Abstract
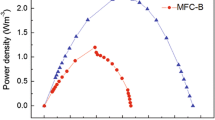
This study assessed cell voltage development, electricity recovery, and microbial community composition in response to initial substrate including acetate, xylose, acetate/xylose 1:1 mixture (ace/xyl), and bioethanol effluent (BE) during microbial fuel cell (MFC) operation at 1000 Ω external resistance. The BE mainly contained 20.5 g/L xylose, 1.8 g/L arabinose, and 2.5 g/L propionic acid. The MFCs initially fed with acetate showed shorter initiation time (1 day), higher average cell voltage (634 ± 9 mV), and higher coulombic efficiency (31.5 ± 0.5 %) than those initially fed with ace/xyl or xylose. However, BE-initiated MFCs only generated 162 ± 1 mV. The acetate-initiated MFCs exhibited longer adaptation time (21 h) and lower cell voltage (645 ± 10 mV) when the substrate was switched to xylose, whereas substrate switching to BE produced the highest voltage (656 mV), maximum power density (362 ± 27 mW/m2), maximum current density (709 ± 27 mA/m2), and coulombic efficiency (25 ± 0.5 %) in the acetate-initiated MFCs. The microbial community in acetate-initiated MFCs was less diverse and contained more electrogenic bacteria (13.9 ± 0.4 %) including Geobacter sulfurreducens and Desulfuromonas acetexigen than the MFCs initially fed with ace/xyl, xylose, and BE. After switching the substrate to xylose and subsequently to BE, the microbial community in the acetate-initiated MFCs became more diverse, while no significant changes were observed in ace/xyl-, xylose-, and BE-initiated MFCs. The results showed that initial substrate affected the power generation and the capability to adapt to the substrate alteration in MFCs. Acetate-initiated MFCs showed best performance in utilizing BE.







Similar content being viewed by others
References
Ambye-Jensen M, Thomsen ST, Kádár Z, Meyer AS (2013) Ensiling of wheat straw decreases the required temperature in hydrothermal pretreatment. Biotechnol Biofuels 6:116
Bond DR, Lovley DR (2003) Electricity production by Geobacter sulfurreducens attached to electrodes. Appl Environ Microbiol 69:1548–1555
Chae KJ, Choi MJ, Lee JW, Kim KY, Kim IS (2009) Effect of different substrates on the performance, bacterial diversity, and bacterial viability in microbial fuel cells. Bioresour Technol 100:3518–3525
Ezeji T, Qureshi N, Blaschek HP (2007) Butanol production from agricultural residues: impact of degradation products on Clostridium beijerinckii growth and butanol fermentation. Biotechnol Bioeng 97:1460–1469
Fan Y, Sharbrough E, Liu H (2008) Quantification of the internal resistance distribution of microbial fuel Cells. Environ Sci Technol 42:8101–8107
Feng Y, Wang X, Logan BE, Lee H (2008) Brewery wastewater treatment using air-cathode microbial fuel cells. Appl Microbiol Biotechnol 78:873–880
Finster K, Bak F, Pfennig N (1994) Desulfuromonas acetexigens sp. nov., a dissimilatory sulfur-reducing eubacterium from anoxic freshwater sediments. Arch Microbiol 161:328–332
Futamata H, Bretschger O, Cheung A, Kan J, Owen R, Nealson HN (2013) Adaptation of soil microbes during establishment of microbial fuel cell consortium fed with lactate. J Biosci Bioeng 115:58–63
He Z, Mansfeld F (2009) Exploring the use of electrochemical impedance spectroscopy (EIS) in microbial fuel cell studies. Energy Environ Sci 2:215–219
Huang L, Angelidaki I (2008) Effect of humic acids on electricity generation integrated with xylose degradation in microbial fuel cells. Biotechnol Bioeng 100:413–422
Huang L, Zeng RJ, Angelidaki I (2008) Electricity production from xylose using a mediator-less microbial fuel cell. Bioresour Technol 99:4178–4184
Hutchinson AJ, Tokash JC, Logan BE (2011) Analysis of carbon fiber brush loading in anodes on startup and performance of microbial fuel cells. J Power Sour 196:9213–9219
Ishii S, Shimoyama T, Hotta Y, Watanabe K (2008) Characterization of a filamentous biofilm community established in a cellulose-fed microbial fuel cell. BMC Microbiol 8:6
Katz E, Willner I (2003) Probing biomolecular interactions at conductive and semiconductive surfaces by impedance spectroscopy: routes to impedimetric immunosensors, DNA-sensors, and enzyme biosensors. Electroanalysis 15:913–947
Ketep SF, Bergel A, Bertrand M, Achouak W, Fourest E (2013) Lowering the applied potential during successive scratching/re-inoculation improves the performance of microbial anodes for microbial fuel cells. Bioresour Technol 127:448–455
Liu Y, Harnisch F, Fricke K, Sietmann R, Schröder U (2008) Improvement of the anodic bioelectrocatalytic activity of mixed culture biofilms by a simple consecutive electrochemical selection procedure. Biosens Bioelectron 24:1012–1017
Lovley DR (2006) Bug juice: harvesting electricity with microorganisms. Nat Rev Microbiol 4:497–508
Lovley DR (2008) The microbe electric: conversion of organic matter to electricity. Curr Opin Biotechnol 19:564–571
Lovley DR, Phillips EJP (1988) Novel mode of microbial energy metabolism : organic carbon oxidation coupled to dissimilatory reduction of iron or manganese. Appl Environ Microbiol 54:1472–1480
Mäkinen AE, Lay C-H, Nissilä ME, Puhakka JA (2013) Bioelectricity production on xylose with a compost enrichment culture. Int J Hydrogen Energy 38:15606–15612
Mansfeld F (2003) The use of electrochemical techniques for the investigation and monitoring of microbiologically influenced corrosion and its inhibition – a review. Mater Corros 54:489–502
Marzorati M, Wittebolle L, Boon N, Daffonchio D, Verstraete W (2008) How to get more out of molecular fingerprints: practical tools for microbial ecology. Environ Microbiol 10:1571–1581
Muyzer G, Waal ECD, Uitterlinden AG (1993) Profiling of complex microbial populations by denaturing gradient gel electrophoresis analysis of polymerase chain reaction-amplified genes coding for 16S rRNA. Appl Environ Microbiol 59:695–700
Nevin KP, Richter H, Covalla SF, Johnosen JP, Woodard TL, Orloff AL, Jia H, Zhang M, Lovely DR (2008) Power output and columbic efficiencies from biofilms of Geobacter sulfurreducens comparable to mixed community microbial fuel cells. Environ Microbiol 10:2505–2514
Petrik S, Kádár Z, Márová I (2013) Utilization of hydrothermally pretreated wheat straw for production of bioethanol and carotene-enriched biomass. Bioresour Technol 133:370–377
Rabaey K, Lissens G, Siciliano SD, Verstraete W (2003) A microbial fuel cell capable of converting glucose to electricity at high rate and efficiency. Biotechnol Lett 25:1531–1535
Rabaey K, Ossieur W, Verhaege M, Verstraete W (2005) Continuous microbial fuel cells convert carbohydrates to electricity. Water Sci Technol 52:515–523
Read ST, Dutta P, Bond PL, Keller J, Rabaey K (2010) Initial development and structure of biofilms on microbial fuel cell anodes. BMC Microbiol 10:98
Rezaei F, Xing D, Wagner R, Regan JM, Richard TL, Logan BE (2009) Simultaneous cellulose degradation and electricity production by Enterobacter cloacae in a microbial fuel cell. Appl Environ Microbiol 75:3673–3678
Rice EW, Baird RB, Eaton AD, Clesceri LS (2012) Standard methods for the examination of water and wastewater. 22nd ed. Washington DC, USA. APHA, AWA, WEF. 2540C
Rice EW, Rodgers MR, Wesley IV, Johnoson CH, Tanner SA (1999) Isolation of Arcobacter butzleri from ground water. Lett Appl Microbiol 28:31–35
Sevda S, Dominguez-Benetton X, Vanbroekhoven K, Wever HD, Sreekrishnan TR (2013) High strength wastewater treatment accompanied by power generation using air cathode microbial fuel cell. Appl Energy 105:194–206
Sun G, Thygesen A, Ale MT, Mensah M, Poulsen F, Meyer AS (2014) The significance of the initiation process parameters and reactor design for maximizing the efficiency of microbial fuel cells. Appl Microbiol Biotechnol 98:2415–2427
Thomsen MH, Thygesen A, Thomsen AB (2009) Identification and characterization of fermentation inhibitors formed during hydrothermal treatment and following SSF of wheat straw. Appl Microbiol Biotechnol 83:447–455
Thygesen A, Poulsen FW, Min B, Angelidaki I, Thomsen AB (2009) The effect of different substrates and humic acid on power generation in microbial fuel cell operation. Bioresour Technol 100:1186–1191
Thygesen A, Thomsen AB, Possemiers S, Verstraete W (2010) Integration of microbial electrolysis Cells (MECs) in the biorefinery for production of ethanol, H2 and phenolics. Waste Biomass Valor 1:9–20
Thygesen A, Marzorati M, Boon N, Thomsen AB, Verstraete W (2011) Upgrading of straw hydrolysate for production of hydrogen and phenols in a microbial electrolysis cell (MEC). Appl Microbiol Biotechnol 89:855–865
Wagner M, Loy A, Nogueira R, Ulrike P, Lee N, Daims H (2002) Microbial community composition and function in wastewater treatment plants. Antonie Van Leeuwenhoek 81:665–680
Wang S, Huang L, Gan L, Quan X, Li N, Chen G, Lu L, Xing D, Yang F (2012) Combined effects of enrichment procedure and non-fermentable or fermentable co-substrate on performance and bacterial community for pentachlorophenol degradation in microbial fuel cells. Bioresour Technol 120:120–126
Zhang Y, Angelidaki I (2012) Innovative self-powered submersible microbial electrolysis cell (SMEC) for biohydrogen production from anaerobic reactors. Water Res 46:2727–2736
Zhang Y, Min B, Huang L, Angelidaki I (2011) Electricity generation and microbial community response to substrate changes in microbial fuel cell. Bioresour Technol 102:1166–1173
Acknowledgments
The authors are grateful to Danida Fellowship Centre, Denmark for supporting the research project (Biobased Electricity in Developing Countries, DFC No. 11–091 Risø). The financial support from China Scholarship Council (CSC) for Guotao Sun’s Ph.D. project is gratefully acknowledged. Tomas Fernqvist, Ingelis Larsen, and Annette Eva Jensen, DTU are thanked for analytical work.
Compliance with ethical standards
It is declared that this work has not been published elsewhere and that all ethical requirements are followed including avoidance of data manipulation and plagiarism. The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 118 kb)
Rights and permissions
About this article
Cite this article
Sun, G., Thygesen, A. & Meyer, A.S. Acetate is a superior substrate for microbial fuel cell initiation preceding bioethanol effluent utilization. Appl Microbiol Biotechnol 99, 4905–4915 (2015). https://doi.org/10.1007/s00253-015-6513-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-015-6513-5