Abstract
Understanding the mechanisms that link sensory stimuli to animal behavior is a central challenge in neuroscience. The quantitative description of behavioral responses to defined stimuli has led to a rich understanding of different behavioral strategies in many species. One important navigational cue perceived by many vertebrates and insects is the e-vector orientation of linearly polarized light. Drosophila manifests an innate orientation response to this cue (‘polarotaxis’), aligning its body axis with the e-vector field. We have established a population-based behavioral paradigm for the genetic dissection of neural circuits guiding polarotaxis to both celestial as well as reflected polarized stimuli. However, the behavioral mechanisms by which flies align with a linearly polarized stimulus remain unknown. Here, we present a detailed quantitative description of Drosophila polarotaxis, systematically measuring behavioral parameters that are modulated by the stimulus. We show that angular acceleration is modulated during alignment, and this single parameter may be sufficient for alignment. Furthermore, using monocular deprivation, we show that each eye is necessary for modulating turns in the ipsilateral direction. This analysis lays the foundation for understanding how neural circuits guide these important visual behaviors.





Similar content being viewed by others
References
Brunner D, Labhart T (1987) Behavioral evidence for polarization vision in crickets. Physiol Entomol 12:1–10. doi:10.1111/j.1365-3032.1987.tb00718.x
Clark DA, Bursztyn L, Horowitz MA, Schnitzer MJ, Clandinin TR (2011) Defining the computational structure of the motion detector in Drosophila. Neuron 70:1165–1177. doi:10.1016/j.neuron.2011.05.023
Fortini M, Rubin GM (1991) The optic lobe projection pattern of polarization-sensitive photoreceptor cells in Drosophila melanogaster. Cell Tissue Res 265:185–191. doi:10.1007/BF00318153
Hardie RC (1984) Properties of photoreceptors R7 and R8 in dorsal marginal ommatidia in the compound eyes of Musca and Calliphora. J Comp Physiol A 154:157–165. doi:10.1007/BF00604981
Hassenstein B, Reichardt W (1956) Systemtheoretische Analyse der Zeit-, Reihenfolgen- und Vorzeichenauswertung bei der Bewegungsperzeption des Rüsselkäfers Chlorophanus. Z Naturforsch 11:513–524
Heinze S, Homberg U (2007) Maplike representation of celestial e-vector orientations in the brain of an insect. Science 327:385–398. doi:10.1126/science.1135531
Homberg U, Heinze S, Pfeiffer K, Kinoshita M, el Jundi B (2011) Central neural coding of sky polarization in insects. Phil Trans R Soc Lond B Biol Sci 366:680–687. doi:10.1098/rstb.2010.0199
Horvath G, Majer J, Horvath L, Szivak I, Kriska G (2008) Ventral polarization vision in tabanids: horseflies and deerflies (Diptera: Tabanidae) are attracted to horizontally polarized light. Naturwissenschaften 95:1093–1100. doi:10.1007/s00114-008-0425-5
Kamermans M, Hawryshyn C (2011) Teleost polarization vision: how it might work and what it might be good for. Phil Trans R Soc Lond B Biol Sci 366:742–756. doi:10.1098/rstb.2010.0211
Katsov AY, Clandinin TR (2008) Motion processing streams in Drosophila are behaviorally specialized. Neuron 59:322–335. doi:10.1016/j.neuron.2008.05.022
Labhart T (1980) Specialized photoreceptors at the dorsal rim of the retina of the honeybee’s compound eye: polarization and angular sensitivity. J Comp Physiol A 141:19–30. doi:10.1007/BF00611874
Labhart T (1986a) The electrophysiology of photoreceptors in different eye regions of the desert ant, Cataglyphis-bicolor. J Comp Physiol A 158:1–7. doi:10.1007/BF00614514
Labhart T (1986b) How polarization-sensitive interneurons of crickets perform at low degrees of polarization. J Exp Biol 199:1467–1475
Labhart T (1988) Polarization-opponent interneurons in the insect visual system. Nature 331:435–437. doi:10.1038/331435a0
Labhart T, Meyer EP (1999) Detectors for polarized skylight in insects: a survey of ommatidial specializations in the dorsal rim area of the compound eye. Microsc Res Tech 47:368–379. doi:10.1002/(SICI)1097-0029(19991215)47:6<368:AID-JEMT2>3.0.CO;2-Q
Labhart T, Hodel B, Valenzuela I (1984) The physiology of the cricket’s compound eye with particular reference to the anatomically specialized dorsal rim area. J Comp Physiol A 155:289–296. doi:10.1007/BF00610582
Maisak MS, Haag J, Ammer G, Serbe E, Meier M, Leonhardt A, Schilling T, Bahl A, Rubin GM, Nern A, Dickson BJ, Reiff DF, Hopp E, Borst A (2013) A directional tuning map of Drosophila elementary motion detectors. Nature 500:212–216. doi:10.1038/nature12320
Marshall J, Cronin TW, Kleinlogel S (2007) Stomatopod eye structure and function: a review. Arthropod Struct Dev 36:420–448. doi:10.1016/j.asd.2007.01.006
Meyer EP, Domanico V (1999) Microvillar orientation in the photoreceptors of the ant Cataglyphis bicolor. Cell Tissue Res 295:355–361. doi:10.1007/s004410051242
Reppert SM, Zhu H, White RH (2004) Polarized light helps monarch butterflies navigate. Curr Biol 14:155–158. doi:10.1016/j.cub.2003.12.034
Rossel S, Wehner R (1986) Polarization vision in bees. Nature 323:128–131. doi:10.1038/323128a0
Sakura M, Lambrinos D, Labhart T (2008) Polarized skylight navigation in insects: model and electrophysiology of e-vector coding by neurons in the central complex. J Neurophysiol 99:667–682. doi:10.1152/jn.00784.2007
Schwind R (1983) A polarization-sensitive response of the flying water bug Notonecta glauca to UV light. J Comp Physiol A 150:87–91. doi:10.1007/BF00605291
Shashar N, Sabbah S, Aharoni N (2005) Migrating locusts can detect polarized reflections to avoid flying over the sea. Biol Lett 1:472–475. doi:10.1098/rsbl.2005.0334
Silies M, Gohl DM, Fisher YE, Freifeld L, Clark DA, Clandinin TR (2013) Modular use of peripheral input channels tunes motion-detecting circuitry. Neuron 79:111–127. doi:10.1016/j.neuron.2013.04.029
Straw AD, Branson K, Neuman TR, Dickenson MR (2011) Multi-camera real-time three-dimensional tracking of multiple flying animals. J R Soc Interface 8:395–409. doi:10.1098/rsif.2010.0230
Takemura SY, Bharioke A, Lu Z, Nern A, Vitaladevuni S, Rivlin PK, Katz WT, Olbris DJ, Plaza SM, Winston P, Zhao T, Horne JA, Fetter RD, Takemura S, Blazek K, Chang LA, Ogundeyi O, Saunders MA, Shapiro V, Sigmund C, Rubin GM, Scheffer LK, Meinertzhagen IA, Chklovskii DB (2013) A visual motion detection circuit suggested by Drosophila connectomics. Nature 500:175–181. doi:10.1038/nature12450
Tuthill JC, Nern A, Holtz SL, Rubin GM, Reiser MB (2013) Contributions of the 12 neuron classes in the fly lamina to motion vision. Neuron 79:128–140. doi:10.1016/j.neuron.2013.05.024
Von Frisch K (1949) Die Polarisation des Himmelslichtes als orientierender Faktor bei den Tänzen der Bienen. Experientia 5:142–148. doi:10.1007/BF02174424
Von Philipsborn A, Labhart T (1990) A behavioral-study of polarization vision in the fly, musca-domestica. J Comp Physiol A 167:737–743. doi:10.1007/BF00189764
Vowles DM (1950) Sensitivity of ants to polarized light. Nature 165:282–283. doi:10.1038/165282a0
Wada S (1974) Spezielle randzonale Ommatidien der Fliegen (Diptera: Brachycera): Architektur und Verteilung in den Komplexaugen. Z Morph Tiere 77:87–125. doi:10.1007/BF00374212
Wehner R (2001) Polarization vision—a uniform sensory capacity? J Exp Biol 204:2589–2596
Wehner R (2003) Desert ant navigation: how miniature brains solve complex tasks. J Comp Physiol A 189:579–588. doi:10.1007/s00359-003-0431-1
Wehner R, Müller M (1985) Does intraocular transfer occur in visual navigation by ants? Nature 315:228–229. doi:10.1038/315228a0
Weir PT, Dickinson MH (2012) Flying Drosophila orient to sky polarization. Curr Biol 22:21–27. doi:10.1016/j.cub.2011.11.026
Wernet MF, Labhart T, Baumann F, Mazzoni EO, Pichaud F, Desplan C (2003) Homothorax switches function of Drosophila photoreceptors from color to polarized light sensors. Cell 115:267–279. doi:10.1016/S0092-8674(03)00848-1
Wernet MF, Velez MM, Clark DA, Baumann-Klausener F, Brown JR, Klovstad M, Clandinin TR (2012) Genetic dissection reveals two separate retinal substrates for polarization vision in Drosophila. Curr Biol 22:12–20. doi:10.1016/j.cub.2011.11.028
Wildermuth H (1998) Dragonflies recognize the water of rendezvous and oviposition sites by horizontally polarized light: a behavioral field test. Naturwissenschaften 85:297–302. doi:10.1007/s001140050504
Wiltschko W, Wiltschko R (2012) Global navigation in migratory birds: tracks, strategies, and interactions between mechanisms. Curr Opin Neurobiol 22:328–335. doi:10.1016/j.conb.2011.12.012
Wolf R, Gebhardt B, Gademann R, Heisenberg M (1980) Polarization sensitivity of course control in Drosophila melanogaster. J Comp Physiol A 139:177–191. doi:10.1007/BF00657080
Wunderer H, Smola U (1982) Fine-structure of ommatidia at the dorsal eye margin of Calliphora-Erythrocephala Meigen (Diptera, Calliphoridae)—an eye region specialized for the detection of polarized-light. Int J Insect Morphol Embryol 11:25–38. doi:10.1016/0020-7322(82)90035-6
Yonehara K, Roska B (2013) Motion detection: neuronal circuit meets theory. Cell 154:1188–1189. doi:10.1016/j.cell.2013.08.027
Zhu Y, Nern A, Zipursky SL, Frye MA (2009) Peripheral visual circuits functionally segregate motion and phototaxis behaviors in the fly. Curr Biol 19:613–619. doi:10.1016/j.cub.2009.02.053
Zufall F, Schmitt M, Menzel R (1989) Spectral and polarized-light sensitivity of photoreceptors in the compound eye of the cricket (Gryllus-Bimaculatus). J Comp Physiol A 164:597–608. doi:10.1007/BF00614502
Acknowledgments
The authors thank Thomas Labhart, Bob Schneeveis, and David Profitt for technical assistance. This work was supported by the Helen Hay Whitney Foundation (MFW), the Jane Coffin Childs Foundation (DAC), a Ruth L. Kirschstein Graduate Fellowship Award (MMV), and by an NIH Director’s Pioneer Award (DP1 OD003530) to TRC. This work was also supported by a Burroughs-Wellcome Career Development Award (TRC), a Mcknight Scholar Award (TRC), and Klingenstein Fellowship (TRC), and a Searle Scholar Award (TRC).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
359_2014_910_MOESM1_ESM.tif
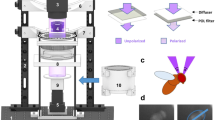
Supplemental Fig S1. Experimental setup and procedures. a Schematic of the experimental setup used to present linearly polarized UV light from above to populations of Drosophila, which are illuminated with IR light (red), and are filmed from below (camera). A polarization filter (Polarizer) is facing the flies, with a diffuser facing the light source. IR = infrared light. UV = polarized UV light. b Summary of the stimulus protocol used. Top: A computer-controlled servomotor rotated the polarization filter in 45° increments, remaining still for 5 s at each position. Different motor positions are shown. Below: Polar histograms of fly angular headings are shown for each motor position, and indicate that flies align their body axis with the orientation of the e-vector (double-headed arrows) of linearly polarized light. The orientation of incident e-vectors is symbolized using double-headed arrows. (TIFF 1502 kb)
359_2014_910_MOESM2_ESM.tif
Supplemental Fig S2. Ventral behavior is not mediated by reflections. Series of controls confirming that ventral polarotaxis by flies walking upside down on the ceiling of the arena is not the result of POL stimuli being reflected off the arena floor. A series of modifications was added to the experimental setup (from left to right): the arena floor plate was tilted (either at 30° or 45°), was sanded, replaced by a non-reflecting surface), or by a nylon mesh. In all cases, polarotactic alignment behavior persisted, demonstrating that flies perceived the stimulus directly with their ventral eye. (TIFF 1291 kb)
Rights and permissions
About this article
Cite this article
Velez, M.M., Wernet, M.F., Clark, D.A. et al. Walking Drosophila align with the e-vector of linearly polarized light through directed modulation of angular acceleration. J Comp Physiol A 200, 603–614 (2014). https://doi.org/10.1007/s00359-014-0910-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00359-014-0910-6