I (2008) Improved group-specific PCR primers for denaturing gradient gel electrophoresis analysis of the genetic diversity of complex microbial communities. ISME J 2:379–392
Müller H, Berg G (2008) Impact of formulation procedures on the effect of the biocontrol agent Serratia plymuthica HRO-C48 on Verticillium wilt in oilseed rape. Biocontrol 53:905–916
Müller H, Westendorf C, Leitner E, Chernin L, Riedel K, Schmidt S, Eberl L, Berg G (2009) Quorum-sensing effects in the antagonistic rhizosphere bacterium Serratia plymuthica HRO-C48. FEMS Microbiol Ecol 67:468–478
Nadeem SM, Zahir ZA, Naveed M, Asghar HN, Arshad M (2010) Rhizobacteria capable of producing ACC-deaminase may mitigate salt stress in wheat. Soil Sci Soc America J 74:533–542
Parker BC, Bold HC, Deason TR (1961) Facultative heterotrophy in some chlorococcacean algae. Science 133:761–763
Ribbeck-Busch K, Roder A, Hasse D, de Boer W, Martínez JL, Hagemann M, Berg G (2005) A molecular biological protocol to distinguish potentially human pathogenic Stenotrophomonas maltophilia from plant-associated Stenotrophomonas rhizophila. Environ Microbiol 7:1853–1858
Ryan RP, Fouhy Y, Garcia BF, Watt SA, Niehaus K, Yang L, Tolker-Nielsen J, Dow M (2008) Interspecies signalling via the Stenotrophomonas maltophilia diffusible signal factor influences biofilm formation and polymyxin tolerance in Pseudomonas aeruginosa. Mol Microbiol 68:75–86
Ryan RP, Monchy S, Cardinale M, Taghavi S, Crossman L, Avison MB, Berg G, van de Lelie D, Dow JM (2009) The versatility and adaptation of bacteria from the genus Stenotrophomonas. Nat Rev Microbiol 7:514–525
Scherwinski K, Grosch R, Berg G (2008) Effect of bacterial antagonists on lettuce: active biocontrol of Rhizoctonia solani and negligible, short-term effects on non target microorganisms. FEMS Microbiol Ecol 64:106–116
Schippers B, Bakker AW, Bakker PAHM (1987) Interactions of deleterious and beneficial rhizosphere microorganisms and the effect of cropping practices. Ann Rev Phytopathol 25:339–358
Schmidt CS, Lorenz D, Wolf GA (2001) Biological control of the grapevine dieback fungus Eutypa lata II: Influence of formulation additives and transposon mutagenesis on the antagonistic activity of Bacillus subtilis and Erwinia herbicola. J Phytopathol 149:437–445
Schwieger F, Tebbe CC (1998) A new approach to utilize PCR single strand-conformation polymorphism for 16S rRNA gene-based microbial community analysis. Appl Environ Microbiol 64:4870–4876
Shoup S, Lewis LA (2003) Polyphyletic origin of parallel basal bodies in swimming cells of Chlorophycean green algae (Chlorophyta). J Phycol 39:789–796
Suckstorff I, Berg G (2003) Evidence for dose-dependent effects on plant growth by Stenotrophomonas strains from different origin. J Appl Microbiol 95:656–663
Suslow (2004) Key points of control and management of microbial food safety: Information for producers, handlers and processors of fresh market tomatoes. Publication 8150, University of California Division of Agriculture and Natural Resources, Oakland, CA, USA.
Tanji KK, Kielen NC (2002) Agricultural drainage water management in arid and semi-arid areas. FAO irrigation and drainage paper 61. FAO (Food and Agriculture Organisation of the United Nations), Rome
Weller DM (2007) Pseudomonas biocontrol agents of soilborne pathogens: looking back over 30 years. Phytopathology 97:250–256
White TJ, Bruns TD, Lee S, Taylor J (1990) Analysis of phylogenetic relationship by amplification and direct sequencing of ribosomal RNA genes. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR protocols: a guide to methods and applications. Academic, New York, pp 315–322
Williams P (2007) Quorum sensing, communication and cross-kingdom signalling in the bacterial world. Microbiology 153:3923–3938
Wiyono S, Schulz D, Wolf GA (2008) Improvement of the formulation and antagonistic activity of Pseudomonas fluorescens B5 through selective additives in the pelleting process. Biol Control 46:348–357
Wolf A, Fritze A, Hagemann M, Berg G (2002) Stenotrophomonas rhizophila sp. nov., a novel plant-associated bacterium with antifungal properties. Int J Syst Evol Microbiol 52:1937–1944
Acknowledgments
We wish to thank Dilfuza Egamberdiyeva (Tashkent) for the provision of seeds, Michael Fürnkranz (Graz) for very valuable advices and Katja Drofenigg (Graz) for technical support. This study was funded by the Austrian Science Foundation FWF by a grant to G.B.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schmidt, C.S., Alavi, M., Cardinale, M. et al. Stenotrophomonas rhizophila DSM14405T promotes plant growth probably by altering fungal communities in the rhizosphere. Biol Fertil Soils 48, 947–960 (2012). https://doi.org/10.1007/s00374-012-0688-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00374-012-0688-z