Abstract
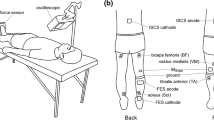
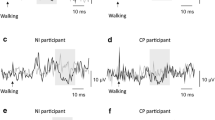
Stimulating sensory afferents can increase corticospinal excitability. Intensive use of a particular part of the body can also induce reorganization of neural circuits (use-dependent plasticity) in the central nervous system (CNS). What happens in the CNS when the nerve stimulation is applied in concert with the use of particular muscle groups? The purpose of this study was to investigate short-term effects of electrical stimulation of the common peroneal (CP) nerve during walking on motor-evoked potentials (MEPs) in the ankle flexors and extensors in healthy subjects. Since the stimulation was applied during the swing phase of the step cycle when the ankle flexors are active, this is referred to as functional electrical stimulation (FES). The following questions were addressed: (1) can FES during walking increase corticospinal excitability more effectively than passively received repetitive nerve stimulation and (2) does walking itself improve the descending connection. FES was delivered using a foot drop stimulator that activates ankle dorsiflexors during the swing phase of the step cycle. MEPs in the tibialis anterior (TA) and soleus muscles were measured before, between, and after periods of walking with or without FES, using transcranial magnetic stimulation. After 30 min of walking with FES, the half-maximum peak-to-peak MEP (MEPh) in the TA increased in amplitude and this facilitatory effect lasted for at least 30 min. In contrast, walking had no effects on the TA MEPh without FES. The increase in the TA MEPh with FES (~40%) was similar to that with repetitive CP nerve stimulation at rest. The soleus MEPh was also increased after walking with FES, but not without FES, which differs from the previous observation with CP nerve stimulation at rest. With FES, the TA silent period at MEPh was unchanged or slightly decreased, while it increased after walking without FES. Increased cortical excitability accompanied by unchanged cortical inhibition (no changes in the silent period with FES) suggests that FES did not simply increase general excitability of the cortex, but had specific effects on particular cortical neurons.





Similar content being viewed by others
References
Barbeau H, Visintin M (2003) Optimal outcomes obtained with body-weight support combined with treadmill training in stroke subjects. Arch Phys Med Rehabil 84:1458–1465
Barbeau H, Norman K, Fung J, Visintin M, Ladouceur M (1998) Does neurorehabilitation play a role in the recovery of walking in neurological populations? Ann N Y Acad Sci 860:377–392
Barbeau H, Ladouceur M, Mirbagheri MM, Kearney RE (2002) The effect of locomotor training combined with functional electrical stimulation in chronic spinal cord injured subjects: walking and reflex studies. Brain Res Brain Res Rev 40:274–291
Belanger M, Stein RB, Wheeler GD, Gordon T, Leduc B (2000) Electrical stimulation: can it increase muscle strength and reverse osteopenia in spinal cord injured individuals? Arch Phys Med Rehabil 81:1090–1098
Bertasi V, Bertolasi L, Frasson E, Priori A (2000) The excitability of human cortical inhibitory circuits responsible for the muscle silent period after transcranial brain stimulation. Exp Brain Res 132:384–389
Brasil-Neto JP, Valls-Sole J, Pascual-Leone A, Cammarota A, Amassian VE, Cracco R, Maccabee P, Cracco J, Hallett M, Cohen LG (1993) Rapid modulation of human cortical motor outputs following ischaemic nerve block. Brain 116:511–525
Burridge JH, Taylor PN, Hagan SA, Wood DE, Swain ID (1997) The effects of common peroneal stimulation on the effort and speed of walking: a randomized controlled trial with chronic hemiplegic patients. Clin Rehabil 11:201–210
Butefisch CM, Davis BC, Wise SP, Sawaki L, Kopylev L, Classen J, Cohen LG (2000) Mechanisms of use-dependent plasticity in the human motor cortex. Proc Natl Acad Sci U S A 97:3661–3665
Charlton CS, Ridding MC, Thompson PD, Miles TS (2003) Prolonged peripheral nerve stimulation induces persistent changes in excitability of human motor cortex. J Neurol Sci 208:79–85
Chen R, Lozano AM, Ashby P (1999) Mechanism of the silent period following transcranial magnetic stimulation. Evidence from epidural recordings. Exp Brain Res 128:539–542
Chen R, Cohen LG, Hallett M (2002) Nervous system reorganization following injury. Neuroscience 111:761–773
Colombo G, Wirz M, Dietz V (1998) Effect of locomotor training related to clinical and electrophysiological examinations in spinal cord injured humans. Ann N Y Acad Sci 860:536–538
Devanne H, Lavoie BA, Capaday C (1997) Input-output properties and gain changes in the human corticospinal pathway. Exp Brain Res 114:329–338
Dietz V, Duysens J (2000) Significance of load receptor input during locomotion: a review. Gait Posture 11:102–110
Dietz V, Colombo G, Jensen L (1994) Locomotor activity in spinal man. Lancet 344:1260–1263
Dietz V, Colombo G, Jensen L, Baumgartner L (1995) Locomotor capacity of spinal cord in paraplegic patients. Ann Neurol 37:574–582
Dietz V, Wirz M, Jensen L (1997) Locomotion in patients with spinal cord injuries. Phys Ther 77:508–516
Dietz V, Wirz M, Colombo G, Curt A (1998) Locomotor capacity and recovery of spinal cord function in paraplegic patients: a clinical and electrophysiological evaluation. Electroencephalogr Clin Neurophysiol 109:140–153
Dobkin BH, Apple D, Barbeau H, Basso M, Behrman A, Deforge D, Ditunno J, Dudley G, Elashoff R, Fugate L, Harkema S, Saulino M, Scott M (2003) Methods for a randomized trial of weight-supported treadmill training versus conventional training for walking during inpatient rehabilitation after incomplete traumatic spinal cord injury. Neurorehabil Neural Repair 17:153–167
Drew T, Jiang W, Widajewicz W (2002) Contributions of the motor cortex to the control of the hindlimbs during locomotion in the cat. Brain Res Brain Res Rev 40:178–191
Fuhr P, Agostino R, Hallett M (1991) Spinal motor neuron excitability during the silent period after cortical stimulation. Electroencephalogr Clin Neurophysiol 81:257–262
Garvey MA, Ziemann U, Becker DA, Barker CA, Bartko JJ (2001) New graphical method to measure silent periods evoked by transcranial magnetic stimulation. Clin Neurophysiol 112:1451–1460
Gerrits HL, de Haan A, Sargeant AJ, Dallmeijer A, Hopman MT (2000) Altered contractile properties of the quadriceps muscle in people with spinal cord injury following functional electrical stimulated cycle training. Spinal Cord 38:214–223
Hamdy S, Rothwell JC, Aziz Q, Singh KD, Thompson DG (1998) Long-term reorganization of human motor cortex driven by short-term sensory stimulation. Nat Neurosci 1:64–68
Ikeda A, Ohara S, Matsumoto R, Kunieda T, Nagamine T, Miyamoto S, Kohara N, Taki W, Hashimoto N, Shibasaki H (2000) Role of primary sensorimotor cortices in generating inhibitory motor response in humans. Brain 123:1710–1721
Jacobs KM, Donoghue JP (1991) Reshaping the cortical motor map by unmasking latent intracortical connections. Science 251:944–947
Jiang W, Drew T (1996) Effects of bilateral lesions of the dorsolateral funiculi and dorsal columns at the level of the low thoracic spinal cord on the control of locomotion in the adult cat. I. Treadmill walking. J Neurophysiol 76:849–866
Kably B, Drew T (1998) Corticoreticular pathways in the cat. II. Discharge activity of neurons in area 4 during voluntary gait modifications. J Neurophysiol 80:406–424
Kaelin-Lang A, Luft AR, Sawaki L, Burstein AH, Sohn YH, Cohen LG (2002) Modulation of human corticomotor excitability by somatosensory input. J Physiol 540:623–633
Keck ME, Pijnappels M, Schubert M, Colombo G, Curt A, Dietz V (1998) Stumbling reactions in man: influence of corticospinal input. Electroencephalogr Clin Neurophysiol 109:215–223
Khaslavskaia S, Ladouceur M, Sinkjaer T (2002) Increase in tibialis anterior motor cortex excitability following repetitive electrical stimulation of the common peroneal nerve. Exp Brain Res 145:309–315
Knash ME, Kido A, Gorassini M, Chan KM, Stein RB (2003) Electrical stimulation of the human common peroneal nerve elicits lasting facilitation of cortical motor-evoked potentials. Exp Brain Res 153:366–377
Ladouceur M, Barbeau H (2000a) Functional electrical stimulation-assisted walking for persons with incomplete spinal injuries: changes in the kinematics and physiological cost of overground walking. Scand J Rehabil Med 32:72–79
Ladouceur M, Barbeau H (2000b) Functional electrical stimulation-assisted walking for persons with incomplete spinal injuries: longitudinal changes in maximal overground walking speed. Scand J Rehabil Med 32:28–36
Levy LM, Ziemann U, Chen R, Cohen LG (2002) Rapid modulation of GABA in sensorimotor cortex induced by acute deafferentation. Ann Neurol 52:755–761
Liepert J, Graef S, Uhde I, Leidner O, Weiller C (2000) Training-induced changes of motor cortex representations in stroke patients. Acta Neurol Scand 101:321–326
Maegele M, Muller S, Wernig A, Edgerton VR, Harkema SJ (2002) Recruitment of spinal motor pools during voluntary movements versus stepping after human spinal cord injury. J Neurotrauma 19:1217–1229
Malouin F, Richards CL, Jackson PL, Dumas F, Doyon J (2003) Brain activations during motor imagery of locomotor-related tasks: a PET study. Hum Brain Mapp 19:47–62
Martin TP, Stein RB, Hoeppner PH, Reid DC (1992) Influence of electrical stimulation on the morphological and metabolic properties of paralyzed muscle. J Appl Physiol 72:1401–1406
McKay DR, Ridding MC, Thompson PD, Miles TS (2002) Induction of persistent changes in the organisation of the human motor cortex. Exp Brain Res 143:342–349
Nathan PW (1994) Effects on movement of surgical incisions into the human spinal cord. Brain 117:337–346
Pascual-Leone A, Davey NJ, Rothwell J, Wassermann EM, Puri BK (2002) Handbook of transcranial magnetic stimulation. Arnold, London
Petersen NT, Butler JE, Marchand-Pauvert V, Fisher R, Ledebt A, Pyndt HS, Hansen NL, Nielsen JB (2001) Suppression of EMG activity by transcranial magnetic stimulation in human subjects during walking. J Physiol 537:651–656
Ridding MC, Rothwell JC (1995) Reorganisation in human motor cortex. Can J Physiol Pharmacol 73:218–222
Ridding MC, Rothwell JC (1999) Afferent input and cortical organisation: a study with magnetic stimulation. Exp Brain Res 126:536–544
Ridding MC, Taylor JL (2001) Mechanisms of motor-evoked potential facilitation following prolonged dual peripheral and central stimulation in humans. J Physiol 537:623–631
Ridding MC, Brouwer B, Miles TS, Pitcher JB, Thompson PD (2000) Changes in muscle responses to stimulation of the motor cortex induced by peripheral nerve stimulation in human subjects. Exp Brain Res 131:135–143
Ridding MC, McKay DR, Thompson PD, Miles TS (2001) Changes in corticomotor representations induced by prolonged peripheral nerve stimulation in humans. Clin Neurophysiol 112:1461–1469
Sacco P, Thickbroom GW, Thompson ML, Mastaglia FL (1997) Changes in corticomotor excitation and inhibition during prolonged submaximal muscle contractions. Muscle Nerve 20:1158–1166
Schubert M, Curt A, Jensen L, Dietz V (1997) Corticospinal input in human gait: modulation of magnetically evoked motor responses. Exp Brain Res 115:234–246
Schubert M, Wohlfarth K, Rollnik JD, Dengler R (1998) Walking and fatigue in multiple sclerosis: the role of the corticospinal system. Muscle Nerve 21:1068–1070
Schubert M, Curt A, Colombo G, Berger W, Dietz V (1999) Voluntary control of human gait: conditioning of magnetically evoked motor responses in a precision stepping task. Exp Brain Res 126:583–588
Shimizu T, Hino T, Komori T, Hirai S (2000) Loss of the muscle silent period evoked by transcranial magnetic stimulation of the motor cortex in patients with cervical cord lesions. Neurosci Lett 286:199–202
Sinkjaer T, Khaslavskaia S (2002) Tibialis anterior motor cortex excitability following repetitive electrical stimulation (rES) of CP nerve depends on the voluntary motor control drive during the time of rES. Soc Neurosci Abstr 562:4
Solomonow M, Reisin E, Aguilar E, Baratta RV, Best R, D’Ambrosia R (1997) Reciprocating gait orthosis powered with electrical muscle stimulation (RGO II). II. Medical evaluation of 70 paraplegic patients. Orthopedics 20:411–418
Stefan K, Kunesch E, Cohen LG, Benecke R, Classen J (2000) Induction of plasticity in the human motor cortex by paired associative stimulation. Brain 123:572–584
Stefan K, Kunesch E, Benecke R, Cohen LG, Classen J (2002) Mechanisms of enhancement of human motor cortex excitability induced by interventional paired associative stimulation. J Physiol 543:699–708
Stein RB (1998) Assembly for functional electrical stimulation. Continuation in part. US patent 5814093
Stein RB, Gordon T, Jefferson J, Sharfenberger A, Yang JF, de Zepetnek JT, Belanger M (1992) Optimal stimulation of paralyzed muscle after human spinal cord injury. J Appl Physiol 72:1393–1400
Stein RB, Belanger M, Wheeler G, Wieler M, Popovic DB, Prochazka A, Davis LA (1993) Electrical systems for improving locomotion after incomplete spinal cord injury: an assessment. Arch Phys Med Rehabil 74:954–959
Taylor PN, Burridge JH, Dunkerley AL, Wood DE, Norton JA, Singleton C, Swain ID (1999) Clinical use of the Odstock dropped foot stimulator: its effect on the speed and effort of walking. Arch Phys Med Rehabil 80:1577–1583
Trompetto C, Buccolieri A, Marinelli L, Abbruzzese G (2001) Differential modulation of motor evoked potential and silent period by activation of intracortical inhibitory circuits. Clin Neurophysiol 112:1822–1827
Weber M, Eisen AA (2002) Magnetic stimulation of the central and peripheral nervous systems. Muscle Nerve 25:160–175
Werhahn KJ, Mortensen J, Kaelin-Lang A, Boroojerdi B, Cohen LG (2002) Cortical excitability changes induced by deafferentation of the contralateral hemisphere. Brain 125:1402–1413
Wernig A, Muller S (1992) Laufband locomotion with body weight support improved walking in persons with severe spinal cord injuries. Paraplegia 30:229–238
Wernig A, Muller S, Nanassy A, Cagol E (1995) Laufband therapy based on ‘rules of spinal locomotion’ is effective in spinal cord injured persons. Eur J Neurosci 7:823–829
Wernig A, Nanassy A, Muller S (1999) Laufband (treadmill) therapy in incomplete paraplegia and tetraplegia. J Neurotrauma 16:719–726
Wieler M, Stein RB, Ladouceur M, Whittaker M, Smith AW, Naaman S, Barbeau H, Bugaresti J, Aimone E (1999) Multicenter evaluation of electrical stimulation systems for walking. Arch Phys Med Rehabil 80:495–500
Wirz M, Colombo G, Dietz V (2001) Long term effects of locomotor training in spinal humans. J Neurol Neurosurg Psychiatry 71:93–96
Wu T, Sommer M, Tergau F, Paulus W (2000) Modification of the silent period by double transcranial magnetic stimulation. Clin Neurophysiol 111:1868–1872
Ziemann U, Netz J, Szelenyi A, Homberg V (1993) Spinal and supraspinal mechanisms contribute to the silent period in the contracting soleus muscle after transcranial magnetic stimulation of human motor cortex. Neurosci Lett 156:167–171
Ziemann U, Rothwell JC, Ridding MC (1996) Interaction between intracortical inhibition and facilitation in human motor cortex. J Physiol 496:873–881
Ziemann U, Corwell B, Cohen LG (1998a) Modulation of plasticity in human motor cortex after forearm ischemic nerve block. J Neurosci 18:1115–1123
Ziemann U, Hallett M, Cohen LG (1998b) Mechanisms of deafferentation-induced plasticity in human motor cortex. J Neurosci 18:7000–7007
Ziemann U, Muellbacher W, Hallett M, Cohen LG (2001) Modulation of practice-dependent plasticity in human motor cortex. Brain 124:1171–1181
Acknowledgements
We thank Dr. Monica Gorassini for helpful comments on the manuscript. This work was supported by the Canadian Institutes of Health Research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kido Thompson, A., Stein, R.B. Short-term effects of functional electrical stimulation on motor-evoked potentials in ankle flexor and extensor muscles. Exp Brain Res 159, 491–500 (2004). https://doi.org/10.1007/s00221-004-1972-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-004-1972-4