Abstract
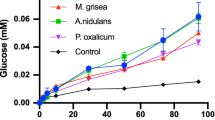
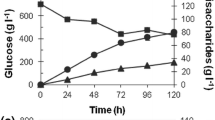
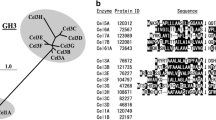
β-glucosidases (BGs) from Aspergillus fumigatus, Aspergillus niger, Aspergillus oryzae, Magnaporthe grisea, Neurospora crassa, and Penicillium brasilianum were purified to homogeneity, and investigated for their (simultaneous) hydrolytic and transglycosylation activity in samples with high concentrations of either cellobiose or glucose. The rate of the hydrolytic process (which converts one cellobiose to two glucose molecules) shows a maximum around 10–15 mM cellobiose and decreases with further increase in the concentration of substrate. At the highest investigated concentration (100 mM cellobiose), the hydrolytic activity for the different enzymes ranged from 10% to 55% of the maximum value. This decline in hydrolysis was essentially compensated by increased transglycosylation (which converts two cellobiose to one glucose and one trisaccharide). Hence, it was concluded that the hydrolytic slowdown at high substrate concentrations solely relies on an increased flow through the transglycosylation pathway and not an inhibition that delays the catalytic cycle. Transglycosylation was also detected at high product (glucose) concentrations, but in this case, it was not a major cause for the slowdown in hydrolysis. The experimental data was modeled to obtain kinetic parameters for both hydrolysis and transglycosylation. These parameters were subsequently used in calculations that quantified the negative effects on BG activity of respectively transglycosylation and product inhibition. The kinetic parameters and the mathematical method presented here allow estimation of these effects, and we suggest that this may be useful for the evaluation of BGs for industrial use.





Similar content being viewed by others
References
Baumann MJ, Murphy L, Lei N, Krogh KBRM, Borch K, Westh P (2011) Advantages of isothermal titration calorimetry for xylanase kinetics in comparison to chemical-reducing-end assays. Anal Biochem 410:19–26. doi:10.1016/j.ab.2010.11.001
Bhat MK, Bhat S (1997) Cellulose degrading enzymes and their potential industrial applications. Biotechnol Adv 15:583–620. doi:10.1016/S0734-9750(97)00006-2
Bhat KM, Gaikwad JS, Maheshwari R (1993) Purification and characterization of an extracellular β-glucosidase from the thermophilic fungus Sporotrichum thermophile and its influence on cellulase activity. J Gen Microbiol 139:2825–2832
Bhatia Y, Mishra S, Bisaria VS (2002) Microbial β-glucosidases: cloning, properties, and applications. Crit Rev Biotechnol 22:375–407. doi:10.1080/07388550290789568
Bock K, Pedersen C, Pedersen H (1984) C-13 nuclear magnetic-resonance data for oligosaccharides. Adv Carbohydr Chem Biochem 42:193–225. doi:10.1016/s0065-2318(08)60125-0
Bohlin C, Olsen SN, Morant MD, Patkar S, Borch K, Westh P (2010) A comparative study of activity and apparent inhibition of fungal β-glucosidases. Biotechnol Bioeng. doi:10.1002/bit.22885
Briggner LE, Wadsö I (1991) Test and calibration processes for microcalorimeters, with special reference to heat-conduction instruments used with aqueous systems. J Biochem Biophys Methods 22:101–118. doi:10.1016/0165-022X(91)90023-P
Calsavara LPV, Moraes FFD, Zanin GM (1999) Modeling cellobiose hydrolysis with integrated kinetic models. Appl Biochem Biotechnol 77–79:789–806
Chauve M, Mathis H, Huc D, Casanave D, Monot F,Ferreira NL (2010) Comparative kinetic analysis of two fungal beta-glucosidases. Biotechnol Biofuels 3 doi:10.1186/1754-6834-3-3
Chirico WJ, Brown RD (1987) Purification and characterization of a β-glucosidase from Trichoderma reesei. Eur J Biochem 165:333–341. doi:10.1111/j.1432-1033.1987.tb11446.x
Christakopoulos P, Goodenough PW, Kekos D, Macris BJ, Claeyssens M, Bhat MK (1994) Purification and characterization of an extracellular β-glucosidase with transglycosylation and exo-glucosidase activities from Fusarium oxysporum. Eur J Biochem 224:379–385. doi:10.1111/j.1432-1033.1994.00379.x
Cornish-Bowden A (2004) Fundamentals of enzyme kinetics, 3rd edn. Portland Press Ltd., London, UK
Crombie HJ, Chengappa S, Hellyer A, Reid JSG (1998) A xyloglucan oligosaccharide-active, transglycosylating beta-d-glucosidase from the cotyledons of nasturtium (Tropaeolum majus L) seedlings—purification, properties and characterization of a cDNA clone. Plant J 15:27–38. doi:10.1046/j.1365-313X.1998.00182.x
Decker CH, Visser J, Schreier P (2000) β-glucosidases from five black Aspergillus species: study of their physico-chemical and biocatalytic properties. J Agric Food Chem 48:4929–4936. doi:10.1021/jf000434d
Decker CH, Visser J, Schreier P (2001) β-glucosidase multiplicity from Aspergillus tubingensis CBS 643.92: purification and characterization of four β-glucosidases and their differentiation with respect to substrate specificity, glucose inhibition and acid tolerance. Appl Microbiol Biotechnol 55:157–163
Goldberg RN, Bell D, Tewari YB, McLaughlin MA (1991) Thermodynamics of hydrolysis of oligosaccharides. Biophys Chem 40:69–76. doi:10.1016/0301-4622(91)85030-T
Grous W, Converse A, Grethlein H, Lynd L (1985) Kinetics of cellobiose hydrolysis using cellobiase composites from Trichoderma reesei and Aspergillus niger. Biotechnol Bioeng 27:463–470. doi:10.1002/bit.260270411
Gueguen Y, Chemardin P, Arnaud A, Galzy P (1995) Purification and characterization of intracellular β-glucosidase from Botrytis cinerea. Enzyme Microb Technol 17:900–906. doi:10.1016/0141-0229(94)00143-F
Han YJ, Chen HZ (2008) Characterization of beta-glucosidase from corn stover and its application in simultaneous saccharification and fermentation. Bioresour Technol 99:6081–6087. doi:10.1016/j.biortech.2007.12.050
Harnpicharnchai P, Champreda V, Sornlake W, Eurwilaichitr L (2009) A thermotolerant β-glucosidase isolated from an endophytic fungi, Periconia sp., with a possible use for biomass conversion to sugars. Protein Expr Purif 67:61–69. doi:10.1016/j.pep.2008.05.022
Hong J, Ladisch MR, Gong CS, Wankat PC, Tsao GT (1981) Combined product and substrate-inhibition equation for cellobiase. Biotechnol Bioeng 23:2779–2788. doi:10.1002/bit.260231212
Jeoh T, Baker JO, Ali MK, Himmel ME, Adney WS (2005) Beta-d-glucosidase reaction kinetics from isothermal titration microcalorimetry. Anal Biochem 347:244–253. doi:10.1016/j.ab.2005.09.031
Kawai R, Igarashi K, Kitaoka M, Ishii T, Samejima M (2004) Kinetics of substrate transglycosylation by glycoside hydrolase family 3 glucan (1-->3)-[beta]-glucosidase from the white-rot fungus Phanerochaete chrysosporium. Carbohydr Res 339:2851–2857. doi:10.1016/j.carres.2004.09.019
Korotkova OG, Semenova MV, Morozova VV, Zorov IN, Sokolova LM, Bubnova TM, Okunev ON, Sinitsyn AP (2009) Isolation and properties of fungal β-glucosidases. Biochem Mosc 74:569–577. doi:10.1134/s0006297909050137
Krogh K, Harris PV, Olsen CL, Johansen KS, Hojer-Pedersen J, Börjesson J, Olsson L (2009) Characterization and kinetic analysis of a thermostable GH3 β-glucosidase from Penicillium brasilianum. Appl Microbiol Biotechnol 86:143–154. doi:10.1007/s00253-009-2181-7
Langston J, Sheehy N, Xu F (2006) Substrate specificity of Aspergillus oryzae family 3 β-glucosidase. Biochim Biophys Acta—Prot Proteom 1764:972–978. doi:10.1016/j.bbapap.2006.03.009
Lin J, Pillay B, Singh S (1999) Purification and biochemical characteristics of β-d-glucosidase from a thermophilic fungus, Thermomyces lanuginosus-SSBP. Biotechnol Appl Biochem 30:81–87
Lymar ES, Li B, Renganathan V (1995) Purification and characterization of a cellulose-binding β-glucosidsase from cellulose-degrading cultures of Phanerochaete chrysosporium. Appl Environ Microbiol 61:2976–2980
Montero MA, Romeu A (1992) Kinetic study on the β-glucosidase-catalyzed reaction of Trichoderam viride cellulase. Appl Microbiol Biotechnol 38:350–353
Murray P, Aro N, Collins C, Grassick A, Penttilä M, Saloheimo M, Tuohy M (2004) Expression in Trichoderma reesei and characterisation of a thermostable family 3 β-glucosidase from the moderately thermophilic fungus Talaromyces emersonii. Prot Expr Purif 38:248–257. doi:10.1016/j.pep.2004.08.006
Parry NJ, Beever DE, Owen E, Vandenberghe I, Van Beeumen J, Bhat MK (2001) Biochemical characterization and mechanism of action of a thermostable beta-glucosidase purified from Thermoascus aurantiacus. Biochem J 353:117–127
Perezpons JA, Cayetano A, Rebordosa X, Lloberas J, Guasch A, Querol E (1994) A β-glucosidase gene (BGL3) from Streptomyces sp. strain-QM-B814—molecular cloning, nucleotide-sequence, purification and characterization of the encoded enzyme, a new member of family 1 glycosyl hydrolases. Eur J Biochem 223:557–565. doi:10.1111/j.1432-1033.1994.tb19025.x
Riou C, Salmon JM, Vallier MJ, Gunata Z, Barre P (1998) Purification, characterization, and substrate specificity of a novel highly glucose-tolerant β-glucosidase from Aspergillus oryzae. Appl Environ Microbiol 64:3607–3614
Saha BC, Bothast RJ (1996) Production, purification, and characterization of a highly glucose-tolerant novel β-glucosidase from Candida peltata. Appl Environ Microbiol 62:3165–3170
Segel IH (1975) Enzyme kinetics: behavior and analysis og rapid equilibrium and steady-state enzyme systems. John Wiley & Sons Inc., New York
Seidle HF, Huber RE (2005) Transglucosidic reactions of the Aspergillus niger Family 3 beta-glucosidase: qualitative and quantitative analyses and evidence that the transglucosidic rate is independent of pH. Arch Biochem Biophys 436:254–264. doi:10.1016/j.abb.2005.02.017
Seidle HF, Marten I, Shoseyov O, Huber RE (2004) Physical and kinetic properties of the family 3 β-glucosidase from Aspergillus niger which is important for cellulose breakdown. Protein J 23:11–23. doi:10.1023/B:JOPC.0000016254.58189.2a
Seidle HF, Allison SJ, George E, Huber RE (2006) Trp-49 of the family 3 beta-glucosidase from Aspergillus niger is important for its transglucosidic activity: creation of novel beta-glucosidases with low transglucosidic efficiencies. Arch Biochem Biophys 455:110–118. doi:10.1016/j.abb.2006.09.016
Takahash K, Hiromi K, Ono S (1965) Calorimetric studies on hydrolysis of glucosides. 3. Heats of hydrolysis of alpha-1,4 glucosidic linkages involved in higer glucosides. J Biochem (Tokyo) 58:255
Tewari YB, Goldberg RN (1989) Thermodynamics of hydrolysis of disaccharides—cellobiose, gentiobiose, isomaltose, and maltose. J Biol Chem 264:3966–3971
Tewari YB, Lang BE, Decker SR, Goldberg RN (2008) Thermodynamics of the hydrolysis reactions of 1,4-beta-d-xylobiose, 1,4-beta-d-xylotriose, d-cellobiose, and d-maltose. J Chem Thermodyn 40:1517–1526. doi:10.1016/j.jct.2008.05.015
Wallecha A, Mishra S (2003) Purification and characterization of two beta-glucosidases from a thermo-tolerant yeast Pichia etchellsii. BBA-Prot Proteom 1649:74–84. doi:10.1016/S1570-9639(03)00163-8
Yan TR, Lin CL (1997) Purification and characterization of a glucose-tolerant beta-glucosidase from Aspergillus niger CCRC 31494. Biosci Biotechnol Biochem 61:965–970
Yan BX, Sun YQ (1997) Domain structure and conformation of a cellobiohydrolase from Trichoderma pseudokiningii S-38. J Protein Chem 16:59–66
Yeoh HH, Tan TK, Koh SK (1986) Kinetic properties of beta-glucosidase from Aspergillus ornatus. Appl Microbiol Biotechnol 25:25–28
Yoon JJ, Kim KY, Cha CJ (2008) Purification and characterization of thermostable beta-glucosidase from the brown-rot basidiomycete Fomitopsis palustris grown on microcrystalline cellulose. J Microbiol 46:51–55. doi:10.1007/s12275-007-0230-4
Zhang YHP, Lynd LR (2003) Cellodextrin preparation by mixed-acid hydrolysis and chromatographic separation. Anal Biochem 322:225–232. doi:10.1016/j.ab.2003.07.021
Acknowledgement
We thank Torben Lund and Jacob Krake for the help with the HILIC-ESI-MS setup. This work was supported by funds from the Danish Agency for Science, Technology, and Innovation (Grant no. 2104-07-0028) and the Department of Energy (Award Number DE-FC36-08GO18080).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
The online resource shows in detail how Eqs. 3 and 4 is derived and how SAS is used to obtain the maximum likelihood values of the kinetic parameters. It also shows how calculation of glucose, cellobiose, and triose as a function of time could be determined using a reaction scheme which includes hydrolysis and transglycosylation of cellobiose as well as hydrolysis of triose and product inhibition by glucose.
ESM 1
(DOC 588 kb)
Rights and permissions
About this article
Cite this article
Bohlin, C., Praestgaard, E., Baumann, M.J. et al. A comparative study of hydrolysis and transglycosylation activities of fungal β-glucosidases. Appl Microbiol Biotechnol 97, 159–169 (2013). https://doi.org/10.1007/s00253-012-3875-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-012-3875-9