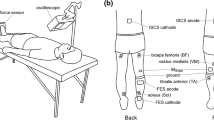
Abstract
The purpose of this study was to investigate short-term effects of walking with functional electrical stimulation (FES) on inhibitory and excitatory spinal reflexes in healthy subjects. The FES was applied to the common peroneal (CP) nerve during the swing phase of the step cycle when the ankle flexors are active. We have previously shown that corticospinal excitability for the tibialis anterior (TA) muscle increased after 30 min of FES-assisted walking. An increase of corticospinal excitability could be due to the changes in spinal and/or cortical excitability. Thus, we wished to examine whether a short-term application of FES would increase spinal motoneuronal excitability. Changes could also result from effects on inhibitory as well as excitatory pathways, but to our knowledge no studies have investigated short-term effects of FES on spinal inhibitory pathways. Therefore, we measured reciprocal and presynaptic inhibition, as well as reflex excitability, before and after FES-assisted walking. As controls, effects of FES-like stimulation at rest and walking without stimulation were tested in separate sessions. The TA H-reflex amplitude did not increase after FES in any of the conditions tested, so we have no evidence that FES increases spinal excitability for the TA. The soleus H-reflex decreased slightly (10%) after FES-assisted walking, and remained decreased for at least 30 min. However, the control experiment indicated that this decrease was associated with walking and not with stimulation. Thirty minutes of FES did not produce any significant effects on spinal inhibitory pathways examined in the present study. In conclusion, the soleus H-reflex showed a small but consistent decrease and no spinal circuits examined showed an increase, as was observed in the corticospinal excitability. Thus, we suggest that a short-term application of FES increases the excitability of the cortex or its connections to the spinal cord more effectively than that of spinal pathways.






Similar content being viewed by others
References
Ashby P, Wiens M (1989) Reciprocal inhibition following lesions of the spinal cord in man. J Physiol 414:145–157
Bajd T, Gregoric M, Vodovnik L, Benko H (1985) Electrical stimulation in treating spasticity resulting from spinal cord injury. Arch Phys Med Rehabil 66:515–517
Barbeau H, Ladouceur M, Mirbagheri MM, Kearney RE (2002) The effect of locomotor training combined with functional electrical stimulation in chronic spinal cord injured subjects: walking and reflex studies. Brain Res Rev 40:274–291
Barbeau H, Norman K, Fung J, Visintin M, Ladouceur M (1998) Does neurorehabilitation play a role in the recovery of walking in neurological populations? Ann N Y Acad Sci 860:377–392
Bennett DJ, Li Y, Harvey PJ, Gorassini M (2001a) Evidence for plateau potentials in tail motoneurons of awake chronic spinal rats with spasticity. J Neurophysiol 86:1972–1982
Bennett DJ, Li Y, Siu M (2001b) Plateau potentials in sacrocaudal motoneurons of chronic spinal rats, recorded in vitro. J Neurophysiol 86:1955–1971
Brown P (1994) Pathophysiology of spasticity. J Neurol Neurosurg Psychiatry 57:773–777
Burridge J, Wood D, Taylor P, Swain I, Hagan S (1997) The effect of common peroneal nerve stimulation on quadriceps spaciticity in hemiplegia. Physiotherapy 83:82–89
Capaday C, Cody FW, Stein RB (1990) Reciprocal inhibition of soleus motor output in humans during walking and voluntary tonic activity. J Neurophysiol 64:607–616
Capaday C, Lavoie BA, Comeau F (1995) Differential effects of a flexor nerve input on the human soleus H-reflex during standing versus walking. Can J Physiol Pharmacol 73:436–449
Crone C, Hultborn H, Jespersen B, Nielsen J (1987) Reciprocal Ia inhibition between ankle flexors and extensors in man. J Physiol 389:163–185
Crone C, Hultborn H, Mazieres L, Morin C, Nielsen J, Pierrot-Deseilligny E (1990) Sensitivity of monosynaptic test reflexes to facilitation and inhibition as a function of the test reflex size: a study in man and the cat. Exp Brain Res 81:35–45
Crone C, Nielsen J (1989) Methodological implications of the post activation depression of the soleus H-reflex in man. Exp Brain Res 78:28–32
Crone C, Nielsen J, Petersen N, Ballegaard M, Hultborn H (1994) Disynaptic reciprocal inhibition of ankle extensors in spastic patients. Brain 117:1161–1168
Delwaide PJ (1987) Spasticity: from pathophysiology to therapy. Acta Neurochir Suppl 39:91–95
Delwaide PJ, Oliver E (1988) Short-latency autogenic inhibition (IB inhibition) in human spasticity. J Neurol Neurosurg Psychiatry 51:1546–1550
Devanne H, Lavoie BA, Capaday C (1997) Input–output properties and gain changes in the human corticospinal pathway. Exp Brain Res 114:329–338
Dietz V (1997) Neurophysiology of gait disorders: present and future applications. Electroencephalogr Clin Neurophysiol 103:333–355
Dietz V (2002) Proprioception and locomotor disorders. Nat Rev Neurosci 3:781–790
Earles D, Vardaxis V, Koceja D (2001) Regulation of motor output between young and elderly subjects. Clin Neurophysiol 112:1273–1279
Faist M, Mazevet D, Dietz V, Pierrot-Deseilligny E (1994) A quantitative assessment of presynaptic inhibition of Ia afferents in spastics. Differences in hemiplegics and paraplegics. Brain 117:1449–1455
Gerrits HL, de Haan A, Sargeant AJ, Dallmeijer A, Hopman MT (2000) Altered contractile properties of the quadriceps muscle in people with spinal cord injury following functional electrical stimulated cycle training. Spinal Cord 38:214–223
Goulet C, Arsenault AB, Bourbonnais D, Laramee MT, Lepage Y (1996) Effects of transcutaneous electrical nerve stimulation on H-reflex and spinal spasticity. Scand J Rehabil Med 28:169–176
Granat MH, Ferguson AC, Andrews BJ, Delargy M (1993) The role of functional electrical stimulation in the rehabilitation of patients with incomplete spinal cord injury–observed benefits during gait studies. Paraplegia 31:207–215
Greve JM, Muszkat R, Schmidt B, Chiovatto J, Barros Filho TE, Batisttella LR (1993) Functional electrical stimulation (FES): muscle histochemical analysis. Paraplegia 31:764–770
Hultborn H (2003) Changes in neuronal properties and spinal reflexes during development of spasticity following spinal cord lesions and stroke: studies in animal models and patients. J Rehabil Med S41:46–55
Hultborn H, Illert M, Nielsen J, Paul A, Ballegaard M, Wiese H (1996) On the mechanism of the post-activation depression of the H-reflex in human subjects. Exp Brain Res 108:450–462
Joodaki MR, Olyaei GR, Bagheri H (2001) The effects of electrical nerve stimulation of the lower extremity on H-reflex and F-wave parameters. Electromyogr Clin Neurophysiol 41:23–28
Katz R, Pierrot-Deseilligny E (1982) Recurrent inhibition of alpha-motoneurons in patients with upper motor neuron lesions. Brain 105:103–124
Katz RT, Rymer WZ (1989) Spastic hypertonia: mechanisms and measurement. Arch Phys Med Rehabil 70:144–155
Khaslavskaia S, Ladouceur M, Sinkjaer T (2002) Increase in tibialis anterior motor cortex excitability following repetitive electrical stimulation of the common peroneal nerve. Exp Brain Res 145:309–315
Khaslavskaia S, Sinkjaer T (2005) Motor cortex excitability following repetitive electrical stimulation of the common peroneal nerve depends on the voluntary drive. Exp Brain Res 162:497–502
Kido A, Tanaka N, Stein RB (2004a) Spinal excitation and inhibition decrease as humans age. Can J Physiol Pharmacol 82:238–248
Kido A, Tanaka N, Stein RB (2004b) Spinal reciprocal inhibition in human locomotion. J Appl Physiol 96:1969–1977
Kido Thompson A, Stein RB (2004) Short-term effects of functional electrical stimulation on motor evokes potentials in ankle flexor and extensor muscles. Exp Brain Res 159:491–500
Knash ME, Kido A, Gorassini M, Chan KM, Stein RB (2003) Electrical stimulation of the human common peroneal nerve elicits lasting facilitation of cortical motor-evoked potentials. Exp Brain Res 153:366–377
Ladouceur M, Barbeau H (2000a) Functional electrical stimulation-assisted walking for persons with incomplete spinal injuries: changes in the kinematics and physiological cost of overground walking. Scand J Rehabil Med 32:72–79
Ladouceur M, Barbeau H (2000b) Functional electrical stimulation-assisted walking for persons with incomplete spinal injuries: longitudinal changes in maximal overground walking speed. Scand J Rehabil Med 32:28–36
Lavoie BA, Devanne H, Capaday C (1997) Differential control of reciprocal inhibition during walking versus postural and voluntary motor tasks in humans. J Neurophysiol 78:429–438
Levin MF, Hui-Chan CW (1992) Relief of hemiparetic spasticity by TENS is associated with improvement in reflex and voluntary motor functions. Electroencephalogr Clin Neurophysiol 85:131–142
Mao CC, Ashby P, Wang M, McCrea D (1984) Synaptic connections from large muscle afferents to the motoneurons of various leg muscles in man. Exp Brain Res 56:341–350
Meunier S, Kwon J, Cohen LG (2004) Intrinsically encoded spinal cord plasticity during performance of a visuomotor task. Soc Neurosci (Abstract: 418.411)
Meunier S, Pierrot-Deseilligny E (1998) Cortical control of presynaptic inhibition of Ia afferents in humans. Exp Brain Res 119:415–426
Mirbagheri MM, Ladouceur M, Barbeau H, Kearney RE (2002) The effects of long-term FES-assisted walking on intrinsic and reflex dynamic stiffness in spastic spinal-cord-injured subjects. IEEE Trans Neural Syst Rehabil Eng 10:280–289
Mizuno Y, Tanaka R, Yanagisawa N (1971) Reciprocal group I inhibition on triceps surae motoneurons in man. J Neurophysiol 34:1010–1017
Morita H, Crone C, Christenhuis D, Petersen NT, Nielsen JB (2001) Modulation of presynaptic inhibition and disynaptic reciprocal Ia inhibition during voluntary movement in spasticity. Brain 124:826–837
Nielsen J, Petersen N, Crone C (1995) Changes in transmission across synapses of Ia afferents in spastic patients. Brain 118:995–1004
O’Dwyer NJ, Ada L (1996) Reflex hyperexcitability and muscle contracture in relation to spastic hypertonia. Curr Opin Neurol 9:451–455
O’Dwyer NJ, Ada L, Neilson PD (1996) Spasticity and muscle contracture following stroke. Brain 119:1737–1749
Perez MA, Field-Fote EC, Floeter MK (2003) Patterned sensory stimulation induces plasticity in reciprocal ia inhibition in humans. J Neurosci 23:2014–2018
Potisk KP, Gregoric M, Vodovnik L (1995) Effects of transcutaneous electrical nerve stimulation (TENS) on spasticity in patients with hemiplegia. Scand J Rehabil Med 27:169–174
Rochester L, Barron MJ, Chandler CS, Sutton RA, Miller S, Johnson MA (1995) Influence of electrical stimulation of the tibialis anterior muscle in paraplegic subjects. 2. Morphological and histochemical properties. Paraplegia 33:514–522
Rushton DN (2003) Functional electrical stimulation and rehabilitation—an hypothesis. Med Eng Phys 25:75–78
Schmit BD, Benz EN (2002) Extensor reflexes in human spinal cord injury: activation by hip proprioceptors. Exp Brain Res 145:520–527
Schmit BD, Benz EN, Rymer WZ (2002) Afferent mechanisms for the reflex response to imposed ankle movement in chronic spinal cord injury. Exp Brain Res 145:40–49
Seib TP, Price R, Reyes MR, Lehmann JF (1994) The quantitative measurement of spasticity: effect of cutaneous electrical stimulation. Arch Phys Med Rehabil 75:746–750
Shefner JM, Berman SA, Sarkarati M, Young RR (1992) Recurrent inhibition is increased in patients with spinal cord injury. Neurology 42:2162–2168
Sinkjaer T, Toft E, Larsen K, Andreassen S, Hansen HJ (1993) Non-reflex and reflex mediated ankle joint stiffness in multiple sclerosis patients with spasticity. Muscle Nerve 16:69–76
Solomonow M, Reisin E, Aguilar E, Baratta RV, Best R, D’Ambrosia R (1997) Reciprocating gait orthosis powered with electrical muscle stimulation (RGO II). Part II: Medical evaluation of 70 paraplegic patients. Orthopedics 20:411–418
Stein RB (1998) Assembly for functional electrical stimulation. In: Continuation in part US patent 5814093
Stein RB, Belanger M, Wheeler G, Wieler M, Popovic DB, Prochazka A, Davis LA (1993) Electrical systems for improving locomotion after incomplete spinal cord injury: an assessment. Arch Phys Med Rehabil 74:954–959
Stein RB, Chong SL, James KB, Kido A, Bell GJ, Tubman LA, Belanger M (2002) Electrical stimulation for therapy and mobility after spinal cord injury. Prog Brain Res 137:27–34
Stein RB, Gordon T, Jefferson J, Sharfenberger A, Yang JF, de Zepetnek JT, Belanger M (1992) Optimal stimulation of paralyzed muscle after human spinal cord injury. J Appl Physiol 72:1393–1400
Tanaka R (1974) Reciprocal Ia inhibition during voluntary movements in man. Exp Brain Res 21:529–540
Taylor PN, Burridge JH, Dunkerley AL, Wood DE, Norton JA, Singleton C, Swain ID (1999) Clinical use of the Odstock dropped foot stimulator: its effect on the speed and effort of walking. Arch Phys Med Rehabil 80:1577–1583
Taylor S, Ashby P, Verrier M (1984) Neurophysiological changes following traumatic spinal lesions in man. J Neurol Neurosurg Psychiatry 47:1102–1108
Thilmann AF, Fellows SJ, Garms E (1991) The mechanism of spastic muscle hypertonus. Variation in reflex gain over the time course of spasticity. Brain 114:233–244
Wieler M, Stein RB, Ladouceur M, Whittaker M, Smith AW, Naaman S, Barbeau H, Bugaresti J, Aimone E (1999) Multicenter evaluation of electrical stimulation systems for walking. Arch Phys Med Rehabil 80:495–500
Yang JF, Fung J, Edamura M, Blunt R, Stein RB, Barbeau H (1991) H-reflex modulation during walking in spastic paretic subjects. Can J Neurol Sci 18:443–452
Zehr EP, Stein RB (1999) Interaction of the Jendrassik maneuver with segmental presynaptic inhibition. Exp Brain Res 124:474–480
Acknowledgements
We thank Dr. C. Capaday for helpful comments on the manuscript. The Canadian Institutes of Health Research and the Christopher Reeve Paralysis Foundation supported this research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Thompson, A.K., Doran, B. & Stein, R.B. Short-term effects of functional electrical stimulation on spinal excitatory and inhibitory reflexes in ankle extensor and flexor muscles. Exp Brain Res 170, 216–226 (2006). https://doi.org/10.1007/s00221-005-0203-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-005-0203-y