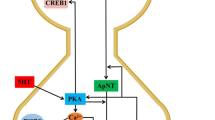
Abstract
Previously, a cannabinoid-dependent form of long-term depression (LTD) was discovered at the polysynaptic connection between the touch mechanosensory neuron and the S interneuron (Li and Burrell in J Comp Physiol A 195:831–841, 2009). In the present study, the physiological properties of this cannabinoid-dependent LTD were examined. Increases in intracellular calcium in the S interneuron are necessary for this form of LTD in this circuit. Calcium signals contributing to cannabinoid-dependent LTD are mediated by voltage-dependent calcium channel and release of calcium from intracellular stores. Inositol triphosphate receptors, but not ryanodine receptors, appear to mediate this store-released calcium signal. Cannabinoid-dependent LTD also requires activation of metabotropic serotonin receptors, possibly a serotonin type 2-like receptor. Finally, this form of LTD involves the stimulation of nitric oxide synthase and a decrease in cyclic adenosine monophosphate signaling, both of which appeared to be downstream of cannabinoid receptor activation. Based on these findings, the cellular signaling mechanisms of cannabinoid-dependent LTD in the leech are remarkably similar to vertebrate forms of cannabinoid-dependent synaptic plasticity.





Similar content being viewed by others
Abbreviations
- α-Me-5-HT:
-
Alpha-methyl-5-hydroxytryptamine
- 2-AG:
-
2-Arachidonyl glycerol
- AMPA:
-
α-Amino-3-hydroxy-5-methylisoxazole-4-propionic acid
- AM251:
-
1-(2,4-Dichlorophenyl)-5-(4-iodophenyl)-4-methyl-N-1-piperidinyl-1H-pyrazole-3-carboxamide
- ANOVA:
-
Analysis of variance
- AP5:
-
2-Amino-5-phosphonopentanoic acid
- BAPTA:
-
2-Bis(o-aminophenoxy)ethane-N,N,N′,N′-tetraacetic acid
- Ca2+ :
-
Calcium
- cAMP:
-
Cyclic adenosine monophosphate
- CNS:
-
Central nervous system
- DMSO:
-
Dimethyl sulfoxide
- d-NAME:
-
NG-nitro-d-arginine methyl ester
- EGTA:
-
Ethylene glycol tetraacetic acid
- EPSP:
-
Excitatory post-synaptic potential
- 5HT:
-
5-Hydroxytryptamine (serotonin)
- 5-HT2 R:
-
5-Hydroxytryptamine (serotonin) type 2 receptor
- IP3 :
-
Inositol triphosphate
- KAc :
-
Potassium acetate
- LFS:
-
Low frequency stimulation
- l-NAME:
-
NG-nitro-l-arginine methyl ester
- LTD:
-
Long-term depression
- LTP:
-
Long-term potentiation
- MCPG:
-
Methyl-4-carboxyphenylglycine
- MK801:
-
(5R,10S)-(+)-5-methyl-10,11-dihydro-5H-dibenzo[a,d]cyclohepten-5,10-imine
- Ni2+ :
-
Nickel
- NMDAR:
-
N-methyl-d-aspartic acid receptor
- VDCC:
-
Voltage-dependent Ca2+ channels
References
Alger BE (2002) Retrograde signaling in the regulation of synaptic transmission: focus on endocannabinoids. Prog Neurobiol 68:247–268
Angstadt JD, Grassmann JL, Theriault KM, Levasseur SM (2005) Mechanisms of postinhibitory rebound and its modulation by serotonin in excitatory swim motor neurons of the medicinal leech. J Comp Physiol A 191:715–732
Barbas D, Campbell A, Castellucci VF, DesGroseillers L (2005) Comparative localization of two serotonin receptors and sensorin in the central nervous system of Aplysia californica. J Comp Neurol 490:295–304
Bender VA, Bender KJ, Brasier DJ, Feldman DE (2006) Two coincidence detectors for spike timing-dependent plasticity in somatosensory cortex. J Neurosci 26:4166–4177
Best AR, Regehr WG (2008) Serotonin evokes endocannabinoid release and retrogradely suppresses excitatory synapses. J Neurosci 28:6508–6515
Burke RD, Angerer LM, Elphick MR, Humphrey GW, Yaguchi S, Kiyama T, Liang S, Mu X, Agca C, Klein WH, Brandhorst BP, Rowe M, Wilson K, Churcher AM, Taylor JS, Chen N, Murray G, Wang D, Mellott D, Olinski R, Hallböök F, Thorndyke MC (2006) A genomic view of the sea urchin nervous system. Dev Biol 300:434–460
Burrell BD, Li Q (2008) Co-induction of long-term potentiation and long-term depression at a central synapse in the leech. Neurobiol Learn Mem 90:275–279
Burrell BD, Sahley CL (2004) Multiple forms of long-term potentiation and long-term depression converge on a single interneuron in the leech CNS. J Neurosci 24:4011–4019
Burrell BD, Sahley CL (2005) Serotonin mediates learning-induced potentiation of excitability. J Neurophysiol 94:4002–4010
Burrell BD, Sahley CL, Muller KJ (2001) Non-associative learning and serotonin induce similar bi-directional changes in excitability of a neuron critical for learning in the medicinal leech. J Neurosci 21:1401–1412
Burrell BD, Sahley CL, Muller KJ (2003) Progressive recovery of learning during regeneration of a single synapse in the medicinal leech. J Comp Neurol 457:67–74
Calviño MA, Szczupak L (2008) Spatial-specific action of serotonin within the leech midbody ganglion. J Comp Physiol A 194:523–531
Cercós MG, De-Miguel FF, Trueta C (2009) Real-time measurements of synaptic autoinhibition produced by serotonin release in cultured leech neurons. J Neurophysiol 102:1075–1085
Chevaleyre V, Takahashi KA, Castillo PE (2006) Endocannabinoid-mediated synaptic plasticity in the CNS. Annu Rev Neurosci 29:37–76
Chevaleyre V, Heifets BD, Kaeser PS, Südhof TC, Castillo PE (2007) Endocannabinoid-mediated long-term plasticity requires cAMP/PKA signaling and RIM1alpha. Neuron 54:801–812
Cohen JE, Onyike CU, McElroy VL, Lin AH, Abrams TW (2003) Pharmacological characterization of an adenyl cyclase-coupled 5-HT receptor in Aplysia: comparison with mammalian 5-HT receptors. J Neurophysiol 89:1440–1455
Connelly WM, Baggott MJ (2009) Role of endocannabinoids in 5-HT2 receptor-mediated effects. J Neurophysiol 101:5–7
Crisp KM, Mesce KA (2006) Beyond the central pattern generator: amine modulation of decision-making neural pathways descending from the brain of the medicinal leech. J Exp Biol 209:1746–1756
Crisp KM, Muller KJ (2006) A 3-synapse positive feedback loop regulates the excitability of an interneuron critical for sensitization in the leech. J Neurosci 26:3524–3531
De Petrocellis L, Melck D, Bisogno T, Milone A, Di Marzo V (1999) Finding of the encocannabinoid signaling system in Hydra, a very primitive organism: possible role in the feeding response. Neuroscience 92:377–387
Dernovici S, Starc T, Dent JA, Ribeiro P (2007) The serotonin receptor SER-1 (5-HT2ce) contributes to the regulation of locomotion in Caenorhabditis elegans. Dev Neurobiol 67:189–204
Egertova M, Elphick MR (2007) Localization of CiCBR in the invertebrate chordate Ciona intestinalis: evidence of an ancient role for cannabinoid receptors as axonal regulators of neuronal signaling. J Comp Neurol 502:660–672
Elphick MR, Egertova M (2005) The phylogenetic distribution and evolutionary origins of endocannabinoid signaling. Handb Exp Pharmacol 168:283–297
Freund TF, Katona I, Piomelli D (2003) Retrograde signaling in the regulation of synaptic transmission: focus on endocannabinoids. Physiol Rev 83:1017–1066
Gaudry Q, Kristan WB Jr (2009) Behavioral choice by presynaptic inhibition of tactile sensory terminals. Nat Neurosci 12:1450–1457
Gerdeman GL, Ronesi J, Lovinger DM (2002) Postsynaptic endocannabinoid release is critical to long-term depression in the striatum. Nat Neurosci 5:446–451
Giuffrida A, Parsons LH, Kerr TM, Rodríguez de Fonseca F, Navarro M, Piomelli D (1999) Dopamine activation of endogenous cannabinoid signaling in dorsal striatum. Nat Neurosci 2:358–363
Glanzman DL (2010) Common mechanisms of synaptic plasticity in vertebrates and invertebrates. Curr Biol 20:R31–R36
Grey KB, Burrell BD (2010) Co-induction of LTP and LTD and its regulation by protein kinases and phosphatases. J Neurophysiol 103:2737–3746
Hillard CJ, Muthian S, Kearn CS (1999) Effects of CB(1) cannabinoid receptor activation on cerebellar granule cell nitric oxide synthase activity. FEBS Lett 459:277–281
Isokawa M, Alger BE (2005) Retrograde endocannabinoid regulation of GABAergic inhibition in the rat dentate gyrus granule cell. J Physiol 567:1001–1010
Ivanov AI, Calabrese RL (2006) Graded inhibitory synaptic transmission between leech interneurons: assessing the roles of two kinetically distinct low-threshold Ca currents. J Neurophysiol 96:218–234
Jeon YJ, Yang KH, Pulaski JT, Kaminski NE (1996) Attenuation of inducible nitric oxide synthase gene expression by delta 9-tetrahydrocannabinol is mediated through the inhibition of nuclear factor-kappa B/Rel activation. Mol Pharmacol 50:334–341
Kim J, Isokawa M, Ledent C, Alger BE (2002) Activation of muscarinic acetylcholine receptors enhances the release of endogenous cannabinoids in the hippocampus. J Neurosci 22:10182–10191
Kiss T, Hiripi L, Papp N, Elekes K (2003) Dopamine and serotonin receptors mediating contractions of the snail, Helix pomatia, salivary duct. Neuroscience 116:775–790
Kristan WB Jr, Calabrese RL, Friesen WO (2005) Neuronal control of leech behavior. Prog Neurobiol 76:279–327
Lemak MS, Bravarenko NI, Bobrov MY, Bezuglov VV, Ierusalimsky VN, Storozhuk MV, Malyshev AY, Balaban PM (2007) Cannabinoid regulation in identified synapse of terrestrial snail. Eur J Neurosci 26:3207–3214
Leung HT, Tseng-Crank J, Kim E, Mahapatra C, Shino S, Zhou Y, An L, Doerge RW, Pak WL (2008) DAG lipase activity is necessary for TRP channel regulation in drosophila photoreceptors. Neuron 58:884–896
Li Q, Burrell BD (2009) Two forms of long-term depression in a polysynaptic pathway in the leech CNS: one NMDA receptor-dependent and the other cannabinoid-dependent. J Comp Physiol A 195:831–841
Lohr C, Deitmer JW (1997) Intracellular Ca2+ release mediated by metabotropic glutamate receptor activation in the leech giant glial cell. J Exp Biol 200:2565–2573
Lu FM, Hawkins RD (2006) Presynaptic and postsynaptic Ca and CamKII contribute to long-term potentiation at synapses between individual CA3 neurons. Proc Natl Acad Sci USA 103:4262–4269
Lu J, Dalton JF 4th, Stokes DR, Calabrese RL (1997) Functional role of Ca2+ currents in graded and spike-mediated synaptic transmission between leech heart interneurons. J Neurophysiol 77:1779–1794
Maejima T, Hashimoto K, Yoshida T, Aiba A, Kano M (2001) Presynaptic inhibition caused by retrograde signal from metabotropic glutamate to cannabinoid receptors. Neuron 31:463–475
Matias I, Bisogno T, Melck D, Vandenbulcke F, Verger-Bocquet M, De Petrocellis L, Sergheraert C, Breton C, Di Marzo V, Salzet M (2001) Evidence for an endocannabinoid system in the central nervous system of the leech Hirudo medicinalis. Brain Res Mol Brain Res 87:145–159
McPartland JM (2004) Phylogenomic and chemotaxonomic analysis of the endocannabinoid system. Brain Res Rev 45:18–29
McPartland JM, Glass M (2003) Functional mapping of cannabinoid receptor homologs in mammals, other vertebrates and invertebrates. Gene 312:297–303
McPartland JM, Di Marzo V, De Petrocellis L, Mercer A, Glass M (2001) Cannabinoid receptors are absent in insects. J Comp Neurol 436:423–429
McPartland JM, Agraval J, Gleeson D, Heasman K, Glass M (2006) Cannabinoid receptors in invertebrates. J Evol Biol 19:366–373
Modney BK, Sahley CL, Muller KJ (1997) Regeneration of a central synapse restores nonassociative learning. J Neurosci 17:6478–6482
Moss BL, Fuller AD, Sahley CL, Burrell BD (2005) Serotonin modulates axo-axonal coupling between neurons critical for learning in the leech. J Neurophysiol 94:2575–2589
Muller KJ, Scott SA (1981) Transmission at a ‘direct’ electrical connexion mediated by an interneurone in the leech. J Physiol 311:565–583
Ohno-Shosaku T, Matsui M, Fukudome Y, Shosaku J, Tsubokawa H, Taketo MM, Manabe T, Kano M (2003) Postsynaptic M1 and M3 receptors are responsible for the muscarinic enhancement of retrograde endocannabinoid signaling in the hippocampus. Eur J Neurosci 18:109–116
Parrish JC, Nichols DE (2006) Serotonin 5-HT(2A) receptor activation induces 2-arachidonoylglycerol release through a phospholipase c-dependent mechanism. J Neurochem 99:1164–1175
Penzo MA, Peña JL (2009) Endocannabinoid-mediated long-term depression in the avian midbrain expressed presynaptically and postsynaptically. J Neurosci 29:4131–4139
Rawls SM, Gomez T, Raffa RB (2007) An NMDA antagonist (LY 235959) attenuates abstinence-induced withdrawal of planarians following acute exposure to a cannabinoid agonist (WIN 55212–2). Pharmacol Biochem Behav 86:499–504
Robbe D, Kopf M, Remaury A, Bockaert J, Manzoni OJ (2002) Endogenous cannabinoids mediate long-term synaptic depression in the nucleus accumbens. Proc Natl Acad Sci USA 99:8348–8384
Sahley CL, Modney BK, Boulis NM, Muller KJ (1994) The S cell: an interneuron essential for sensitization and full dishabituation of leech shortening. J Neurosci 14:6715–6721
Salzet M, Stefano GB (2002) The endocannabinoid system in invertebrates. Prostaglandins Leukot Essent Fatty Acids 66:353–361
Salzet M, Breton C, Bisogno T, Di Marzo V (2000) Comparative biology of the endocannabinoid system possible role in the immune response. Eur J Biochem 267:4917–4927
Silverman-Gavrila LB, Lew RR (2002) An IP3-activated Ca2+ channel regulates fungal tip growth. J Cell Sci 115:5013–5025
Sjostrom PJ, Turrigiano GG, Nelson SB (2003) Neocortical LTD via coincident activation of presynaptic NMDA and cannabinoid receptors. Neuron 39:641–654
Soler-Llavina GJ, Sabatini BL (2006) Synapse-specific plasticity and compartmentalized signaling in cerebellar stellate cells. Nat Neurosci 9:798–806
Trueta C, Sánchez-Armass S, Morales MA, De-Miguel FF (2004) Calcium-induced calcium release contributes to somatic secretion of serotonin in leech Retzius neurons. J Neurobiol 61:309–316
Varma N, Carlson GC, Ledent C, Alger BE (2001) Metabotropic glutamate receptors drive the endocannabinoid system in hippocampus. J Neurosci 21:RC188
Watts SW, Cohen ML, Mooney PQ, Johnson BG, Schoepp DD, Baez M (1994) Disruption of potential alpha-helix in the G loop of the guinea pig 5-hydroxytryptamine2 receptor does not prevent receptor coupling to phosphoinositide hydrolysis. Neurochemistry 62:934–943
Acknowledgments
The authors thank Drs. Brenda Moss and Kathryn Grey for their helpful comments during the preparation of this manuscript. Supported by grants from the South Dakota Spinal Cord/Traumatic Brain Injury Research Council (BDB) and by a subproject of the National Institutes of Health Center of Biomedical Research Excellence (NIH/COBRE) grant (P20 RR015567).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplemental Figure 1
: LFS (1Hz, 900 s) induced depression in the electrical component of T-to-S synaptic connection and this depression was blocked by EGTA injection into the S cell (one-way ANOVA, F2, 17 = 40.9, P < 0.001, Newman Keuls post-hoc test between EGTA + LFS group and saline LFS group, P <0.001). EGTA treatment without LFS did not alter the electrical EPSP (Post-hoc Newman Keuls test between EGTA + LFS group and EGTA without LFS group, P > 0.05) (EPS 2194 kb)
Supplemental Figure 2
: The role of intracellular and extracellular Ca2+ sources on depression of the electrical EPSP (a) Ni2+ (1 mM) prevented the LTD following LFS (one-way ANOVA of the Ni2+ + LFS and Saline + LFS groups, F1,8 = 29.13, P < 0.001), but did not affect basal synaptic transmission (n = 5 in each group; one-way ANOVA for no LFS groups, F1,8 = 2.74, P > 0.05). (b) Similar to its effect on chemical synaptic transmission, thapsigargin (1 µM) blocked LTD of the electrical EPSP (One-way ANOVA, F2, 12 = 4.63, P < 0.05; post-hoc Newman Keuls test between Thapsigargin + LFS and DMSO + LFS group, P < 0.05). Both TMB-8 (50 µM) and ryanodine (50 µM) also prevented LTD of the electrical synapse (post-hoc Newman Keuls test, Ryanodine + LFS and DMSO + LFS group P < 0.05; TMB-8 + LFS and DMSO + LFS group P < 0.05) (EPS 2303 kb)
Supplemental Figure 3
: The effect of 5-HT and metabotropic glutamate receptor antagonists on depression of the electrical EPSP. Depression was blocked by the 5-HT receptor antagonists methysergide and ritanserin, but not affected by the metabotropic glutamate receptor antagonist MCPG (one-way ANOVA of LFS groups, F3, 18 = 14.83, P < 0.001; post-hoc Newman Keuls test between Saline + LFS and MCPG + LFS groups, P > 0.05; post-hoc Newman Keuls test between Saline + LFS and Methysergide + LFS group, P < 0.001; post-hoc Newman Keuls test between Saline + LFS and Ritanserin + LFS group, P < 0.01) (EPS 2230 kb)
Supplemental Figure 4
: The effect of nitric oxide synthase inhibitor on depression of the electrical EPSP. Both D-NAME and L-NAME bath application had no affect on T-to-S electrical transmission when LFS was omitted. When L-NAME and LFS were combined a partial reduction in LTD of the electrical synapse was observed, but this effect was not statistically significant (one-way ANOVA of the L-NAME + LFS and D-NAME + LFS groups, F1,8 = 1.65, P > 0.05). However, L-NAME did block depression of the electrical EPSP following CP55 940 treatment (one-way ANOVA of the CP55 940 + L-NAME and CP55 940 + D-NAME groups, F1, 6 = 8.30, P < 0.05) (EPS 2206 kb)
Supplemental Figure 5
: The effect of cAMP phosphodiesterase inhibitor on depression of the electrical EPSP. Rolipram (0.1 µM) prevented LTD of the electrical EPSP following LFS (one-way ANOVA of the Rolipram + LFS and DMSO + LFS groups, F1, 8 = 35.12, P < 0.001). Rolipram also blocked LTD of the electrical EPSP following CP55 940 treatment (one-way ANOVA of the CP55 940 and CP55 940 + Rolipram groups, F1, 10 = 7.71, P < 0.05) (EPS 2215 kb)
Rights and permissions
About this article
Cite this article
Li, Q., Burrell, B.D. Properties of cannabinoid-dependent long-term depression in the leech. J Comp Physiol A 196, 841–851 (2010). https://doi.org/10.1007/s00359-010-0566-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00359-010-0566-9